All published articles of this journal are available on ScienceDirect.
A Rare Case of Kawasaki-like Multisystem Inflammatory Syndrome following COVID-19 in an Adult
Abstract
In this case report, we describe a Kawasaki-like multisystem inflammatory syndrome (MIS-A) in a 33-year old man that occurred 19 days after a SARS-CoV-2 infection. The main features at presentation were profound myocarditis, bilateral non-purulent conjunctivitis, mediastinal lymphadenopathy, and acute kidney failure and laboratory evidence of hyperinflammation. He received ACE-inhibition and beta-blockers for his heart failure and made a fairly rapid spontaneous recovery over the subsequent 8 days.
1. INTRODUCTION
Kawasaki disease is an acute and usually self-limiting vasculitis of the medium calibre vessels, which exclusively affects children [1]. Although the underlying aetiology is unknown, various lines of evidence point to viral infections playing a central role in triggering the disease [1]. The COVID-19 pandemic has been associated with outbreaks of a Kawasaki-type disease that has also predominantly affected children. This has been termed the multisystem inflammatory syndrome in children (MIS-C). More recently, it has been appreciated that SARS-CoV-2 infection may also trigger a similar multisystem inflammatory syndrome in adults (MIS-A). We report a case of MIS-A in a 33-year old man with spontaneous resolution.
2. CASE REPORT
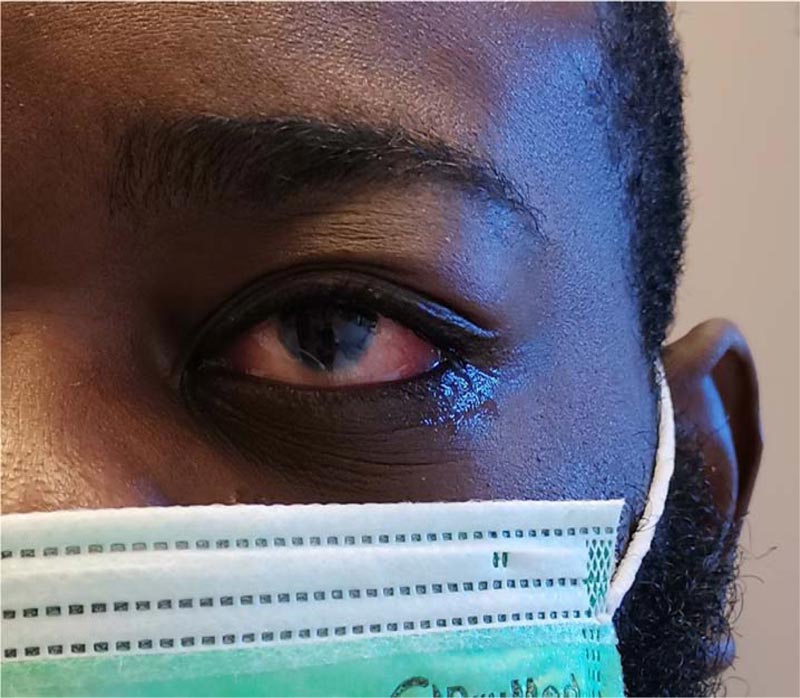
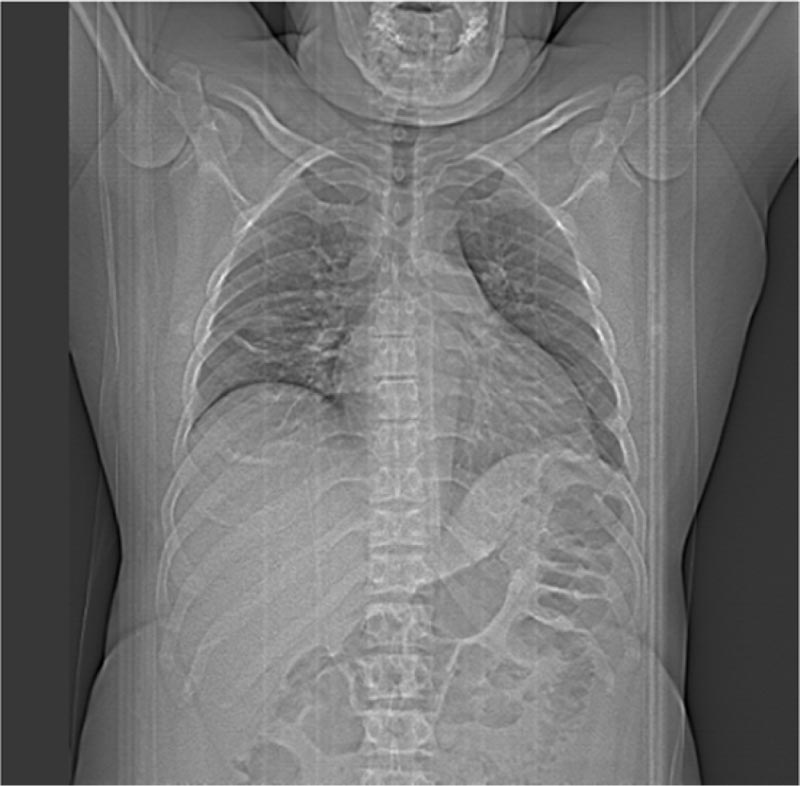
A 33-year-old male with no past medical history was presented to the emergency department with diarrhoea and abdominal pain. His nasopharyngeal swab was positive for SARS-CoV-2 and he was diagnosed with mild COVID-19 gastroenteritis. Apart from a body mass index (BMI) of 33,9 kg/m2 (<25,0 kg/m2), he had no risk factors for severe COVID-19. He was managed as an outpatient with paracetamol and within a week had complete resolution of his symptoms. Nineteen days after his initial presentation, he developed gradual onset orthopnoea, fever, myalgia and fatigue. These symptoms were progressive and necessitated ICU admission 5 days later with multiorgan failure. At this point, his predominant symptoms were severe dyspnoea and palpitations. On clinical examination, he was febrile, had bilateral non-purulent conjunctivitis (Fig. 1) and signs of heart failure. Laboratory results showed a type I respiratory failure with a pO2 of 79,2 mmHg (83,0-108,0 mmHg) on room air, and raised leucocytes 20,30 x109/L (3,50-9,80 x109/L), neutrophils 17,50 x109/L (1,56-7,08 x109/L), CRP 550 mg/L (< 6 mg/L), ferritin 5344 µg/L (< 275 µg/L), LDH 625 U/L (< 220 U/L), CK 418 U/L (< 200 U/L) and triglycerides 276 mg/dL (< 150 mg/dL) (Table 1). In addition, he had a marked elevation of his highly sensitive troponin 6834,7 ng/L (< 34,2 ng/L) and NT-pro BNP 10599,0 pg/mL (< 125,0 pg/mL). He was not oliguric but had biochemical evidence of acute kidney failure with a creatinine of 2,20 mg/dL (0,50-0,80 mg/dL), an estimated GFR (MDRD) of 35 mL/min/1,73m2 (>90 mL/min/1,73m2) and proteinuria 872,2 mg/24hours (<140,0 mg/24hours). A nasopharyngeal swab and bronchoalveolar lavage were both negative for SARS-CoV-2 on real-time polymerase chain reaction (RT-PCR). Bacterial blood cultures were negative. Echocardiogram revealed a reduced ejection fraction (LVEF) of 30%, mild left ventricular hypertrophy and a small pericardial effusion. His D-dimers were elevated 3,28 mg/L (<0,5 mg/L), but computed tomography angiography (CT-A) was negative for pulmonary embolism. This CT-scan did, however reveal paratracheal and subcarinal lymphadenopathy (Fig. 2).
Coronary angiography revealed normal coronary vasculature and magnetic resonance imaging of the heart showed no structural abnormalities or signs of inflammation (Fig. 3).
A diagnosis of a multisystem inflammatory syndrome (myocarditis, acute kidney failure, adenopathy and bilateral non-purulent conjunctivitis) with a Kawasaki-like disease prior to a recent SARS-CoV-2 infection was made. His heart failure was treated with beta blocker and ACE-inhibition. His fever and laboratory markers of hyperinflammation settled spontaneously over the course of a week and thus, no immune-modulatory therapy was commenced. By the time of his discharge 8 days after admission, his cardiac left ventricular ejection fraction had recovered to 62%.
| Laboratory Results | Hospital Admission (Day 0) |
Hospital Discharge (Day 8) |
Reference Values |
|---|---|---|---|
| Leucocytes (x109/L) | 20,30 | 13,20 | 3,50 – 9,80 |
| Neutrofils (x109/L) | 17,50 | 18,01 | 1,56 – 7,08 |
| CRP (mg/L) | 550 | 63,6 | < 6 |
| Ferritin (µg/L) | 5344 | N.A. | < 275 |
| LDH (U/L) | 625 | 437 | < 220 |
| CK (U/L) | 418 | 122 | < 200 |
| Triglycerides (mg/dL) | 276 | N.A. | < 150 |
| Hs-troponins (ng/L) | 4809,1 | 136,0 | < 34,2 |
| NT-pro BNP (pg/mL) | 10599,0 | N.A. | < 125,0 |

3. DISCUSSION
COVID-19 is characterized by a number of inflammatory syndromes [2]. In adults, the most common of these is COVID-19-associated hyperinflammatory syndrome (cHIS), which occurs 10-12 days following symptom onset. It typically effects patients with proinflammatory conditions, for example, obesity and diabetes. This cytokine release storm is often complicated by respiratory failure, cardiac pathology and thrombotic events [2-5]. Therapy is mainly immunosuppressive with options including corticosteroids, immunoglobulins, selective cytokine blockade and JAK-inhibition [5]. cHIS is very rare in children, who are more likely to have pauci- or asymptomatic SARS-CoV-2 infection. One of the possible explanations is a less developed adaptive immune system and a more robust innate immune response [2].
On the other hand, a small proportion of children and an even smaller proportion of adults have been noted to develop a Kawasaki-type illness 2 to 5 weeks after onset of COVID-19 symptoms [2, 6, 7]. Various features, including the timing of this multisystem inflammatory syndrome in children/adults (MIS-C/A) suggest that it is mediated by an abnormal immunological response that triggers systemic hyperinflammation [2, 6].
In spite of similar features, there are many differences between Kawasaki disease and MIS-C/A. Some of these include older age, impairment of cardiac function and less coronary involvement in MIS-C/A [8].
Authors from the Centers for Disease Control and Prevention (CDC) have recently reviewed the presentation of 27 adults with MIS-A [5]. They proposed as a working case definition that cases should fulfill the following five criteria: 1) a severe illness requiring hospitalization in a person aged ≥21 years; 2) a positive test result for current or previous SARS-CoV-2 infection during admission or in the previous 12 weeks; 3) severe dysfunction of one or more extrapulmonary organ systems (e.g., hypotension or shock, cardiac dysfunction, arterial or venous thrombosis or thromboembolism, or acute liver injury); 4) laboratory evidence of severe inflammation (e.g., elevated CRP, ferritin, D-dimer, or interleukin-6); 5) absence of severe respiratory illness. Patients should also not have alternative diagnoses such as bacterial sepsis [2]. They found that the cases presented with a variety of cardiovascular, gastrointestinal, dermatologic, and neurologic symptoms. Only 9 patients had no underlying medical conditions. Of note six were, like our patient, obese [6]. All the patients had evidence of cardiac dysfunction and 13 had gastrointestinal symptoms on admission. Ten patients required ICU admission. Two patients died and the long-term follow-up was not reported for the remainder who were all discharged from the hospital. An autopsy performed on one of the deceased revealed evidence of mediastinal adenopathy, small-vessel vasculitis, particularly in the cardiac vasculature and new pulmonary thrombi [2].
Our patient met the suggested CDC definition of MIS-A. His laboratory findings were also typical for MIS-A with cardiac and renal involvement – very high elevations of CRP, ferritin, triglyceride, troponins and NT-pro BNP [6]. His age of thirty-three is similar to the other cases in current literature. He had no typical polymorphous exanthema or mucosal involvement seen in classic Kawasaki disease [9]. His fairly rapid spontaneous recovery without immunomodulatory therapy is fairly novel [6]. We cannot however be sure that the preceding SARS COV-2 infection was the cause of his presentation. The optimal therapy for MIS-C/A is unknown, but in the CDC case series, 7 were treated with intravenous immunoglobulin (standard therapy for Kawasaki disease) and 2 with tocilizimab (IL-6 inhibitor). Alternative IL-6 inhibitors such as sarilumab and siltuximab may also be considered. A recent case series describes 5 adults with MIS-A myocarditis similar to our patient. All of them were admitted to the ICU and two needed intravenous immunoglobulins. No deaths were reported [10]. A recent observational study of 111 individuals with MIS-C found that children treated with IVIG and methylprednisolone vs IVIG alone had a favorable clinical course [11]. On the basis of these findings, certain guidelines now recommend combined IVIG/corticosteroids for moderate or severe MIS-C [12].
CONCLUSION
This case report should serve as a reminder for clinicians to consider MIS-A in adults with signs and symptoms compatible with the current working case definition. If nucleic acid testing for SARS-CoV-2 is negative, serological or antigen tests should be performed. Although there are no randomized controlled trials as to the benefit of intravenous globulins, the evidence from Kawasaki disease suggests that this could be beneficial in fulminant cases or cases without spontaneous resolution [3-6]. Further studies are required to investigate the role of specific therapies like immunoglobulins, corticosteroids and selective cytokine blockade (tocilizumab and anakinra) [10]. In the context of active vaccination against SARS-CoV-2 infection we need to be aware of MIS-C/A as a possible adverse event of immunization [8].
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Written and informed consent from the patient was obtained for publishing this case report.
STANDARDS OF REPORTING
CARE guidelines were followed to conduct this study.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


