All published articles of this journal are available on ScienceDirect.
Pimozide Inhibits the AcrAB-TolC Efflux Pump in Escherichia coli
Abstract
Efflux pump inhibitors (EPIs) are attractive compounds to reverse multidrug-resistance in clinically relevant bacterial pathogens. In this study we tested the ability of the neuroleptic drug pimozide to inhibit the Escherichia coli AcrAB-TolC efflux pump, whose overproduction confers resistance to various antimicrobial agents. A real-time Nile red efflux assay in the AcrAB – overproducing strain 3-AG100 revealed that pimozide was capable of full inhibition of this pump at a concentration of 100 µM, which is far below its intrinsic MIC (>1mM). However, MIC assay demonstrated very little effect of pimozide with regard to reduction in MICs of various antimicrobial compounds.
Only oxacillin MICs were reduced twofold in the presence of pimozide at 100 and 200 µM.
Since pimozide did considerably enhance accumulation of ethidium bromide in a fluorescence assay, ethidium bromide MIC assays in the presence and absence of this putative EPI were performed. They revealed that pimozide was able to reduce the MICs of ethidium bromide by 4-fold. In line with previous reports we suggest that the capability of EPIs to restore the susceptibility to antimicrobial agents can be highly substrate-specific due to different substrate binding sites.
INTRODUCTION
The Escherichia coli AcrB multidrug efflux pump is a membrane protein that recognizes many chemically unrelated compounds including various dyes and antibiotics and expels them in collaboration with the so called membrane fusion protein AcrA and the outer membrane protein TolC which are both needed for function. The pump belongs to the resistance-nodulation-division (RND) family of efflux pumps that are common in gram-negative microorganisms. Based on various X-ray crystallographic, mutagenic and biochemical studies the tripartite efflux pump AcrAB-TolC is one of the best characterized bacterial efflux pumps [1-10]. It is also the only RND pump in E. coli that contributes significantly to antibiotic resistance against clinically relevant antibiotics due to its constitutive expression. Exposure to antimicrobial agents can further increase efflux-mediated resistance due to mutations in the acrR or marR gene leading to upregulation of the efflux pump [11]. Since the above mutations can relatively rapidly be selected by exposure to only one antibiotic and since the substrate spectrum of AcrAB-TolC is so large, generation of multidrug resistance via efflux of antimicrobial compounds is a real threat to treatment options in the clinic.
Since the antimicrobial drug pipeline is relatively empty – especially against gram-negatives – efflux pump inhibitors (EPIs) that revert multidrug resistance are an attractive target. Interestingly, several working groups have recently described psychotropic drugs, namely phenothiazines [12, 13] and selective serotonine reuptake inhibitors (SSRIs) [14] that are capable of inhibiting the AcrAB-TolC efflux pump in E. coli. By screening more psychotropic drugs we discovered that the neuroleptic drug pimozide is also a model efflux pump inhibitor.
MATERIALS AND METHODS
Bacterial Strains and Growth Conditions
E. coli strains 3-AG100 (a multidrug-resistant mutant (gyrAmarR) with acrB overexpression obtained from E. coli K-12 strain AG100 after repeated exposure to a fluoroquinolone) and the acrB knockout strain 1-DC14 [15] were grown in LB broth (1% tryptone, 0.5% yeast extract, and 1% NaCl), or on LB agar (1.5%) plates (Roth, Karlsuhe, Germany).
Chemicals
All chemicals were obtained from Sigma-Aldrich (Taufkirchen, Germany). Since pimozide is poorly soluble in water it was dissolved in hot lactic acid (0.03 %).
Susceptibility Testing
The MICs of a range of antimicrobial agents in the presence and absence of pimozide were determined in a 96-well microtiter plate using strains incubated overnight at 37°C in a volume of 100µl/well by a standard LB broth microdilution procedure and a final inoculum of 5 X 105 CFU/ml. MIC testing was done in triplicate. Custom 96-well microtiter plates containing selected antimicrobials at increasing concentrations were purchased from Merlin Diagnostics (Bornheim, Germany).
RESULTS
Fluorescence Assays
The protocols for the Nile red efflux [16] and ethidium bromide (EtBr) accumulation [17] assay have been published previously. The only modification was that the standard 20 mM potassium phosphate buffer (pH 7) amended with 1 mM MgCl2 used in the Nile red efflux assay was also used in the ethidium bromide accumulation assay. Moreover, the EtBr concentration was increased to 10 µM.
Using AcrAB-TolC overproducing strain 3-AG100 pimozide at concentrations of up to 200 µM did not reduce the MIC of the following antibiotics: tetracycline, chloramphenicol, rifampicin, and linezolid (Table 1). Oxacillin MICs were reduced twofold at 200 µM but not at 100 µM. The levofloxacin MICs behaved somewhat paradoxical with twofold reduction at 100 µM but no reduction at 200 µM.
Synergy of Pimozide with Selected Antibacterial Drugs and Ethidium in E. coli.
| Strain | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Levofloxacin | Tetracycline | Chloramphenicol | Rifampicin | Oxacillin | Linezolid | EtBr | |||||||||||||||
| No EPI | + pimozide 100 µM | + pimozide 200 µM | No EPI | + pimozide 100 µM | + pimozide 200 µM | No EPI | + pimozide 100 µM | + pimozide 200 µM | No EPI | + pimozide 100 µM | + pimozide 200 µM | No EPI | + pimozide 100 µM | + pimozide 200 µM | No EPI | + pimozide 100 µM | + pimozide 200 µM | No EPI | + pimozide 100 µM | + pimozide 200 µM | |
| 3-AG100 | 2 | 1 | 2 | 4 | 4 | 4 | 16 | 16 | 16 | 16 | 16 | 16 | 512 | 256 | 256 | 1024 | 1024 | 1024 | 512 | 256 | 128 |
| 1-DC14 | 0.125 | n.g. a) | n.g. | 0.5 | n.g. | n.g. | 1 | n.g. | n.g. | 4 | n.g. | n.g. | 0.5 | n.g. | n.g. | 16 | n.g. | n.g. | 8 | n.g. | n.g. |
a) no growth.
Adding the dye EtBr to the MIC panel, we determined that EtBr MICs were reduced 4-fold. The intrinsic MIC for pimozide was found to be > 1000 µM, whereas in the AcrB-deficient strain 1-DC14 it was 100 µM.
Nile Red Efflux and EtBr Accumulation Assays
Notable retardation of Nile red efflux in strain 3-AG100 was observed in the presence of pimozide at a threshold concentration of 25 µM (Fig. 1). Adding higher concentration of pimozid led to increasing efflux retardation in a dose-dependent manner. At 100 µM Nile red efflux was almost completely abolished.
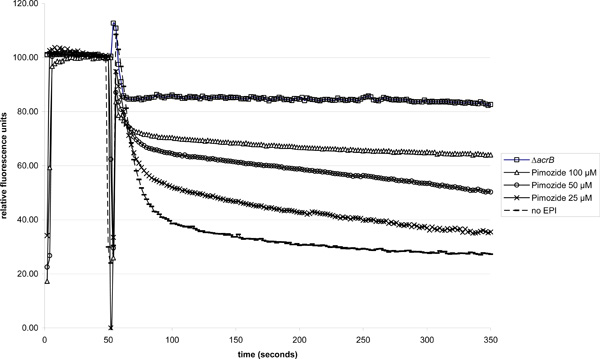
Dose-dependent Nile red efflux inhibition in 3-AG100 in the presence of pimozide (ΔacrB strain 1-DC14 used for comparison). Addition of glucose (final concentration 50 mM) 50 seconds into the experiment to trigger efflux.
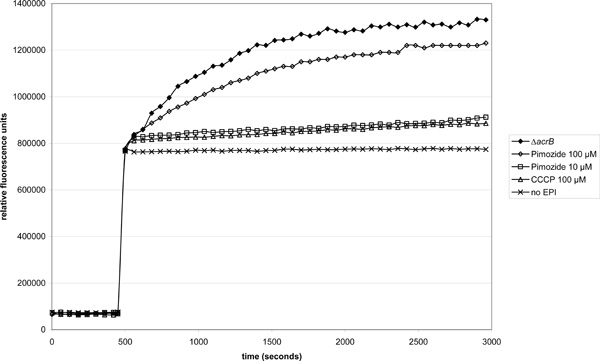
Effect of pimozide on EtBr accumulation in the acrAB-overexpressing strain 3-AG100 (ΔacrB strain 1-DC14 used for comparison). Pimozide or CCCP were added 50 seconds and EtBr (final concentration 10 µM) 500 seconds into the experiment. 50 mM glucose was present in the buffer throughout the assay.
Adding the pimozide just 50 s before energization yielded the same results. Controls with 0.03 % lactic acid and no pimozide did not alter the substrate efflux.
In line with these observations, 100 µM pimozide led also to a pronounced increase in EtBr accumulation that was even stronger than 100 µM carbonyl cyanide m-chlorophenyl hydrazone (CCCP) (Fig. 2).
DISCUSSION
In the Nile red efflux and EtBr accumulation assays the neuroleptic drug pimozide behaved like an EPI, in the case of Nile red comparable to Phe-Arg-β-naphthylamine (PAßN) [18]. However, the effect in the MIC assays was generally much less pronounced. Only in the case of EtBr could a fourfold reduction in MIC be observed. It thus seems that the effect of pimozide is highly substrate-specific. Such behaviour has been described before in the well-studied EPI PAßN. Although this EPI potentiates the activity of a wide variety of antimicrobial compounds it fails to do so with ethidium bromide [19]. As recently suggested by molecular docking [20] and X-ray crystallographic [8] studies we hypothesize that the observed discrepancy is mainly due to different EPI and substrate binding sites so that the activity of a given EPI might be highly substrate specific. It seems that pimozide is an extreme case in this regard since we could only demonstrate that it interferes with the transport of EtBr and Nile red but not with a range of clinically relevant antimicrobials.
While this observation and the fact that the plasma peak levels of this drug required for EPI activity cannot be safely reached in humans preclude its clinical usefulness, we suggest that pimozide can still be valuable as a “narrow-spectrum” model EPI to learn more about substrate – EPI interactions.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflicts of interest.
ACKNOWLEDGEMENTS
Declared none.


