All published articles of this journal are available on ScienceDirect.
Pleural Infections: Antimicrobial Susceptibility Patterns of Bacterial Isolates and Associated Factors in Suspected Hospitalized Patients at Arba Minch General Hospital, Southern Ethiopia
Abstract
Background:
Pleural infection remains a clinical problem with high morbidity and mortality all over the world. Antimicrobial options are uncertain due to the lack of understanding of the bacteriology of pleural infection. Thus, the goal of this study was to determine bacteriological profiles, antimicrobial susceptibility patterns, and factors associated with pleural infection.
Methods:
An institution-based cross-sectional study was undertaken in Arba Minch General Hospital from 1st January to 30th November 2020. Socio-demographic, clinical, and behavioral data were collected using a pre-tested, structured questionnaire. Adducted amounts of pleural fluid samples were collected aseptically with sterile test tubes. Standard procedures were utilized for bacterial identification, and antimicrobial susceptibility testing was done using Kirby–Disk Bauer's diffusion method. Version 25 of the Statistical Package for the Social Sciences (SPSS) was used to analyze the data. The p-value of 0.05 was considered statistically significant.
Results:
A total of 152 hospitalized patients were enrolled in this study. The overall magnitude of bacterial pleural infection was 27.6%. The predominant bacteria isolated were Staphylococcus aureus with 34.9%, followed by Escherichia coli with 11.6%. Multidrug-resistant (MDR) isolates account for around 48.8% of all isolates, with Gram-positive bacterial isolates accounting for 71.4%. Methicillin-resistant was found in 26.7% and 33.3% of isolated Staphylococcus aureus (MRSA) and coagulase-negative staphylococci (CoNS), respectively. One isolate of Enterococcus spp. was found to be vancomycin-resistant. All isolated Streptococcus pneumoniae and Streptococcus pyogenes were susceptible to penicillin, erythromycin, cotrimoxazole, and vancomycin. Likewise, isolated Pseudomonas aeruginosa was susceptible to piperacillin. The pleural infection showed a statistically significant association with age group less than or equal to 25 years old (p<0.001), hospital admission for more than seven days (p<0.007), chest tube drainage (p<0.021), previous history of pneumonia (p<0.029) and habit of alcohol drinking (p<0.029).
Conclusion:
The overall rate of culture-confirmed pleural infection was high, and a considerable percentage of bacteria isolates showed increased resistant to routinely used antibiotics, indicating that more attention is needed to follow the spread and emergence of drug-resistant bacterial agents.
1. INTRODUCTION
Pleural infections are still a common clinical problem that affects people of all ages, genders, and ethnicities, with a particularly high frequency in children and the elderly [1-3]. Despite modern healthcare and improvements in medicine, this infection has remained a significant clinical concern in both developed and developing countries in the 21st century [4, 5]. About 15% of individuals with pleural infections who seek medical attention are in serious condition [6]. Pleuritic chest discomfort, cough, purulent sputum, fever, chills, weight loss, anorexia, dyspnea, and night sweats are all symptoms of pleural infection [5-7]. Pleural infection can either be confined or spread across the pleural cavity, resulting in “parapneumonic effusion” [2, 6].
The global rate of bacterial-related pleural infection has grown from 7.6% to 14.9%, with a 20% mortality rate [5, 8]. In elderly or chronically sick individuals, the mortality risk for bacterial-related pleural infection ranges from 25% to 75% [5, 6, 9, 10]. The most common causes of infection are bacterial invasion into the sterile pleural space or trans-diaphragmatic spreads of abdominal infection, and it is strongly associated with complications of hospital and community-acquired pneumonia, thoracic injuries, chest trauma, chest tube drainage, and postsurgical complications, immune-compromised states, and other behavioral factors (alcohol misuse and cigarette smoking) [11-15]. More than 20%–57% of patients with bacterial pneumonia admitted to the hospital develop parapneumonic effusions, which are associated with a 15%–20% mortality risk [6, 8, 9, 16]. Pleural infections have been related to 30% of thoracic surgical treatment problems, and 15% to 40% of pleural infections require surgical intervention, raising the risk of complications [17].
In developing countries, pleural infections are more commonly caused by Staphylococcus aureus, Streptococcus pneumoniae, Streptococcus pyogenes, Escherichia coli, Klebsiella species, Pseudomonas species, and Haemophilus influenza [1, 18-20]. These microbes' ability to colonize the body surface of a compromised host rapidly after being displaced, as well as their ability to survive in the community and hospital environment by developing antimicrobial and disinfectant resistance, may explain their role as etiological agents in pleural infections [18, 19].
In the early stages of pleural infections, antimicrobial therapy with or without chest tube drainage remains the treatment of choice [1, 18]. However, the diagnosis and treatment of pleural infections have become a severe challenge in clinical practice due to the increasing rise of antibiotic resistance in bacterial populations [7, 8]. Furthermore, the emergence of multidrug-resistant bacteria exacerbates the situation, and it has become a serious problem, especially in developing countries, as a result of overcrowding in hospitals, irrational prescription of more focused and less broad-spectrum antibiotics, an increasing trend of self-medication, and a lack of infection prevention programs [20-24]. The infectious organism, on the other hand, must be identified as rapidly as possible, and the therapies to which it is vulnerable must be implemented [1-8, 20, 23].
In Ethiopia, as its bacteriology is poorly described, antibiotic drug choices for pleural infection are uncertain [20, 24]. In the study area, patients with clinical signs and symptoms of pleural infection are treated empirically with broad-spectrum antibiotics without a culture report because of the absence of a microbiological laboratory in the hospital setting. However, determining the actual etiologic agent as well as its antimicrobial susceptibility profile is crucial for better treatment and drug resistance prevention. The goal of this study was to determine the bacteriological profile and antimicrobial susceptibility pattern of isolates, as well as their associated factors, among hospitalized pleural infection suspects.
2. MATERIALS AND METHODS
2.1. Study Area, Design, Period, and Study Population
The study was carried out at Arba Minch General Hospital, southern Ethiopia, from 1st January to 30th November 2020. Arba Minch is located in Ethiopia's southern region, 454 kilometers from Addis Ababa, the country's capital. The hospital has currently been delivering its health care services in the medical, surgical, gynecological, and pediatric wards to more than 1.5 million people from Gamo and other nearby zones. A cross-sectional study was conducted to determine the bacteriological profile and antimicrobial susceptibility patterns of isolates and their associated factors among pleural infection suspected hospitalized patients at Arba Minch General Hospital, Southern Ethiopia. The study comprised 152 patients with pleural effusion who were admitted to Arba Minch General Hospital's medical, surgical, and pediatric wards with at least three of the following pleural infection signs and symptoms: fever, chest discomfort, cough, purulent sputum, and dyspnea [5]. Patients who were unable to give complete responses to questionnaires (severely ill patients) during the collection period were excluded.
2.2. Sample Size and Sampling Procedure
The sample size was calculated using a single population proportion. A P value of 0.237 was chosen from the previous study done in Addis Ababa, Ethiopia [ 20 ], by considering only pleural infection. After considering 95% of the confidence interval (Z = 1.96) and 5% of marginal error (d = 0.05), the initial sample size was 278. Because the source population was 10,000 and the annual admission case was extremely rare, the sample size was reduced to 145 using a correction formula, and the final sample size was consolidated to 152 by computing a 5% non-response rate. The study participants were consecutively enrolled until the final sample size was achieved.
2.3. Data Collection and Laboratory Procedures
2.3.1. Data Collection
Two well-trained clinical nurses and one physician data collector aided the data collection. Data on socio-demographic (age, sex, residence, level of education, and occupation) and clinical and behavioral factors (history of thoracic surgery, hospital admission period, chest tube drainage, history of pneumonia, underlying disease (HIV/AIDS status, and Hepatitis B or C status), body mass index (BMI), cigarette smoking, and alcohol drinking) were collected through a structured face-to-face interview along with cross-checking of their medical records. Chest radiography has also been considered the initial screening tool for pleural infections and the presence of pleural effusion. As a result, chest radiography typically reveals non–free-flowing effusion. For instance, the presence of the “D sign” suggests the diagnosis in the correct clinical setting, and it should be noted that chest radiography misses >10% of significant parapneumonic effusions (i.e., ≥10 mm in depth), particularly in the setting of a lower-lobe parenchymal consolidation [5, 7].
2.3.2. Sample Collection and Processing
Using a sterile test tube, the trained physician aseptically collected about 2-3 mL of pleural fluid from each clinically suspected patient (n=152). Within one hour of collection, the fluid samples were transported to the Microbiology and Parasitology Laboratory at Arba Minch University College of Medicine and Health Sciences and processed for microbiological analysis. As a preliminary result, primary Gram staining, acid-fast staining, and differential WBC count were performed directly from collected fluids until culture data was obtained.
2.4. Culture and Identification
The pleural fluid samples were primarily inoculated onto 5% sheep blood agar (HiMedia Pvt. Ltd), chocolate agar (HiMedia Pvt. Ltd), and MacConkey agar (HiMedia Pvt. Ltd) and then incubated aerobically at 37°C for 48 hours, while the inoculated chocolate agar was incubated at 35–37°C for 72 hours in a candle jar to provide a 5–10% CO2 concentration. After incubation, pure cultures of bacterial isolates were subsequently subjected to species identification and confirmation. By adopting standard laboratory methods [25], biochemical (coagulase (Remel Europe Ltd, Dartford, UK), catalase (Remel Europe Ltd, Dartford, UK), Bile esculin test (HiMedia laboratory Pvt. Ltd, Mumbai, India), Bacitracin and optochin (all were from Oxoid Ltd, Basingstoke, England), Triple Sugar Iron (HiMedia laboratory Pvt. Ltd, Mumbai-India), Sulphur Indole Motility (SIM) (HiMedia laboratory Pvt. Ltd, Mumbai, India), Citrate (HiMedia laboratory Pvt. Ltd, Mumbai, India), Urea (HiMedia laboratory Pvt Ltd, Mumbai, India), and Oxidase (Remel Europe Ltd, Dartford, UK)), colony characterization and Gram reaction properties of bacterial isolates were determined.
2.5. Antimicrobial Susceptibility Testing
An antimicrobial susceptibility test was performed on Mueller Hinton agar by using Kirby-Bauer disk diffusion methods using the following antibiotics (Oxoid Ltd, Basingstoke, Hampshire, UK) as per Clinical and Laboratory Standards Institute (CLSI) guidelines [26]: Gram-positive bacteria isolates were tested by penicillin (10μg), cefoxitin (30μg), gentamicin (10μg), erythromycin (15μg), tetracycline (30 μg), doxycycline (30μg), ciprofloxacin (5μg), clindamycin (10μg), cotrimoxazole (25μg), vancomycin (30μg), and chloramphenicol (30μg) antibiotics; while Gram-negative bacteria isolates were tested with ampicillin (10μg), piperacillin (100μg), cefoxitin (30μg), cefepime (30μg), ceftriaxone (5μg), ceftazidime (30μg), meropenem (10μg), gentamicin (10μg), tetracycline (30 μg), doxycycline (30μg), ciprofloxacin (5μg), and chloramphenicol (30μg) antibiotics. Then, after overnight incubation, the diameter of the zone of inhibition was measured in millimeters and reported as susceptible, intermediate, or resistant as per the CLSI guidelines. Then, after overnight incubation, the diameter of the zone of inhibition was measured in millimeters and reported as susceptible, intermediate, or resistant as per the CLSI guidelines. Isolated bacteria that resist at least one antimicrobial drug in three or more antimicrobial categories are defined as multidrug resistant [26, 27].
2.6. Quality Management
Data quality was assured by closely following standard operating procedures and providing proper training for data and sample collectors. Culture media were prepared according to the manufacturer's instructions, and the sterility of the culture media was determined by incubating 5% of the batch at 35–37 °C overnight. If the selected media from the batches showed growth, all the bathes were discarded and prepared again aseptically. The performance of the prepared culture media was checked by inoculating the reference strains, Escherichia coli ATCC 25922 and Staphylococcus aureus ATCC 25923, obtained from the Ethiopian Public Health Institute. The potency of antibiotic discs was checked by using Enterococcus faecalis ATCC 29122 as per standard [26]. The overall procedure was supervised daily, and 5% of the questionnaire was pretested and validated before the actual data collection.
2.7. Statistical Analysis
The Statistical Package for Social Sciences (SPSS) software version 25 was used to analyze the data. Descriptive statistics like frequency, mean, and percentage were calculated. Binary logistic regression analysis was used to assess factors associated with pleural infection. Variables with a p-value <0.25 in the bivariable analysis were selected as candidates for the multivariable analysis and fitted into a logistic regression model. Finally, variables with a p-value < 0.05 were considered statistically significant.
2.8. Ethics Approval and Consent to Participate
The study was ethically authorized by the Arba Minch University College of Medicine and Health Sciences Institutional Review Board (Ref. No. CMHS/12033993/11). Besides, Arba Minch General Hospital granted permission. The purpose of the study and the information's confidentiality were both disclosed to the participants. Before the interview, written consent and assent were obtained from the respective participants. Confidentiality was strictly conserved from sample collection up to the final report writing.
2.9. Operational Definitions
Pleural Infection: If there is any growth of pathogenic aerobic bacteria on either of the culture media after overnight incubation of pleural fluid samples
Previous History of Thoracic Surgery: History of thoracic surgical procedures for the last year.
Previous History of Chest Tube Drainage: Patients with a clinical history of chest tube drainage for the last six months.
Previous History of Pneumonia: Patients with a clinical history of pneumonia for the last three months.
Alcoholic Patient: Patient with a habit of drinking alcohol at least three times a week, regardless of the type of alcohol.
Multidrug-Resistant: Bacterial isolates resistant to at least one antimicrobial drug in three or more classes of antibiotics.
3. RESULTS
3.1. Socio-demographic Characteristics
A total of 152 patients with suspicion of pleural infection participated in the study. Of those, 59.2% of the study participants were males. The study participants ranged in age from 11 to 73 years old, with a mean age of 32 years old and an SD of ± 18.6. The majority of the study participants (61.2%) were rural dwellers, and only 7.2% of the participants were illiterate. In occupational terms, 30.3% of the study participants were merchants (Table 1).
3.2. Diversity of Bacteria Isolated from Pleural Fluids
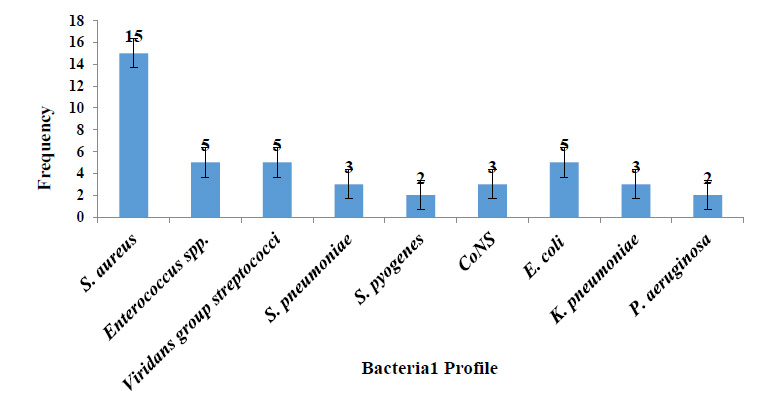
Among the 152 pleural fluid samples subjected to aerobic bacteriological culture, bacterial growth was found in 27.6% (42/152), with one male patient having a mixed infection (Staphylococcus aureus and Klebsiella pneumoniae), bringing the total number of bacteria isolates to forty-three. From the pleural fluid samples, nine species of bacteria isolates with a ratio of 3.3:1 of Gram-positive and Gram-negative bacteria were identified. The predominant isolated bacteria were Staphylococcus aureus with 34.9% (15/43), followed by Enterococcus spp. with 11.6% (5/43), Viridans group streptococci (VGS) with 11.6% (5/43), and Escherichia coli with 11.6% (5/43) (Fig. 1). The preliminary Gram stain yielded 81% (34/42) positive results compared to culture-positive results (42). All the culture-positive pleural fluids had a WBC count of over 21 cells/mm3 with dominant polymorphic differential characteristics. There was no acid-fast positive pleural fluid found overall.
3.3. Socio-Demographic Factors Associated with Pleural Infection
There was no significant difference (p-value = 0.852) in the pleural infection rate among the males and the females (27.8% and 27.4%, respectively), though the samples were more from the male population (Male: Female = 1.45: 1). The rate of pleural infection was highest in the age group of less than or equal to 25 years old (50%) and the difference was statistically significant (p<0.001). Pleural infections have been less common among study participants as their educational levels increased (Table 2).
| Variables | Categories | Frequency (n) | Percentage (%) |
|---|---|---|---|
| Gender | Male Female |
90 62 |
59.2 40.8 |
| Age (years) |
< 25 26-45 46-65 > 66 |
34 36 49 33 |
22.4 23.7 32.2 21.7 |
| Residence | Urban Rural |
59 93 |
38.8 61.2 |
| Educational status | Illiterate Primary Secondary Diploma & above |
11 24 73 44 |
7.2 15.8 48.0 29.0 |
| Occupation | Farmer Housewife Merchant Student Employee |
28 26 46 20 32 |
18.4 17.1 30.3 13.2 21.0 |
| Variables | Pleural Infection | COR [95% CI] | AOR [95% CI] | p-value | ||
|---|---|---|---|---|---|---|
| Negative (%) | Positive (%) | |||||
| Gender | ||||||
|
Male Female |
65 (72.2) 45 (72.6) |
25 (27.8) 17 (27.4) |
1.02 (0.49, 2.10) 1 |
1.09 (0.44, 2.74) 1 |
0.852 | |
| Age (years) | ||||||
|
<=25 26-45 46-65 >=66 |
17 (50.0) 29 (80.6) 36 (73.5) 28 (84.8) |
17 (50.0) 7 (19.4) 13 (26.5) 5 (15.2) |
5.60 (1.75, 17.96) 1.35 (0.38, 4.76) 2.02 (0.64, 6.35) 1 |
13.69 (3.29, 56.96) 3.09 (0.66, 14.49) 3.21 (0.83, 12.38) |
0.001 0.153 0.090 |
|
| Residence | ||||||
|
Urban Rural |
43 (72.9) 67 (72.0) |
16 (27.1) 26 (28.0) |
1 1.04 (0.50, 2.17) |
- | - | |
| Educational status | ||||||
|
Illiterate Primary Secondary Diploma & above |
7 (63.6) 15 (62.5) 55 (75.3) 33 (75.0) |
4 (36.4) 9 (37.5) 18 (24.7) 11 (25.0) |
1.71 (0.42, 6.99) 1.80 (0.62, 5.26) 0.98 (0.41, 2.33) 1 |
- | - | |
| Occupation | ||||||
|
Farmer Housewife Merchant Student Employee |
22 (78.6) 22 (84.6) 33 (71.7) 12 (60) 21 (65.6) |
6 (21.4) 4 (15.4) 13 (28.3) 8 (40.0) 11 (34.4) |
0.52 (0.16, 1.66) 0.35 (0.09, 1.26) 0.75 (0.29, 1.99) 1.27 (0.40, 4.04) 1 |
- | - | |
3.4. Clinical and Behavioral Factors Associated with Pleural Infection
A total of 44.7% of study participants diagnosed with pleural infection were admitted to the hospital for more than seven days, of which 39.7% of patients had a pleural infection. Study participants who had just undergone thoracic surgery (14.5%), chest tube drainage (16.4%), and a previous history of pneumonia (28.9%) had a culture-positive pleural infection of 50%, 48%, and 42.9%, respectively. The majority of study individuals had a BMI in the normal range of 18.5-24.9 kg/m2, with a pleural infection rate of 23.1%. Alcoholic and cigarette-addicted study participants had a pleural infection rate of 41.5% and 33.3%, respectively (Table 3).
Among clinical and behavioral factors, multivariable logistic regression analysis revealed that bacterial associated pleural infection was statistically significant for patients admitted to the hospital for more than seven days [AOR = 3.29, 95% CI: (1.39, 7.78)], patients who had undergone chest tube drainage [AOR = 3.43, 95% CI: (1.21, 9.76)], patients who had a previous history of pneumonia [AOR = 2.74, 95% CI: (1.11, 6.79)], and patients who had the habit of alcohol drinking for more than three days per week [AOR = 5.04; 95% CI: (1.85, 13.73)] (Table 3).
3.5. Antimicrobial Susceptibility Patterns of Bacterial Isolates.
Among Gram-positive bacterial isolates, resistance was observed against tetracycline (63.6%), doxycycline (57.6%), and chloramphenicol (51.5%). However, gentamicin, cefoxitin, cotrimoxazole, clindamycin, and erythromycin were found to be effective against 39.4% to 57.6% of isolated Gram-positive bacteria. 26.7% of S. aureus isolates were found to be methicillin-resistant (MRSA), while methicillin resistance CoNS (MR-CoNS) were found in one-third of the isolates. Penicillin, tetracycline, and chloramphenicol resistance were found in 80% of the Enterococcus spp. identified. One of the Enterococcus spp. isolated was vancomycin-resistant. Penicillin, erythromycin, cotrimoxazole, and vancomycin were all effective in S. pneumoniae and S. pyogenes isolates (Table 4).
| Variables | Pleural Infection | ||||||
| Negative (%) | Positive (%) | COR [95% CI] | AOR [95% CI] | p-value | |||
| Hospital admission | |||||||
| <= 7 days admitted | 69 (82.1) | 15 (17.9) | 1 | 1 | |||
| >7 days admitted | 41 (60.3) | 27 (39.7) | 3.03 (1.45, 6.35) | 3.29 (1.39, 7.78) | 0.007 | ||
| History of thoracic surgery | |||||||
| Yes | 11 (50.0) | 11 (50.0) | 3.19 (1.26, 8.08) | 2.51 (0.76, 8.31) | 0.133 | ||
| No | 98 (75.4) | 32 (24.6) | 1 | 1 | |||
| History of chest tube drainage | |||||||
| Yes | 13 (52.0) | 12 (48.0) | 2.99 (1.23, 7.23) | 3.43 (1.21, 9.76) | 0.021 | ||
| No | 97 (76.4) | 30 (23.6) | 1 | 1 | |||
| History of pneumonia | |||||||
| Yes | 26 (23.6) | 18 (42.9) | 2.42 (1.14, 5.15) | 2.74 (1.11, 6.79) | 0.029 | ||
| No | 84 (76.4) | 24 (57.1) | 1 | 1 | |||
| HIV/AIDS status | |||||||
| Positive | 18 (69.2) | 8 (30.8) | 1.20 (0.48, 3.02) | ||||
| Negative | 92 (73) | 34 (27.0) | 1 | ||||
| Hepatitis B or C status | |||||||
| Positive | 7 (58.3) | 5 (41.7) | 1.99 (0.59, 6.65) | ||||
| Negative | 103 (73.6) | 37 (26.4) | 1 | ||||
| Drinking alcohol | |||||||
| Yes | 24 (58.5) | 17 (41.5) | 2.44 (1.14, 5.23) | 5.04 (1.85, 13.73) | 0.002 | ||
| No | 86 (76.6) | 25 (23.4) | 1 | 1 | |||
| Cigarette smokers | |||||||
| Yes | 28 (66.7) | 14 (33.3) | 1.46 (0.68, 3.17) | ||||
| No | 81 (77.5) | 29 (22.5) | 1 | ||||
| Body Mass Index (BMI) Kg/m2 | |||||||
| <18 | 5 (33.3) | 10 (66.7) | 5.00 (0.70, 35.49) | ||||
| 18.5-24.9 | 100 (76.9) | 30 (23.1) | 0.75 (0.14, 4.06) | ||||
| >25 | 5 (71.4) | 2 (28.6) | 1 | ||||
| Antibiotics | Pattern | Bacteria Isolates | ||||||
|---|---|---|---|---|---|---|---|---|
| S. aureus (n=15) | CoNS (n=3) | VGS (n=5) | S. pneumoniae (n=3) | S. pyogenes (n=2) | Enterococcus spp. (n=5) | Total (n=33) | ||
| No. (%) | No. (%) | No. (%) | No. (%) | No. (%) | No. (%) | No. (%) | ||
| PEN | S I R |
3(20) 2(13.3) 10(66.7) |
1(33.3) 0(0) 2(66.7) |
NR NR NR |
3(100) 0(0) 0(0) |
2(100) 0(0) 0(0) |
1(20) 0(0) 4(80) |
10(30.3) 2(6.1) 16(48.5) |
| CXT | S | 11(73.3) | 2(66.7) | NA | NA | 2(100) | NA | 15(45.5) |
| I | 0(0) | 0(0) | NA | NA | 0(0) | NA | 0(0) | |
| R | 4(26.7) | 1(33.3) | NA | NA | 0(0) | NA | 5(15.2) | |
| GEN | S | 9(60) | 2(66.7) | NA | NA | 2(100) | NA | 13(39.4) |
| I | 3(20) | 1(33.3) | NA | NA | 0(0) | NA | 4(12.1) | |
| R | 3(20) | 0(0) | NA | NA | 0(0) | NA | 3(9.1) | |
| ERY | S | 7(46.7) | 2(66.7) | 4(80) | 3(100) | 2(100) | 1(20) | 19(57.6) |
| I | 3(20) | 1(33.3) | 1(20) | 0(0) | 0(0) | 0(0) | 5(15.2) | |
| R | 5(33.3) | 0(0) | 0(0) | 0(0) | 0(0) | 4(80) | 9(27.3) | |
| TET | S | 3(20) | 1(33.3) | 1(20) | 1(33.3) | 1(50) | 1(20) | 8(24.2) |
| I | 2(13.3) | 1(33.3) | 1(20) | 0(0) | 0(0) | 0(0) | 4(12.1) | |
| R | 10(66.7) | 1(33.3) | 3(60) | 2(66.7) | 1(50) | 4(80) | 21(63.6) | |
| DOX | S | 3(20) | 1(33.3) | NR | 1(33.3) | 0(0) | 1(20) | 6(18.2) |
| I | 1(6.7) | 0(0) | NR | 0(0) | 1(50) | 1(20) | 3(9.1) | |
| R | 11(73.3) | 2(66.7) | NR | 2(66.7) | 1(50) | 3(60) | 19(57.6) | |
| CPR | S | 4(26.7) | 0(0) | 2(40) | NA | 1(50) | 2(40) | 9(27.3) |
| I | 3(20) | 2(66.7) | 2(40) | NA | 1(50) | 0(0) | 8(24.2) | |
| R | 8(53.3) | 1(33.3) | 1(20) | NA | 0(0) | 3(60) | 13(39.4) | |
| CLD | S | 8(53.3) | 2(66.7) | 4(80) | 2(66.7) | 2(100) | NA | 18(54.5) |
| I | 3(20) | 1(33.3) | 1(20) | 0(0) | 0(0) | NA | 5(15.2) | |
| R | 4(26.7) | 0(0) | 0(0) | 1(33.3) | 0(0) | NA | 5(15.2) | |
| COT | S | 7(46.7) | 2(66.7) | NA | 3(100) | 2(100) | NA | 14(42.4) |
| I | 4(26.7) | 1(33.3) | NA | 0(0) | 0(0) | NA | 5(15.2) | |
| R | 4(26.7) | 0(0) | NA | 0(0) | 0(0) | NA | 4(12.1) | |
| VAN | S | NA | NA | 3(60) | 3(100) | 2(100) | 4(80) | 12(36.4) |
| I | NA | NA | 0(0) | 0(0) | 0(0) | 0(0) | 0(0) | |
| R | NA | NA | 2(40) | 0(0) | 0(0) | 1(20) | 3(9.1) | |
| CHL | S | 4(26.7) | 1(33.3) | 2(40) | 2(66.7) | 1(50) | 1(20) | 11(33.3) |
| I | 3(20) | 0(0) | 1(20) | 0(0) | 1(50) | 0(0) | 5(15.2) | |
| R | 8(53.3) | 2(66.7) | 2(40) | 1(33.3) | 0(0) | 4(80) | 17(51.5) | |
On the other hand, more than half of Gram-negative bacterial isolates were resistant to ampicillin and ciprofloxacin, whereas gentamicin (60%) and meropenem (60%) had better responses. Ampicillin resistance was high in E. coli (80%), while ampicillin, cefepime, tetracycline, doxycycline, ciprofloxacin, and ceftriaxone resistance were found in 66.7% of isolated K. pneumoniae. Piperacillin was found to be 100 percent effective against P. aeruginosa isolates. In contrast, all isolated P. aeruginosa were resistant to ceftazidime (Table 5).
| Antibiotics | Pattern | Bacteria Isolates | |||
|---|---|---|---|---|---|
| E. coli (n=5) | K. pneumoniae (n=3) | P. aeruginosa (n=2) | Total (n=10) | ||
| No. (%) | No. (%) | No. (%) | No. (%) | ||
| AMP | S | 1(20) | 1(33.3) | NA | 2(20) |
| I | 0(0) | 0(0) | NA | 0(0) | |
| R | 4(80) | 2(66.7) | NA | 6(60) | |
| PIP | S | NA | NA | 2(100) | 2(20) |
| I | NA | NA | 0(0) | 0(0) | |
| R | NA | NA | 0(0) | 0(0) | |
| CFP | S | 2(40) | 1(33.3) | 1(50) | 4(40) |
| I | 1(20) | 0(0) | 0(0) | 1(10) | |
| R | 2(40) | 2(66.7) | 1(50) | 5(50) | |
| CXT | S | 3(60) | 2(66.7) | NA | 5(50) |
| I | 1(20) | 1(33.3) | NA | 2(20) | |
| R | 1(20) | 0(0) | NA | 1(20) | |
| CTR | S | 2(40) | 1(33.3) | NA | 3(30) |
| I | 1(20) | 0(0) | NA | 1(10) | |
| R | 2(40) | 2(66.7) | NA | 4(40) | |
| CFZ | S | NA | NA | 0(0) | 0(0) |
| I | NA | NA | 0(0) | 0(0) | |
| R | NA | NA | 2(100) | 2(20) | |
| GEN | S | 3(60) | 2(66.7) | 1(50) | 6(60) |
| I | 0(0) | 0(0) | 0(0) | 0(0) | |
| R | 2(40) | 1(33.3) | 1(50) | 4(40) | |
| MER | S | 4(80) | 2(66.7) | 1(50) | 6(60) |
| I | 0(0) | 0(0) | 0(0) | 0(0) | |
| R | 1(20) | 1(33.3) | 1(50) | 3(30) | |
| TET | S | 1(20) | 1(33.3) | NA | 2(20) |
| I | 1(20) | 0(0) | NA | 1(10) | |
| R | 3(60) | 2(66.7) | NA | 5(50) | |
| DOX | S | 1(20) | 1(33.3) | NA | 2(20) |
| I | 1(20) | 0(0) | NA | 1(10) | |
| R | 3(60) | 2(66.7) | NA | 5(50) | |
| CPR | S | 2(40) | 1(33.3) | 1(50) | 4(40) |
| I | 0(0) | 0(0) | 0(0) | 0(0) | |
| R | 3(60) | 2(66.7) | 1(50) | 6(60) | |
| CHL | S | 2(40) | 1(33.3) | NA | 3(30) |
| I | 0(0) | 1(33.3) | NA | 1(10) | |
| R | 3(60) | 1(33.3) | NA | 4(40) | |
|
Bacterial Isolates |
Antimicrobial-Resistant Pattern (%) | |||||
|---|---|---|---|---|---|---|
| R1 | R2 | R3 | R4 | > R5 | Total (> R2) | |
| S. aureus (n=15) | 1(6.7) | 3(20) | 2(13.3) | 1(6.7) | 1(6.7) | 8(53.3) |
| Enterococcus spp. (n=5) | 0(0) | 2(40) | 1(20) | 0(0) | 0(0) | 3(60) |
| VGS (n=5) | 1(20) | 1(20) | 0(0) | 0(0) | 0(0) | 2(40) |
| CoNS (n=3) | 1(33.3) | 0(0) | 0(0) | 0(0) | 0(0) | 1(33.3) |
| S. pneumoniae (n=3) | 1(33.3) | 0(0) | 0(0) | 0(0) | 0(0) | 1(33.3) |
| S. pyogenes (n=2) | 0(0) | 0(0) | 0(0) | 0(0) | 0(0) | 0(0) |
| E. coli (n=5) | 0(0) | 2(40) | 1(20) | 0(0) | 0(0) | 3 (60) |
| K. pneumoniae (n=3) | 0(0) | 0(0) | 1(33.3) | 1(33.3) | 0(0) | 2(66.7) |
| P. aeruginosa (n=2) | 1(50) | 0(0) | 0(0) | 0(0) | 0(0) | 1(50) |
| Total (n=43) | 5(11.6) | 8(18.6) | 5(11.6) | 2(4.7) | 1(2.3) | 21(48.8) |
3.6. Multidrug-Resistant (MDR) Patterns of Bacterial Isolates
In this study, the overall prevalence of MDR bacteria was 48.8% (21/43). Gram-positive and Gram-negative bacterial isolates with MDR were determined to be 45.5% (15/33) and 60% (6/10), respectively. MDR was found in more than half of the S. aureus, Enterococcus spp., E. coli, and K. pneumoniae isolates (Table 6).
4. DISCUSSION
In this study, bacterial culture yielded positive results in 27.6% of suspected pleural infection hospitalized patients, with a 95% confidence interval of (21.1–34.9).This finding was in line with previous studies conducted in India (25.3%, 30.3%) [25, 26], Pakistan (32.9%, 23.2%) [11, 15], and Ethiopia (23.7%, 27.3%) [20, 24]. However, our finding was greater than other studies done in India (14.3% to15.7%) [21, 28, 29]. In contrast, the finding was relatively lower than studies done in northeastern India (37.3%) [17] and Nigeria (43.8%) [8]. The reasons for the discrepancy in the results might be due to study population deference and the lack of use of molecular methods for diagnosis in our study, which greatly enhance the chances of detection of the etiological agents, even in previously treated patients before sampling.
Gram-positive bacteria were isolated in the majority (76.7%), with S. aureus accounting for 34.9%, Enterococcus spp. 11.6%, Viridans group streptococci 11.6%, S. pneumoniae 7%, CoNS 7%, and S. pyogenes 4.7%, while Gram-negative bacteria were isolated in only 23.3%, with E. coli accounting for 11.7%, K. pneumoniae 7%, and P. aeruginosa 4.7%. This finding is consistent with retrospective studies conducted in Pakistan [14] and India [17, 30]. On the other hand, Gram-negative bacteria were found predominantly in the studies done in India [22, 28], Ethiopia [20], and Nigeria [8]. Variations in pathogenic bacteria distribution in the research areas, variances in socio-demographic and clinical status of study participants, and varying infection control methods in various geographical settings could all explain the observed disparities in isolated species.
In this study, S. aureus (34.9%) was the most predominant isolate, which was in line with reports in African and Asian countries where S. aureus has been reported to be more common than S. pneumoniae as a cause of pleural infection and leads to empyema thoracis, which is associated with low socioeconomic status, malnutrition, and delay in diagnosis of pneumonia [1, 18, 19]. The predominance of S. aureus infection seen in this study could be associated with an endogenous source as the organism colonizes the skin and nasopharyngeal area; this could be introduced following any iatrogenic procedures, and the organism may also be transmitted from the hospital environment [1]. Whereas the prevalence of S. pneumoniae (7%) was lower than in earlier investigations [14, 17, 24]. The possible reasons for these varied findings in our study could be populations’ differences in pathogen exposure, the distribution of pneumococcal vaccine, and pre-sampling performed in medical procedures. Respiratory infections caused by pathogenic organisms like S. pneumoniae are also thought to present prominent symptoms at an early stage of the disease. As a result, it is frequently treated earlier, lowering the identification rate of S. pneumoniae from pleural infection [17].
The antimicrobial susceptibility pattern of this study showed that more than half of the Gram-positive bacterial isolates were resistant to chloramphenicol (51.5%), doxycycline (57.6%), and tetracycline (63.6%). However, more than half of the isolated Gram-positive bacteria showed relatively good sensitivity to clindamycin (54.5%) and erythromycin (57.6%). Greater than 66.7% of isolated S.aureus were resistant to penicillin, tetracycline, and doxycycline. In this study, MRSA was observed among 26.7% of isolates, which was lower than studies in Nigeria (37.5%) [8], and a systemic review reported in Oxford, UK (42% to 67%) [1]. One-third of the isolated CoNS were methicillin-resistant. Likewise, vancomycin-resistant Enterococcus (VRE) was observed in 20% of the isolated species. Moreover, 80% of isolated Enterococcus spp. were resistant to penicillin, tetracycline, and chloramphenicol, whereas 100% of isolated S. pneumoniae and S. pyogenes were susceptible to penicillin, erythromycin, cotrimoxazole, and vancomycin. The finding is comparable to the studies conducted in India [21, 23] and Nigeria [8]. In contrast, investigations in India [22] and Ethiopia [20, 24] found that isolated Gram-positive bacteria had a greater rate of erythromycin, clindamycin, and cotrimoxazole resistance. This difference could be related to trends in antibiotic prescription practice for this antibiotic for the empirical treatment, self-medication practice of study participants, and the spread of drug-resistant bacteria.
Gram-negative bacteria isolates also showed resistance to ampicillin (60%), ciprofloxacin (60%), and cefuroxime (60%). However, gentamicin (60%) and meropenem (60%) were effective against Gram-negative isolates. Isolated E. coli showed high resistance to ampicillin (80%). Likewise, 66.7% of isolated K. pneumoniae were resistant to ampicillin, tetracycline, doxycycline, ciprofloxacin, and ceftriaxone. This suggests that routinely prescribed antibiotics are unlikely to be used as empirical therapy for Gram-negative bacterial-associated pleural infections. Piperacillin (100%) was effective against P. aeruginosa isolates, while ceftazidime was not. The overall finding of this study was comparable to previous studies conducted in different parts of India [22, 29] and Nigeria [8]. However, previous studies conducted in Ethiopia [20, 24] reported that Gram-negative bacteria isolated from various sterile bodily fluids were found to be chloramphenicol and gentamicin resistant. The cause of this could be attributed to the increasing use of conventional antibiotics and their dispersion, as well as isolated strain variances.
In this study, 48.8% (21/43) of the isolated bacteria were MDR. The percentages of Gram-positive and Gram-negative MDR bacteria isolates were found to be 45.5% (15/33) and 60% (6/10), respectively. The most frequently isolated pathogen, S. aureus, showed 53.3% (8/15) of an MDR level, whereas more than 60% of isolated Enterococcus spp., E. coli, and K. pneumoniae showed MDR for commonly prescribed antibiotics. This rate of antibiotic resistance might reflect inappropriate use of antibiotics, lack of laboratory diagnostic tests for appropriate antibiotic selection, unavailability of guidelines for the selection of antibiotics, a trend of self-medication, or poor hospital control measures [1, 7, 8, 16, 20-24]. However, the total number of MDR isolates in this investigation was lower than in previous studies conducted in Ethiopia on various bodily fluids [20, 24] and the systemic review reported in Oxford, UK [1]. This could be related to the widespread use of broad-spectrum antibiotics for empirical treatment of infection, the prevalence of drug-resistant bacteria, or isolated strain variances.
Factors that may have contributed to the pathophysiology of bacterial pleural infection were analyzed. As per the analysis, age groups, mainly patients under the age of 25, were more than thirteen times more likely to develop pleural infection compared to other age groups, which is in line with previous studies in Ethiopia [20]. This could be due to a combination of high exposure and immature immunity, and most patients with respiratory tract infections may take Ibuprofen before being admitted to the hospital to reduce fever, which can interfere with immunological responses and bacteria replication because many pathogenic bacteria serotypes are temperature-sensitive [8, 31].
Another group statistically associated with culture-confirmed pleural infection was patients who were admitted to the hospital for more than seven days. Patients admitted for more than seven days were more than three times more likely to develop a bacterial pleural infection than patients admitted for less than seven days. Similarly, different studies at the University of Perth, Western Australia [5, 32] reported that long-term hospitalization leads to nosocomial infections. This might be owing to the hospital's degree of environmental cleanliness, which can serve as a source of bacterial infection through mechanical vehicles. In addition, invasive medical and surgical treatments used to treat other related health problems may introduce bacteria into sites.
Another clinical factor found to be statistically significant for pleural infection was chest tube drainage. Patients who had been treated through chest tube drainage were more than three times at risk of developing a bacterial-associated pleural infection. This finding was similar to other studies conducted at the University of Perth, Western Australia [5] and Royal Berkshire Hospital, UK [13]. The reason for this could be that throughout the drainage, normal flora bacteria and their products might be translocated into the sterile spaces of the pleural cavity. Bacteria can also be transmitted through devices that have not been thoroughly sanitized. Furthermore, the organ membrane may rupture during chest tube installation, resulting in inflammatory reactions that encourage bacterial entry and proliferation.
Likewise, patients with a previous pneumonia history were more than twice as likely to develop a bacterial-associated pleural infection. This is supported by different studies done previously at the University of Perth, Western Australia [5], India [3, 18], and Salt Lake City, USA [16]. Complications of bacterial pneumonia are known to be the cause of pleural infections that may lead to the development of empyema thoracis in all age groups, especially in developed countries where approximately 60% of pleural infections are associated with patients hospitalized with bacterial pneumonia [1, 6, 16, 17, 20].
The patient who had a habit of drinking alcohol (more than three days per week) was also another behavioral factor that was statistically significant for bacterial pleural infection. This result agrees with studies conducted at Royal Berkshire Hospital, UK [13] and New Orleans, south-eastern USA [33-35]. This could be due to the pathophysiology and critical components involved in neutrophil-driven lung immunity in response to bacterial infection being altered by prolonged alcohol use. In addition, regular alcohol consumption speeds up inflammatory responses, reduces anti-inflammatory cytokines, and disrupts the oropharyngeal flora's microbiota; these could make patients more vulnerable to bacterial illness.
4.1. Limitation of the study
Limitations of this study include the fact that conventional aerobic culture techniques were used for the identification of bacterial pathogens, which may have missed the event of antimicrobial use before sampling since our study participants were hospital admitted. Antimicrobial tests recommended using the minimum inhibition concentration (MIC) approach were not carried out. Due to the lack of appropriate culture media and an organized laboratory setup, strictly anaerobic and fastidious bacteria, as well as fungus and viruses, were not included in this study. The signs and symptoms of pleural infections can be due to the pathogens not included in this study. This study was also conducted with a limited sample size by using the correction formula. This may not reflect the entire scenario of the study. This highlights the need for further studies to be conducted on larger sample sizes and advanced techniques to better understand the possible pathogens of pleural infection in this part of the country.
CONCLUSION
In this study, the magnitude of bacterial pleural infections was comparable to that of other similar investigations. Bacterial-associated pleural infections have been associated with being under 25 years old, being admitted to the hospital for more than a week, having a history of chest tube drainage, a clinical history of pneumonia, and the habit of drinking alcohol. The most common cause of pleural infections was Gram-positive bacteria with a high level of antibiotic resistant. S. aureus and E. coli were the most common species among Gram-positive and Gram-negative isolates, respectively, and more than half of the isolates were resistant to commonly prescribed antibiotics in the hospital. MRSA and VRE were observed among 26.7% and 20% of isolates, respectively. Thus, the nature of bacterial isolates and their antibiotic susceptibility profiles should be considered in pleural infection therapy and management, and the hospital should work on infection prevention strategies, especially for hospital-acquired drug-resistant bacteria.
LIST OF ABBREVIATIONS
| SPSS | = Statistical Package for the Social Sciences |
| MDR | = Multidrug-resistant |
| CoNS | = Coagulase-negative staphylococci |
| BMI | = Body Mass Index |
AUTHORS' CONTRIBUTIONS
All authors made substantial contributions to the development of the project to data collection, analysis, and interpretation; agreed to submit it to this journal; gave final approval of the version to be published; and agreed to be responsible for all aspects of the project.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was ethically authorized by the Arba Minch University College of Medicine and Health Sciences Institutional Review Board (Ref. No. CMHS/12033993/11).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Written consent and assent were obtained from the respective participants.
STANDARDS OF REPORTING
STROBE guideline has been followed.
AVAILABILITY OF DATA AND MATERIALS
Due to ethical and confidentiality concerns, the data sets evaluated in this work are available from the corresponding author [D.A.] upon reasonable request.
FUNDING
The project was not supported by the sponsoring agencies.
CONFLICT OF INTEREST
The authors declare that they have no conflicts of interest in this work.
ACKNOWLEDGEMENTS
We gratefully acknowledge Arba Minch University, Colleges of Medicine and Health Sciences, Department of Medical Laboratory Science, and we thank everyone who participated in the study for their cooperation.


