All published articles of this journal are available on ScienceDirect.
Identification of Immunogenic Candidate for New Serological Tests for Brucella melitensis by a Proteomic Approach
Abstract
Background:
The diagnosis of brucellosis by serological tests is based on antigen suspensions derived from smooth lipopolysaccharide extracts, which can give false positive results linked to cross-reactivity with other Gram-negative microorganisms, especially Yersinia enterocolitica O:9 and Escherichia coli O157:H7.
Objective:
The objective of the present study was the characterization by proteomic analysis of specific immunogenic proteins not associated with smooth lipopolysaccharide to improve the diagnostic tests used in the ovine brucellosis eradication programs.
Methods:
The serum from a sheep positive to Brucella melitensis was treated to eliminate all antibodies against such lipopolysaccharide and highlight the reaction towards the immunoreactive proteins in Western Blotting.
Results:
The immunoreactive bands were identified by nLC-MS/MS and through bioinformatic tools, it was possible to select 12 potential candidates as protein antigens specific for Brucella melitensis.
Conclusion:
The detection of new antigens not subjected to cross-reactivity with other Gram-negative microorganisms can offer an additional tool for the serological diagnosis of such disease.
1. INTRODUCTION
The brucellosis can affect humans by the contact with ill animals and their parts, such as carcasses or abortion derivatives, and by consumption of contaminated raw milk and unpasteurized dairy products [1]. It is characterized by an acute febrile status known as undulant fever, chills or shaking rigors, malaise, generalized aches, headache, anorexia, and general weakness [2], and other long term clinical signs such as arthralgia, myalgia, hepatomegaly or hepatitis, endocarditis, and meningitis [3]. The effectiveness of the control and prevention of brucellosis depends on the timely diagnosis and isolation of infected animals. The serological tests currently in use for its diagnosis are described in the OIE Manual [4] and internationally recognized. Such tests are based on antigen suspensions derived from smooth Brucella or lipopolysaccharide extracts, but they can result in false-positive serological reactions if the animal has developed antibodies against other Gram-negative bacteria, in particular, Yersinia enterocolitica O:9 and Escherichia coli O157:H7 [5]. The cause of this cross-reactivity is due to the presence of smooth lipopolysaccharide (sLPS). It is a dominant component of almost all Gram-negative bacterial outer membranes, generally consisting of a predominantly lipophilic component lipid A, a core oligosaccharide part and O-polysaccharide (OPS) section called O-antigen [6]. The O-antigens are highly immunogenic and conserved among different Gram-negative strains and species, and therefore antibodies developed against sLPS of E. coli or Y. enterocolitica can also bind Brucella LPS. Therefore, the analyses of Brucella strains at protein level can serve to identify Brucella specific immunogenic targets, that are required to improve the current serological tests to better support the disease eradication plans.
In the present study, Brucella melitensis (BM) immunogenic proteins were investigated to detect epitopes able to distinguish true BM positive animals from those infected with Y. enterocolitica O:9 and E. coli O157:H7, the most important Brucella cross-reactive bacteria. The first step was the localization of proteins to discard the cytosolic ones, because the proteins of the envelope are those that are into contact with the host immune system and stimulate the production of antibodies. Such envelope proteins were studied for their immunogenic potential with the aim to discard the homologue proteins with the major cross-reactive Gram-negative bacteria. Then, a depletion approach was adopted: a BM positive serum was pre-treated with purified sLPS to remove all anti-sLPS antibodies. Western Blotting (WB) analyses were performed to detect specific immunoreactive bands of BM not associated with sLPS. The immunoreactive bands were identified by nLC-ESI-MS/MS and the exposed B-cell epitopes were predicted through bioinformatics tools, as reported by Paci et al. [7]. The bioinformatics pipeline, used to analyze the data obtained by mass spectrometry, was developed to identify potential B-cell target protein candidates for serological diagnosis.
2. MATERIALS AND METHODS
The bacterial strains (BM 16 M), reference antisera and the cultivation of Brucella for proteomic analyses were the same used by Paci et al. [7]. To identify BM immunogenic proteins that are not associated with sLPS, the positive serum for BM was treated for 4 h at 37 °C, with sLPS antigen produced according to OIE Manual.
BM total proteins (5 µg/well) were separated by SDS-PAGE using NuPage® 4-12% Bis-Tris pre-cast gels (Life Technologies) at a constant voltage of 200 V [7, 8] and transferred onto a nitrocellulose membrane using the iBlot Dry Blotting system (Life Technologies) [9]. After blocking with 5% skim milk in PBS + 0.05% Tween 20 (PBST) for 2 h, the membranes were cut into strips, which were incubated with untreated and sLPS-treated sera diluted 1:100 in PBST + 2.5% skim milk and then with Protein G-peroxidase conjugate (Sigma) diluted 1:100000 for 1 h, at room temperature.
The antigen-antibody reactions were visualized by adding the AmershamTM ECL SelectTM Western Blotting Detection Reagent (GE Healthcare) and the images were acquired using the ChemiDoc MP (Bio-Rad) and the Image Lab Software, version 4.0 (Bio-Rad).
Mass spectrometry analyses were performed at IFOM (the FIRC Institute of Molecular Oncology, Milan - Italy) facilities. Two slices of SDS-PAGE gel were excised; then proteins were treated with 10 mM DTT and 55 mM IAA, and they were digested with trypsin. After addiction of 0.1% formic acid, peptides were concentrated and analyzed by nLC-MS/MS using an UPLC EASY-nLC 1000 connected to a quadrupole Orbitrap QExactive-HF (Thermo Fisher) and with a gradient (from 5 to 100% B) of 2% acetonitrile/0.1% formic acid (eluent A) and 80% acetonitrile/0.1% formic acid (eluent B) [10].
Raw data were processed with Proteome Discoverer (version 1.4.1.14, Thermo Scientific) and Mascot (version 2.6.0, Matrix Science), searching against the BM 16M. The obtained data were examined with Scaffold (version 4.8.9, Proteome Software Inc.) and only proteins with greater than 99.0% probability and containing at least 3 peptides (greater than 95% probability) were accepted. Therefore, only proteins detected in at least 2 out of 3 biological replicates were included in bioinformatic analysis. The data were examined using the following software: LipoP 1.0 Server [11]; TMHMM Server version 2.0 [12, 13] and SignalP 4.1 Server [14]; PSORTb version 3.0.2 [15] and CELLO version 2.5 [16, 17].
In order to evaluate the protein antigenicity, the non-cytosolic proteins were examined using Vaxign tool [18-20] and VaxiJen [21]. Such software permitted the prediction of protective antigens, but Vaxign tool integrated open source and internally developed programs and it was specific to vaccine target prediction, while VaxiJen analyzed physicochemical properties of proteins without the need to sequence alignment. The proteins with adhesion scores (obtained by Vaxign tool) higher than 0.5 and between 0.4 and 0.5 were considered antigens with high and intermediate antigenic potential, respectively [22]. The adhesion score measured the probability that the protein is attached to the host cells and it is mediated by adhesins, which are important for bacterial colonization and could be target for vaccine development [23].
Furthermore, high potential antigenic proteins were analyzed by BLASTp to eliminate BM proteins homologous with those of Y. enterocolitica O:9 (taxid:630), E. coli O157:H7 (taxid:562) and proteins similar to those of the Bovidae family (identity greater than 35%). MFDP2 version 2.00 Disorder predictor [24] was used to eliminate proteins with disorder more than 25%.
NetSurfP version 1.1 [25] and BepiPred version 1.0 Server [26] were used to predict the protein secondary structure and B-cell linear epitopes, respectively.
3. RESULTS AND DISCUSSION
3.1. Western Blotting
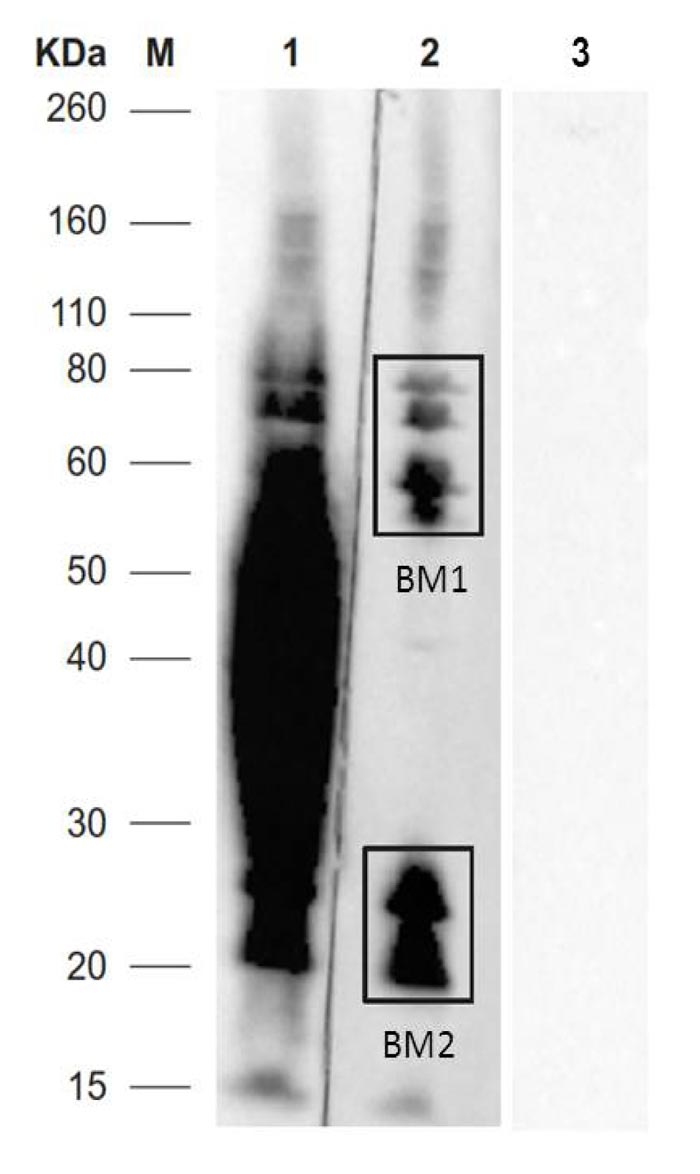
Untreated sera reacted with proteins with molecular weight ranging between 160 and 15 kDa, while sLPS treated sera reacted with 2 areas at 160-55 kDa (BM1) and 18-30 kDa (BM2). Areas with MW at 80-55 kDa (BM1), and 30-18 kDa (BM2) were examined by nLC-ESI-MS/MS. Protein bands with MW ranging from 55 and 30 kDa were recognized by antibodies vs sLPS.
The negative serum samples did not show a reaction with the BM protein extracts (Fig. 1).
3.2. Mass Spectrometry Analysis (nLC-ESI-MS/MS)
The number of proteins identified by mass spectrometry in BM1 and BM2 SDS-PAGE gel area was 526 and 432, respectively. Some proteins were present in both bands and the repeated ones were eliminated. According to the validation criteria, 737 proteins were identified. Further details on the identified proteins were provided as Supplementary material.
3.3. Prediction of Brucella melitensis Immunogenic Proteins
In order to identify BM immunogenic proteins for the improvement of the current serological tests, immunoreactive bands not associated with sLPS were identified by WB and subsequently analyzed by nLC-ESI-MS/MS.
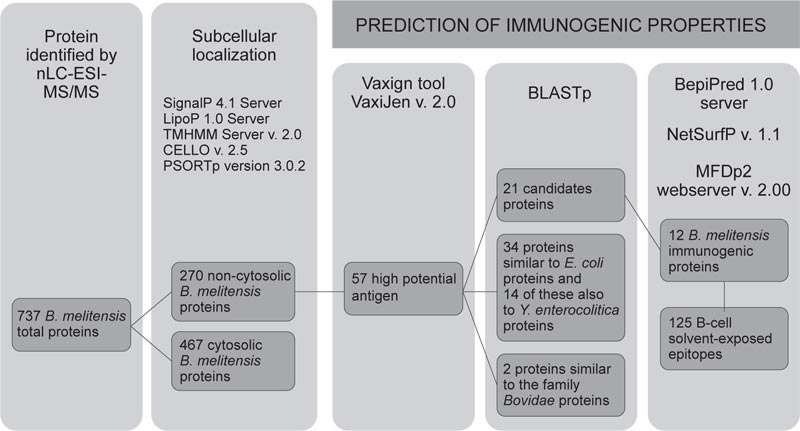
In Fig. (2), the workflow for the prediction of protein candidates was presented. A total of 737 proteins were identified in BM1 and BM2 gel slices (Fig. 1), of which 270 (36.6% of total proteins) were envelope proteins. Then, 69 with “high potential of antigenicity” were identified using the Vaxign tool (adhesion probability greater than 0.5). Among these, 12 proteins were discarded because they showed similarities with other proteins present in humans, pigs, and mice.
Furthermore, 34 proteins showing similarity (identity greater than 35% by BLASTp) with Y. enterocolitica and/or E. coli and with proteins belonging to the family Bovidae (2) were discarded. It is reported that Y. enterocolitica O:9 and E. coli O157:H7 are the most important cross-reactive bacteria with Brucella.
Additional proteins were discarded as previously investigated and resulting cross-reactive (2 proteins: Q8YE89, Q8YHH8 reported by Liang et al. [27]) or showing an unacceptable degree of disorder (7 proteins had a degree of disorder more than 25%, by MFDp2). Disorder proteins lack fixed three-dimensional structures, which is required for the correct and unique function of a protein. These proteins need special tools to be analyzed, although they are functionally important proteins and complement the function of ordered proteins [28]. Table 1 showed the 12 candidate proteins identified and ordered according to their predicted immunogenicity. BepiPred predicted 191 epitopes of BM, NetSurfP and THMM 125 exposed epitopes.
|
UniProt Accession Number |
Protein Name | Adhesion Probabilitya | Protective Antigenb | N° solvent exposed epitope |
Y. enterocolitica proteins Cover-identity % |
E. coli proteins Cover-identity % |
|---|---|---|---|---|---|---|
| Q8YDS0 | Heme transporter BhuA | 0.713 | 0.602 | 19 | 42–26.6 | 38–30.2 |
| Q8YHY8 | Metal chelate outer membrane receptor | 0.650 | 0.631 | 17 | 97–25.7 | 95–29.8 |
| Q8YGF8 | Hypothetical protein | 0.619 | 0.696 | 13 | NSSc | NSSc |
| Q8YDZ8 | Uncharacterized protein | 0.517 | 0.729 | 7 | NSS | NSS |
| C0REV2 | Porin opacity type | 0.743 | 0.629 | 7 | NSS | 93–30.3 |
| Q8YF11 | Trehalose/maltose binding protein | 0.702 | 0.497 | 13 | 11–25.0 | 77–24.8 |
| Q8YG56 | Porin Omp2b | 0.602 | 0.666 | 7 | NSS | NSS |
| Q8YH42 | Membrane lipoprotein lipid attachment site containing protein | 0.736 | 0.510 | 8 | 96–24.2 | 95–23.3 |
| Q8YID7 | Uncharacterized protein | 0.682 | 0.541 | 7 | NSS | 93–26.0 |
| C0RMW9 | Extracellular ligand-binding receptor | 0.609 | 0.588 | 5 | 89–22.9 | 91–24.5 |
| Q8YBP0 | Putative ABC transporter peptide-binding protein | 0.509 | 0.491 | 9 | 57–29.5 | 85–32.4 |
| Q8YIM8 | Uncharacterized protein | 0.504 | 0.496 | 13 | NSS | NSS |
The control of brucellosis is primarily based on its rapid and precise diagnosis, but the conventional methods require a long time of analysis and biosafety level 3 laboratories. For this reason, the serological tests are usually preferred, even if they have a low specificity due to cross-reactivity with other Gram-negative bacteria resulting in false positive reactions [29]. More recently, immunoproteomic methods able to identify new antigens of interest for serodiagnosis have been implemented, not only for Brucella abortus, but also BM and Brucella suis [30]. In the present study, proteomics and bioinformatics analyses focusing on BM envelope proteins were combined to identify BM specific immunogenic targets and improve sensitivity and specificity of the current serological tests. Then, 12 BM immunogenic proteins potentially able to discriminate BM infected animals from those infected by Y. enterocolitica and E. coli were identified. Seven of these were predicted as outer membrane proteins (OMPs); the remaining 5 were predicted as periplasmic. Four out of 12 immunogenic proteins are involved in the transport of substances; the best known is Porin OMP2b (Q8YG56) that allows the small hydrophilic materials to across the outer membrane through the formed diffusion pores [31]. For the other proteins, there are no data available in the literature on their immunogenicity.
This study demonstrated that bioinformatic analysis is a faster and cheaper approach to identify potential targets than traditional laboratory identification techniques also for microorganisms belonging to risk group 3, such as BM. However, the bioinformatic software remains only a predictive tool and the obtained results should be confirmed by the conventional methods.
CONCLUSION
The improvement of serological tests for brucellosis persists as an urgent need for a better diagnosis and control of the disease. The combination of immunoproteomic approach and subsequent in silico analyses to screen B-cell epitopes identified 12 potential candidates as protein antigens specific for BM. The development of diagnostic tests based on new serological antigens not subjected to cross-reactivity with other Gram-negative bacteria can offer an additional tool for the eradication of ovine brucellosis.
AUTHORS’ CONTRIBUTIONS
VP and ML designed the study. VP, IK and FDO performed the experiments. VP, PV, IK, TDF, FP, CDP, FDO, MS, MT and ML performed the data analysis. VP, PV, IK, MS and ML drafted the manuscript. All authors contributed to the interpretation of the results, provided critical feedback, contributed to the writing of the manuscript and have approved the final version.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The authors confirm that the data supporting the findings of this research are available within the article.
FUNDING
This research was funded by the University of Teramo “PROGETTO FARDIB 2019”.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


