All published articles of this journal are available on ScienceDirect.
Magnitude of Multidrug-resistant Bacterial Uropathogens and Associated Factors in Urinary Tract Infection Suspected Adult HIV-Positive Patients in Southern Ethiopia
Abstract
Background:
Due to the reduction of their immunity, people living with the human immunodeficiency virus (HIV) are more susceptible to acquiring urinary tract infections (UTI). Moreover, the emergence of drug-resistant bacterial uropathogens has grown widely and now constitutes a severe clinical problem among HIV-positive patients.
Methods:
An institution-based cross-sectional study was conducted at the ART clinic of Arba Minch General Hospital from 1st January to 30th May, 2021. A total of 251 adult HIV-positive patients clinically suspected of having UTIs were systematically recruited. Socio-demographic and other factors were collected by a pre-tested structured questionnaire. A clean-catch mid-stream urine sample was collected and inoculated onto blood agar, MacConkey agar, and cysteine lactose electrolyte deficient agar (CLED). The inoculated culture media were incubated aerobically at 37°C for 24 hrs. After overnight incubation, significant bacteriuria, 105 CFU/ml, was considered. Bacterial identification was made by standard protocols. Antimicrobial susceptibility testing was done by Kirby Bauer’s disk diffusion method. The data was analyzed using SPSS version 25. A P-value less than 0.05 was used as statistical significance.
Results:
The overall magnitude of multidrug-resistant (MDR) uropathogens was found to be 14.3%, with a 95% CI (10-19.1). Gram-negative bacteria (83.3%) were the predominant MDR isolates, with E. coli (41.7%) followed by K. pneumoniae (22.2%). All isolates of S. aureus and two-thirds of the isolated CoNS were MDR. A total of 30% and 16.7% of Gram-negative isolates were ESBL and carbapenemase producers, respectively, whereas 50% and 33.3% of isolated S. aureus and CoNS were methicillin-resistant, respectively. Antibiotic use in the previous 6 months [AOR = 6.7, 95% CI: (2.53-17.6)], presence of chronic underlying disease [AOR = 7.03, 95% CI: (1.72-28.73)], antibiotic use without a prescription [AOR = 9.7, 95% CI: (3.53-26.675)], CD4+ counts less than 250 cells/mm3 [AOR = 8.05, 95% CI: (2.078–31.154)], and CD4+ count in between 250-500 cells/mm3 [AOR = 2.49, 95% CI: (1.05-5.923)] were found to be statistically significant associated with MDR uropathogens.
Conclusion:
This study found a significant number of multidrug-resistant uropathogens, indicating that healthcare providers should be aware of this and take urgent action to minimize resistance spread, as well as consider current empirical antibiotic therapy for UTI in HIV-positive patients.
1. INTRODUCTION
A bacterial urinary tract infection can range from asymptomatic to a severe kidney infection [1, 2]. The clinical significance of an asymptomatic urinary tract infection (UTI) is the increased likelihood of it becoming symptomatic [2, 3]. The presence of a significant number of bacteria in the urine (greater than or equal to 105) is indicative of a urinary tract infection (UTI) [3, 4].
Each year, more than 150 million people are diagnosed with urinary tract infections (UTIs), which cost the worldwide economy over $6 billion and account for a significant portion of the workload in clinical microbiology laboratories [1-5]. Due to the reduction of their immunity, people living with human immunodeficiency virus (HIV) are more susceptible to urinary tract infections [6-8]. Apart from being at risk of developing UTI, HIV-positive patients are at high risk for recurrences, extensive treatment durations, and more complex infections, including pyelonephritis [1, 3].
The prevalence of UTI in people living with HIV has been documented to be high and varies from place to place, ranging from 4 to 25.3% [6-8]. Escherichia coli, Staphylococcus aureus, Klebsiella species, Coagulase-negative Staphylococci species (CoNS), Enterococcus faecalis, and Pseudomonas aeruginosa were previously identified as the most common bacterial isolates causing UTI in HIV-positive patients [1-8]. More than half of bacterial isolates show moderate to high resistance to commonly prescribed antibiotics [3-6]. Multidrug resistance (MDR) has been demonstrated in 58.3% and up to 78.4% of isolates causing UTI in HIV-positive patients and is reported to be inversely associated with CD4 counts [9-15].
Antimicrobial use for opportunistic infection is a common factor for MDR uropathogens in HIV-positive patients [7]. In particular, the condition is high in developing countries, including Ethiopia, where facilities for urine culture and antimicrobial susceptibility testing are still insufficient, resulting in incorrect diagnosis, conventional antibiotic treatment of urinary tract infections, irrational use of broad-spectrum antibiotics, and inadequate infection prevention programs [15-17].
Despite the fact that Ethiopia is expected to have a large number of HIV-positive patients [18, 19], there is little information on the extent of multidrug-resistant uropathogens among HIV-positive patients. Furthermore, the antibiotics that are indicated for empirical treatment in the general population are also used for HIV-positive individuals. However, the efficiency of these antibiotics in treating UTIs in HIV-positive patients is unknown. Thus, this study aimed to determine the magnitude of multidrug-resistant (MDR) bacterial uropathogens and associated factors in UTI-suspected adult HIV-positive patients in Southern Ethiopia.
2. MATERIALS AND METHODS
2.1. Study Setting and Period
An institution-based cross-sectional study was conducted from 1st January to 30th May, 2021, at the ART clinic of Arba Minch General Hospital. Arba Minch is the administrative center of the Gamo zone, which is 454 kilometers from Ethiopia's capital, Addis Ababa. More than 2 million individuals from the Gamo zone and other neighboring zones receive preventive, curative, and rehabilitative care at the hospital. The hospital also offers adults optional counseling testing and antiretroviral therapy (ART) to more than 1860 HIV-positive patients.
2.2. Study Population and Eligibility Criteria
HIV-positive adult patients at Arba Minch General Hospital ART clinic with at least two of the following signs and symptoms of UTIs were included as a study population: fever, suprapubic discomfort, costovertebral angle pain or tenderness, urine urgency, urinary frequency, or dysuria [20]. Patients were excluded if they were (a) less than 18 years old, (b) taking antibiotics other than cotrimoxazole within the last two weeks, (c) unconscious or had cognitive impairment, or (d) refusing to participate.
2.3. Sample Size and Sampling Technique
The sample size was calculated using the single population proportion formula, based on the following assumptions: the proportion MDR uropathogen (p = 11.2%) was taken from the previous study conducted in Wolayita Sodo, Ethiopia [1], with a 95% confidence level (Zα/2 = 1.96), 4% margin of error, and 5% non-response rate. Therefore, the total sample size was 251. A systematic random sampling technique was employed to recruit the study participants after calculating Kth –value.
2.4. Data Collection Tools and Laboratory Investigations
A pre-tested structured questionnaire administered through the face-to-face interview was used to collect data regarding socio-demographic characteristics as well as clinical and behavioral factors. Other significant medical information was obtained from the patient's medical record, including CD4+ status, UTI treatment history, and other possible comorbidities. Each participant underwent a clinical examination, with special attention to signs and symptoms of UTI (include at least two of the following signs and/or symptoms: fever, suprapubic pain or tenderness, costovertebral angle pain or tenderness, dysuria, frequent or urgent urination, and suprapubic pain or tenderness).
A freshly voided clean-catch midstream urine sample (10–20 mL) was collected from all eligible study participants immediately before initiating antimicrobial therapy using wide-mouth screw-capped leak-proof sterile containers, taking all precautions to avoid contamination. All the urine samples were processed in a microbiology laboratory within 30 minutes of collection. The urine specimen in each patient was cultured on blood agar, MacConkey agar, and cysteine lactose electrolyte deficient agar (CLED) (HiMedia Laboratory Pvt. Ltd., Mumbai, India). One-microliter disposable loops were used for nucleation on culture media plates, and the plates were incubated at 37 °C for 24 hours. In case of a delay in processing the samples, the samples were kept at 2–8 °C in a well-monitored refrigerator and cultured within 6 hours [21].
The urine cultures that grew a bacterial colony count greater than or equal to 105 colony-forming units (CFU) per microliter of a typical urinary tract organism were regarded as significant bacterial growth. A gram stain was performed on the colony for gram reaction identification. The Gram-negative bacteria identification test was performed by using the conventional method of identification, which included Kligler Iron Agar (KIA) (HiMedia laboratory Pvt Ltd, Mumbai, India), Sulphur Indole Motility (SIM) (HiMedia laboratory Pvt. Ltd, Mumbai, India), Citrate (HiMedia laboratory Pvt Ltd, Mumbai, India), and Urea (HiMedia laboratory Pvt Ltd, Mumbai, India), while Gram-positive identification tests were performed by using catalase (Remel Europe Ltd, Dartford, UK), and coagulase (Remel Europe Ltd, Dartford, UK) for staphylococcus spp [22, 23].
2.5. Antimicrobial Susceptibility Testing
An antimicrobial susceptibility test was performed on Mueller Hinton agar by using Kirby-Bauer disk diffusion techniques. Morphologically identical 3-5 pure colonies from 24 hours pure culture were selected, the top of each colony was successively touched by using a one microliter loop, and bacteria were transferred to a tube containing 5 mL of Mueller-Hinton broth (HiMedia laboratory Pvt Ltd, Mumbai, India), then the tube was incubated at 37 °C long enough (8hrs) to produce an organism suspension with slightly to moderate cloudiness. Afterward, the broth was diluted with sterile saline to obtain turbidity equivalent to that of 0.5 McFarland standards. Then, a sterile cotton swab was dipped into adjusted inoculum, and the swab was lifted above the fluid level and rotated against the inside of the tube to remove as much excess inoculum as possible. The entire Mueller-Hinton Agar was streaked with the swab evenly in three directions, and the inoculum was left to dry for 5 minutes with the plate closed. Lastly, selected antibiotic discs (Abtek company of UK) were aseptically placed on the surface of the medium and the diameter of the zone of inhibition was measured by millimeter after overnight incubation and interpreted as sensitive, intermediate, and resistant as per the CLSI guidelines [24].
The following antibiotic discs with their respective concentrations were used: for Gram-positive cocci cefoxitin (30 µg), clindamycin (30 µg), gentamicin (10 µg), ampicillin (10 µg), ciprofloxacin (5 µg), tetracycline (30 µg), and cotrimoxazole (25 µg were used and for Gram-negative bacilli isolates, ceftazidime(30 µg), ceftriaxone (30 µg), cefepime (30 µg), meropenem (10 µg), ciprofloxacin (10 µg), gentamicin (10 µg), cotrimoxazole (25 µg), ampicillin (10 µg), and nitrofurantoin (300 µg) were used. E.coli ATCC 25922 and S.aureus ATCC (25923) were used as control strains to assess the antibiotics' effectiveness. The results were interpreted as sensitive, intermediate and resistant according to the CLSI guidelines after the plate was incubated aerobically at 37°C for 16-18 hours. Multidrug-resistant (MDR) was defined as an organism's resistance to at least one antimicrobial drug in three or more antimicrobial categories [24].
2.6. Identifications of ESβL and Carbapenemase Producing Gram-negative Bacterial Isolates
The double-disc synergy test method was also used to identify extended-spectrum beta-lactamase (ESBL). Potential ESBL producers, Gram-negative bacilli, were isolated with inhibition zones smaller than the CLSI-defined breakpoints: 22 mm for ceftazidime (30 µg) and 25 mm for ceftriaxone (30 µg). When Gram-negative isolates were meropenem intermediate or resistant, carbapenem resistance was detected, and carbapenemase enzyme production was confirmed using the modified Carbapenem Inactivation Methods (mCIM) [25].
2.7. Data Quality Assurance
A pre-test was conducted on 5% of study participants to ensure that the data collection format was feasible in a closely related setting prior to the actual data collection. Standard operating protocols were closely followed, and laboratory materials and reagents, as well as quality control criteria, were kept in good condition. Data completeness was double-checked daily. S. aureus ATCC 25923 and E. coli ATCC 25922 were used as control strains to evaluate the quality of culture media, biochemical tests, and antibiotic disks.
2.8. Statistical Analysis
All datasets were cross-checked for integrity and internal consistency before being encoded, entered, and analyzed using the Statistical Package for Social Science (SPSS) version 25 software. To define the socio-demographic characteristics of the study population, descriptive statistics, such as frequency, mean, and percentage, were determined. Bivariate logistic regression analysis was used to determine the association between MDR uropathogens and their associated factors. Variables with a P-value < 0.25 in the bivariable analysis were included in the final multivariable analysis. Statistical significance was defined as a P-value < 0.05.
3. RESULTS
3.1. Socio-Demographic Characteristics of Study Participants
Among 251 recruited HIV-positive adult patients, 137 (54.6%) were females, and 240 (95.6%) of the participants were urban residents. The mean age of the study participants was 42.91 years with ± 8.31 standard deviation. The majority of the respondents were in the age group of 40–49 years old (52.2%). About 153 (61%) of the study participants were married, and more than half of the study participants attended primary school educational labels (Table 1).
The magnitude of MDR uropathogens was high among females (8%) than males (6.4%). Participants aged 40–49 years had the highest frequency of MDR uropathogens (5.6%). Married study participants had the highest prevalence of MDR uropathogens (7.6%), followed by divorced (4.4%) (Table 1).
| Variables | Categories | MDR Uropathogen | |
| Negative | Positive | ||
| No (%) | No (%) | ||
| Sex | Male | 98(39) | 16(6.4) |
| Female | 117(46.6) | 20(8.0) | |
| Age group in years | 18-29 | 9(3.6) | 3(1.2) |
| 30-39 | 39(15.5) | 6(2.4) | |
| 40-49 | 117(46.6) | 14(5.6) | |
| > 50 | 50(19.9) | 13(5.2) | |
| Residence | Urban | 206(82.1) | 34(13.5) |
| Rural | 9(3.6) | 2(0.8) | |
| Occupation | Employee | 78(31.1) | 11(4.4) |
| Farmer | 19(7.6) | 3(1.2) | |
| Merchant | 44(17.5) | 9(3.6) | |
| Housewife | 63(25.1) | 10(4.0) | |
| Student | 5(2.0) | 1(0.4) | |
| Daily labour | 6(2.4) | 2(0.8) | |
| Marital Status | Single | 16(6.4) | 4(1.6) |
| Married | 134(53.4) | 19(7.6) | |
| Divorced | 42(16.7) | 11(4.4) | |
| Widowed | 23(9.2) | 2(0.8) | |
3.2. Clinical Characteristics of Study Participants
More than half of the study participants, i.e., 148 (59.0%), had a CD4+ count greater than 500 cells/mm3, and the mean CD4+ cell count was 602.08 (SD: 219.76). About 8.4% of the study participants had a previous history of UTI, and 7.2% had been hospitalized in the previous 12 months. In addition, 11.2% of the individuals had taken antibiotics in the previous six months, while 5.6% of those used cotrimoxazole as prophylaxis (Table 2).
Among study participants with previous exposure to UTI, 28.6% (6/21) had MDR uropathogenic isolates. Out of study participants who had antibiotic usage in the last six months, the frequency of isolated MDR uropathogens was found to be 39.3% (11/28). The magnitude of MDR uropathogens among patients who had CD4+ less than 250 cells/mm3 was found to be 33.3% (5/15) (Table 2).
3.3. Diversity of Bacterial Isolates
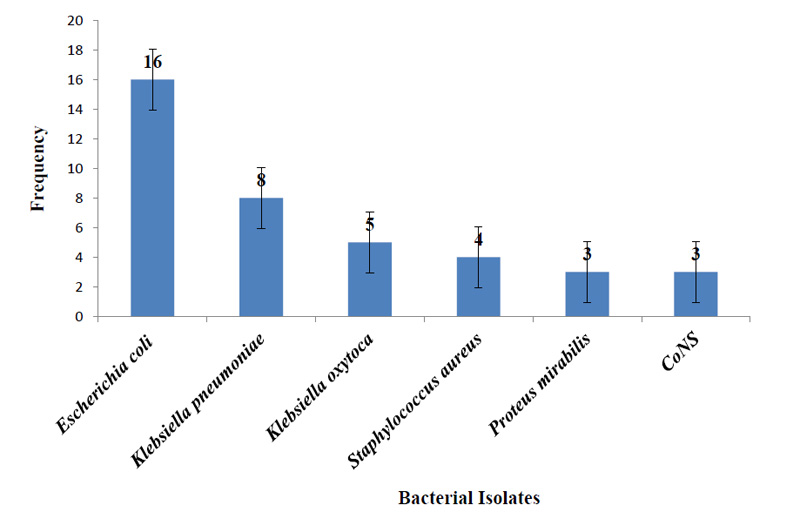
A total of 39 bacterial isolates belonging to five different genera were isolated from 251 UTI-suspected adult HIV patients. Among the bacterial isolates, 32/39 (82.1%) were Gram-negative bacilli, and 7/39 (17.9%) were Gram-positive cocci. Escherichia coli 16/39 was found to be the most frequently isolated organism (41.0%) (Fig. 1).
| Variables | Categories | MDR Uropathogen | |
| Negative | Positive | ||
| No (%) | No (%) | ||
| Previous history of UTI | Yes | 15(6.0) | 6(2.4) |
| No | 200(79.7) | 30(12.0) | |
| Antibiotic usage in the last 6 months | Yes | 17(6.8) | 11(4.4) |
| No | 198(78.9) | 25(10.0) | |
| Presence of chronic underlying disease | Yes | 6(2.4) | 5(2.0) |
| No | 209(83.3) | 31(12.4) | |
| Hospitalization within the last 12 months | Yes | 8(3.2) | 10(4.0) |
| No | 207(82.5) | 26(10.4) | |
| Use of antibiotics without prescription | Yes | 13(5.2) | 11(4.4) |
| No | 208(80.5) | 25(10.4) | |
| Use of cotrimoxazole as prophylaxis | Yes | 9(4.8) | 5(2.0) |
| No | 206(82.1) | 31(12.4) | |
| CD4+ count (cells/mm3) | <250 | 10(4.0) | 5(2.0) |
| 250-500 | 71(28.3) | 17(6.8) | |
| >500 | 134(53.4) | 14(5.6) | |
3.4. Antimicrobial Susceptibility Patterns of Isolated Uropathogens
The isolates' antibiogram testing findings revealed a wide range of resistance and sensitivity patterns. Resistance to cotrimoxazole (78.1%), ampicillin (75%), and gentamicin (65.6%) was found among isolated Gram-negative bacteria. The predominant isolated E. coli strains were resistant to ampicillin (100%) and cotrimoxazole (93.8%). Ceftazidime, ciprofloxacin, cefoxitin, and cefepime were found to be effective against 59% to 96% of isolated Gram-negative uropathogenic bacteria (Table 3).
| Antibiotics | Pattern | Gram-Negative Bacterial Uropathogens | Total (n=32) | |||
| E.coli (n=16) | K.pneumoniae (n=8) | K.oxytoca (n=5) | P.mirabilis (n=3) | |||
| No. (%) | No. (%) | No. (%) | No. (%) | No. (%) | ||
| AMP | S | 0 | NA | 0 | 0 | 0 |
| R | 16(100) | NA | 5(100) | 3(100) | 24(75) | |
| GEN | S | 4(25) | 3(37.5) | 2(40) | 2(66.7) | 11(34.4) |
| R | 12(75) | 5(62.5) | 3(60) | 1(33.3) | 21(65.6) | |
| CPR | S | 12(75) | 6(75) | 4(80) | 2(66.7) | 24(75) |
| R | 4(25) | 2(25) | 1(20) | 1(33.3) | 8(25) | |
| COT | S | 1(6.3) | 2(25) | 2(40) | 2(66.7) | 7(21.9) |
| R | 15(93.8) | 6(75) | 3(60) | 1(33.3) | 25(78.1) | |
| CTR | S | 8(50) | 2(25) | 4(80) | 2(66.7) | 16(50) |
| R | 8(50) | 6(75) | 1(20) | 1(33.3) | 16(50) | |
| CZM | S | 9(56.3) | 4(50.0) | 3(60) | 3(100) | 19(59.4) |
| R | 7(43.8) | 4(50.0) | 2(40) | 0 | 13(40.6) | |
| CXT | S | 12(75) | 4(50.0) | 5(100) | 3(100) | 24(75) |
| R | 4(25) | 4(50) | 0 | 0 | 8(25) | |
| CFP | S | 15(93.8) | 7(87.5) | 4(80) | 3(100) | 29(90.6) |
| R | 1(6.3) | 1(12.5) | 1(20) | 0 | 3(9.4) | |
| MER | S | 14(87.5) | 5(62.5) | 5(100) | 3(100) | 27(84.4) |
| R | 2(12.5) | 3(37.5) | 0 | 0 | 5(15.6) | |
| NIT | S | 13(81.3) | NA | NA | NA | 13(40.6) |
| R | 3(18.8) | NA | NA | NA | 3(9.4) | |
| Antibiotics | Patterns | Gram-Positive Bacterial Uropathogens | Total (n=7) | |
| S.aureus (n=4) | CoNS spp. (n=3) | |||
| No.% | No. % | No.% | ||
| AMP | S | 0(0) | 0(0) | 0(0) |
| R | 4(100) | 3(100) | 7(100) | |
| GEN | S | 2(50) | 2(66.7) | 4(57.1) |
| R | 2(50) | 1(33.3) | 3(42.9) | |
| CPR | S | 1(25) | 1(33.3) | 2(28.6) |
| R | 3(75) | 2(66.7) | 5(71.4) | |
| TET | S | 0(0) | 0(0) | 0(0) |
| R | 4(100) | 3(100) | 7(100) | |
| COT | S | 1(25) | 1(33.3) | 2(28.6) |
| R | 3(75) | 2(66.7%) | 5(71.4) | |
| CXT | S | 2(50) | 1(33.3%) | 3(42.9) |
| R | 2(50) | 2(66.7) | 4(57.1) | |
| CLN | S | 1(25) | 1(33.3) | 2(28.6) |
| R | 3(75) | 2(66.7) | 5(71.4) | |
Among Gram-positive isolates, S.aureus isolates showed a high rate of resistance to ampicillin (100%), tetracycline (100%), cotrimoxazole (75%), and gentamicin (50%). About 50% of the isolated S. aureus was methicillin-resistant (MRSA). Likewise, isolated coagulase-negative staphylococci (CoNS) were resistant to ampicillin (100%) and tetracycline (100%), and 33.3% of isolated CoNS were methicillin-resistant (MR-CoNS). Gentamicin (57.1%) showed better responses against isolated Gram-positive uropathogens (Table 4).
3.5. Multidrug-resistant Isolated Bacterial Uropathogens
Multidrug-resistance (MDR) was seen in 36/39 (92.3%) bacterial uropathogen isolates, and the overall magnitude of MDR uropathogens in the study population was 36/251 (14.3%), with a 95% CI (10-19.1). Gram-negative bacterial isolates accounted for 93.6% of MDR uropathogens. MDR was observed in 100% of K. pneumoniae, P. mirabilis, and S. aureus isolates, followed by E. coli 15/16 (93.7%), K. oxytoca 4/5 (80%), and CoNS 2/3 (66.7%) isolates (Table 5).
| Bacterial Isolates | Antibiogram Pattern (%) | |||||
| R1 | R2 | R3 | R4 | > R5 | Total (> R2) | |
| E. coli (n=16) | 1(6.3) | 5(31.3) | 7(43.6) | 2(12.5) | 1(6.3) | 15(93.7) |
| K. pneumoniae (n=8) | 0(0) | 1(12.5) | 4(50) | 2(25) | 1(12.5) | 8(100) |
| K. oxytoca (n=5) | 1(20) | 2(40) | 1(20) | 1(20) | 0(0) | 4(80) |
| P. mirabilis (n=3) | 0(0) | 2(66.7) | 1(33.3) | 0(0) | 0(0) | 3(100) |
| S. aureus (n=4) | 0(0) | 3(75) | 1(25) | 0(0) | 0(0) | 4(100) |
| CoNS (n=3) | 1(33.3) | 2(66.7) | 0(0) | 0(0) | 0(0) | 2(66.7) |
| Total (n=39) | 3(7.7) | 15(38.5) | 14(35.9) | 5(12.8) | 2(5.1) | 36(92.3) |
| Variables | Categories | MDR Uropathogen | COR [95% CI] | AOR [95% CI] | P-value | |
| Negative | Positive | |||||
| No. (%) | No. (%) | |||||
| Antibiotic usage in the last 6 months | Yes | 17 (6.8) | 11 (4.4) | 5.12 (2.15-12.17) | 6.7 (2.53-17.6)* | 0.001 |
| No | 198 (78.9) | 25 (10.0) | 1 | 1 | ||
| Presence of chronic underlying disease** | Yes | 6 (2.4) | 5 (2.0) | 5.62 (1.62-19.52) | 7.03 (1.72-28.73)* | 0.007 |
| No | 209 (83.3) | 31 (12.4) | 1 | 1 | ||
| Use of antibiotics without prescription | Yes | 13 (5.2) | 11 (4.4) | 6.84 (2.77-16.89) | 9.7 (3.53-26.675)* | 0.001 |
| No | 208 (80.5) | 25 (10.4) | 1 | 1 | ||
| CD4+ count (cells/mm3) | <250 | 10 (4.0) | 5 (2.0) | 4.77 (1.43-15.99) | 8.05 (2.07-31.15)* | 0.003 |
| 250-500 | 71 (28.3) | 17 (6.8) | 2.29 (1.10-4.92) | 2.49 (1.05-5.923)* | 0.038 | |
| >500 | 134 (53.4) | 14(5.6) | 1 | 1 | ||
Abbreviations: MDR, Multidrug-Resistant; COR, Crude Odd Ratio; AOR, Adjusted Odd Ratio; CI: Confidence Interval; CD4, Cluster of Differentiation 4.
3.6. Extended-Spectrum Beta-Lactamase (ESBL) Producer Gram-negative Uropathogens
Nine ESBL producers were found among 32 Gram-negative uropathogenic isolates evaluated using the double-disc synergy test method. In E. coli, the highest frequency was recorded and accounted for 15.7% (5/32) of the total, followed by K. pneumoniae, which accounted for 9.4% (3/32) of the total. The lowest frequency of ESBL production was seen in K. oxytoca isolates, i.e., 3.13% (1/32). In contrast, P. mirabilis isolates were ESBL non-producers.
3.7. Carbapenemase Producer Gram-negative Isolated Uropathogens
The modified Carbapenem Inactivation Method (mCIM) was used to evaluate carbapenemase producer Gram-negative uropathogenic isolates, and only 15.7% (5/32) of uropathogens were carbapenemase enzyme producers. K. pneumoniae was found in 9.4% of the cases (3/32), followed by E. coli accounted for 6.25% (2/32).
3.8. Factors Associated with MDR Uropathogens
Multivariable logistic regression analysis showed that a recent history of antibiotic use [AOR = 6.7, 95% CI: (2.53–17.6)], the presence of chronic underlying disease [AOR = 7.03, 95% CI: (1.72–28.73)], taking antibiotics without a prescription [AOR = 9.7, 95% CI: (3.53-26.675)], CD4+ count between 250 and 500 [AOR = 2.49, 95% CI: (1.05–5.1933)], and CD4+ count less than 250 cells/mm3 [AOR = 8.05, 95% CI: (2.078–31.154)] were found to be statistically significant associated with the development of MDR bacterial uropathogens (Table 6).
4. DISCUSSION
In this study, the total prevalence of bacterial uropathogens in HIV-positive adult patients was 15.5% (95% CI: 11.21–21.34). Gram-negative bacteria made up the majority of the isolates (82.1%). The most common isolate was E. coli (41%), followed by K. pneumoniae (20.5%), with isolated Gram-positive bacteria accounting for just 17.9% of the total, with S. aureus being the most common isolate (10.3%). K. oxytoca was the third most prevalent isolate next to K. pneumoniae, accounting for 12.8%. P. mirabilis and CoNS were the least prevalent isolates, with an isolation rate of 7.7%. This was in line with studies conducted in Kenya [26] and Ethiopia [1, 27, 28].
In this investigation, the total prevalence of MDR bacterial uropathogens was 14.3% [95% CI: (10-19.1)]. This finding was consistent with studies conducted in India (14.0%) [15], Wolayita, Ethiopia (10.6%) [18], and Gondar, Ethiopia (10.08%) [25]. However, the results were higher than those of studies conducted in Hawassa, Ethiopia (8.04%) [3], Addis Ababa, Ethiopia (8.89%) [7], and Nigeria (9%) [29]. In contrast, our finding was lower than the study done in Nigeria (41.9%) [23]. The disparity in results could be attributed to differences in the study population, methodology, and spread of resistant bacteria.
Gram-negative MDR bacteria were the predominant isolates, which accounted for 83.3% (30/36). E. coli (41.7%) was the most frequently exhibited MDR, followed by K. pneumoniae (22.2%), K. oxytoca (11.1%), and S. aureus (11.1%). Gram-negative bacterial isolates were found to be highly resistant to cotrimoxazole (78.1%), ampicillin (75%), and gentamicin (65.6%). This was in line with studies conducted in Ethiopia [3, 25, 30] and Cameroon [31].
ESBL producers were found in 30% (9/30) of Gram-negative MDR bacterial isolates. Of which, 16.7% (5/30), 10% (3/30), and 3.3% (1/30) accounted for E. coli, K. pneumoniae, and K. oxytoca, respectively. On the other hand, 16.7% (5/30) of Gram-negative MDR bacterial isolates were carbapenemase enzyme producers, accounted for K. pneumoniae 10% (3/30) and E. coli 6.7% (2/30). A higher prevalence of MDR was observed among extended-spectrum β-lactamases producing than non-producing isolates, which might be associated with large plasmids that frequently carry the ESBL marker and are capable of incorporating additional determinants coding for resistance to non-β-lactam antimicrobials [25, 31]. Likewise, 50% and 66.7% of isolated S. aureus and CoNS were methicillin-resistant, respectively. The overall finding was in line with the previous studies carried out in Ethiopia [1, 25, 26], Kenya [28], and Tanzania [32]. However, this finding disagreed with other studies done in Ethiopia [30], Nigeria [23], and South Africa [33]. The reason for this discrepancy could be the distribution difference between resistant bacteria and the practice of antibiotic prescription or the frequent prescription of broad-spectrum antibiotics for empirical therapy of ART patients.
In our study, HIV-positive adult patients with a recent history of antibiotic use were found to be 6.7 times more likely to develop an MDR uropathogen. This is similar to studies conducted in Chad [34], India [35], Nigeria [36], and Ethiopia [37]. Frequent antibiotic usage, indiscriminate antibiotic use for opportunistic infections, low drug adherence, and dependence on empiric antibiotic use are all possible reasons for this commonality. Likewise, we found that taking antibiotics without a prescription increased the risk of MDR-uropathogens by 9.7 times. This finding is in line with studies conducted in different parts of Ethiopia [25, 27]. The reason for the association might be that people have the habit of purchasing antibiotics from nearby drug stores without prescriptions from health professionals. This leads to the acquisition of MDR microbes due to the misuse of antibiotics.
Another factor, which had a statistically significant association with MDR bacterial uropathogens, was the presence of chronic underlying disease. HIV-positive adults with other chronic conditions had a 7.03-fold increased chance of getting MDR bacterial uropathogens. This is exhibited by another study evaluating risk factors for MDR acquisition in Addis Ababa, Ethiopia [25, 37]. Use of multiple antibiotics, the presence of autoimmune disease, antibiotic overuse or underuse, antibiotic-induced depletion of the protective commensal microbiota, and the introduction of different microbes during any medical procedure for the treatment of the underlying chronic condition are all possible causes.
Moreover, a CD4+ count below 500 cells/mm3 was statistically associated with MDR bacterial uropathogens. Patients whose CD4 count was between 250 and 500 cells/mm3 were 2.5 times more likely to acquire MDR uropathogens. On the other hand, patients with a CD4+ count of fewer than 250 cells/mm3 were eight times more likely to develop MDR uropathogens. This finding is consistent with most studies conducted in Ethiopia [3, 25], Nigeria [23], Kenya [28], and Cameroon [31]. Low immunity, rendering them more susceptible to opportunistic infection and re-infection, and the likelihood of using wide spectrum antibiotics as prophylactic for preventing opportunistic infections are the most plausible explanations for the increased risk of low CD4+ count.
CONCLUSION
The overall prevalence of MDR bacterial uropathogens among HIV patients in this study was comparable to previous studies. Antibiotic use in the last six months, the presence of a chronic underlying condition, antibiotic use without a prescription, and a CD4+ count of fewer than 500 cells/mm3 increase the odds of MDR uropathogens. Among Gram-negative and Gram-positive bacterial isolates, E. coli and S. aureus were the predominant uropathogens, respectively. More than half of Gram-negative bacterial isolates were ESBL and carbapenemase producers. Moreover, one-half and one-third of isolated S. aureus and CoNS were methicillin-resistant, respectively. The isolation of high MDR bacteria underscores the growing problem of untreatable UTIs, revealing a significant frequency of drug-resistant bacteria in both the community and the hospital environment. To mitigate the infection, measures, such as health education, regular bacterial screening, and antibiotic surveillance, are essential among this group of people.
LIMITATIONS OF THE STUDY
This study was limited as: (a) tests to determine the minimum inhibitory concentration (MIC) of the antimicrobials were not performed, and (b) the fact that the occasion of the use of cotrimoxazole as prophylaxis before sampling might also affect the result.
LIST OF ABBREVIATIONS
| AIDS | = Acquired Immunodeficiency Syndrome; |
| AOR | = Adjusted Odd Ratio; |
| ATCC | = American Type Culture Collection; |
| CD4+ | = Cluster of differentiation 4; |
| CoNS | = Coagulase Negative Staphylococcus; |
| COR | = Crude Odd Ratio; |
| ESB | = Extended-spectrum beta-lactamase; |
| HIV | = Human Immunodeficiency Virus; |
| MDR | = Multidrug-Resistant; |
| MRSA | = Methicillin-Resistant Staphylococcus aureus; |
| UTI | = Urinary Tract Infection. |
AUTHORS’ CONTRIBUTIONS
All authors contributed significantly to the conception, design, data acquisition, data analysis, interpretation, revision for important intellectual content, agreed to submit manuscript to the current journal, and agreed to be accountable for all aspects of the work.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The ethical approval was obtained from the Arba Minch University College of Medicine and Health Sciences Institutional Review Board (Ref. No. IRB/1042/2021). The Arba Minch General Hospital granted official clearance.
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
It was entirely voluntary to take part in the study. The goal of the study, ability to withdraw, and the confidentiality of the information acquired were all explained to all study participants. Before the interview, informed verbal and written consent were obtained.
STANDARDS OF REPORTING
STROBE guideline has been followed.
AVAILABILITY OF DATA AND MATERIALS
Due to ethical and confidentiality concerns, the data are not publicly available; however, it is available from the corresponding author upon reasonable request and with the consent of the ethics committee.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare that they have no conflicts of interest in this work.
ACKNOWLEDGEMENTS
The authors acknowledge the material and reagent support provided by Arba Minch University, Colleges of Medicine and Health Sciences, Department of Medical Laboratory Science. They are grateful to the staff at Arba Minch General Hospital as well as the study participants for their cooperation and willingness to participate.


