RESEARCH ARTICLE
Study Prevalence of Verotoxigenic E.coli Isolated from Urinary Tract Infections (UTIs) in an Iranian Children Hospital
Masoumeh Navidinia1, Abdollah Karimi1, Mohammad Rahbar2, 3, *, Fatemeh Fallah1, Raheleh Radmanesh Ahsani1, Mohammad Ali Malekan1, Mana Hadipour Jahromi1, Zari GholineJad1
Article Information
Identifiers and Pagination:
Year: 2012Volume: 6
First Page: 1
Last Page: 4
Publisher ID: TOMICROJ-6-1
DOI: 10.2174/1874285801206010001
Article History:
Received Date: 07/5/2011Revision Received Date: 15/8/2011
Acceptance Date: 19/8/2011
Electronic publication date: 23/1/2012
Collection year: 2012

open-access license: This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
Abstract
Background and Objectives:
Urinary tract infections (UTI) caused by enterohemorrhagic Escherichia coli (EHEC) is one of the most important diseases in infants and children. If there would not be any useful diagnosis and treatment it may be resulted in diseases such as acute renal failure, thrombocytopenia and hemolytic anemia. The aim of this study was to determine frequency of verotoxigenic E.coli isolates in urine of children with (UTIs) in Mofid children Hospital.
Methods:
During one year from September 2008 to august 2009, urine specimens were taken from children who suspected to UTI admitted to Mofid Children Hospital. E.coli strains that indicated beta hemolytic on sheep blood agar, negative sorbitol fermentation on SMAC (sorbitol macconky agar) and negative motility on SIM were tested by PCR and serologic (VITEC-RPLA kit) methods for detecting toxin genes and production of toxin, respectively.
Results:
Among 12572 urine specimens were taken from children admitted to Mofid hospital, we isolated 378 E.coli from urine samples which only 9 isolates were EHEC. Only five EHEC strains (55%) which produced vtx genes, were detected by serologic and PCR methods.
Conclusion:
The prevalence of urinary infections caused by EHEC strains is very significant because it causes aggravating pathologic effects. Thus we suggest rapid method for identification of this bacteria and proper treatment to Inhibition of unwanted complications.
INTRODUCTION
Escherichia coli is the predominant facultative anaerobe of the human colonic flora. The organism typically colonizes the infant gastrointestinal tract within hours of life, and, thereafter, E. coli and the host derive mutual benefit [1].Verotoxigenic Escherichia coli (VTEC) are named for their ability to produce toxins with a cytopathic effect on Vero cells. They are also referred to as shiga toxin-producing E coli (STEC) because their cytotoxins closely resemble the shiga toxin produced by Shigella dysenteriae 1. Two types of shiga toxin are produced: shiga toxin 1 (Stx1), which is most closely related to that produced by S. dysenteriae, and shiga toxin 2 (Stx2), which has several variants. Entero-hemorrhagic E coli (EHEC) are the subgroup of VTEC organisms with demonstrated human pathogenicity. While most VTEC belong to E coli serogroup O157, verotoxin production has been identified in more than 200 serogroup [2]. Hemolytic uremic syndrome is the most common cause of acute renal failure in children, and the incidence of this syndrome in children is increasing worldwide [3]. E. coli O157:H7 is believed to cause more than 80 percent of the STEC infections that lead to hemolytic uremic syndrome [4]. Hemolytic uremic syndrome primarily occurs in children one to 10 years of age [5,6] with an average annual incidence of one to three cases per 100,000 children [7] and a survival rate of nearly 95 percent. Some studies indicate that rural populations are more at risk than urban populations [8,9] and the incidence is higher in warmer months of year, peaking from June to September [10].
Occurrences may be sporadic or present as an outbreak. A study conducted in the United Kingdom, in which confections intentionally were artificially contaminated with E. coli O157:H7 showed that the Shiga toxin-producing strains could survive for as long as one year, depending on storage conditions [11]. Three to 15 percent of persons who have STEC with diarrhea can develop hemolytic uremic syndrome [12]. Young children and older persons with altered immune response, [13] as well as persons who have been in contact with infected farm animals, are particularly vulnerable. In addition to age, risk factors associated with hemolytic uremic syndrome included bloody diarrhea fever, and elevated white blood cell count and C-reactive protein levels [14]. The use of antibiotics or antimotility/antidiarrheal and antimicrobial agents in early stages of diarrhea has been shown to increase the risk of hemolytic uremic syndrome because the gut is exposed to a greater number of toxins for a longer period as intestinal motility slows [10, 15]. The pathogen is relatively tolerant to acid and can survive in fermented foods and fresh vegetable produce.
Waterborne transmission has also been reported, both from contaminated drinking-water and from recreational waters. Person-to-person contact is an important mode of transmission, particularly in institutional settings, such as day care centers, nursing homes and hospitals [16]. Reports of EHEC as a cause of diarrhea in other African countries are sporadic. In a study of childhood diarrhea in Nigeria, samples of EHEC were isolated from 5.1% of children with diarrhea [17]. A study from Japan in 1996 by Japan reported 9451 cases of EHEC infections, 1808 of which were hospitalized and 12 died; three-quarters of all these cases occurred during six major outbreaks. In the largest outbreak, in SakaiCity, 5727 people (0.5% of the city's population) were affected, and white radish sprouts served at school lunch were the most likely food vehicle. In outbreaks reported in other areas E. coli 0157 was also isolated from the salad and seafood sauce that were served at school lunches. In 1997, E. coli 0157 was detected in wild venison and white radish sprouts associated with sporadic infections [16]. In other parts of Asia, EHEC infections have been reported. In Malaysia, E. coli 0157:H7 has been isolated from the stools of patients with diarrheal illness [18]. In a study at a Bangkok hospital in Thailand, EHEC was identified in 7% of children with bloody diarrhea in whom other enteric pathogens were not identified [19]. In the Republic of Korea, EHEC was isolated from 1.3% of children with no bloody diarrhea. An investigation into the etiology of childhood diarrhea in China, EHEC isolated from 6.8% of children with diarrhea [20]. This article will review the diarrheagenic E. coli strains, which include several emerging pathogens of worldwide public health importance, and will specifically focus on pathogens afflicting humans. We will particularly concentrate on the E. coli strain whose study has advanced most over the last decade, E. coli (EHEC). The indications for culturing for EHEC differ from those for the rest of the diarrheagenic E. coli categories. Routine screening test for E. coli in urine are most often recovered on blood agar and eosin methyleneblue agar, which selectively grow members of the Enterobacteriaceae , biochemical test such as TSI,SIM, citrate, Urease were used, and also serologic test with VTEC-RPLA latex agglutination kit production of toxin and multiplex PCR methods used for identifying vtxB1- and vtxA2 Phenotypes.
MATERIAL AND METHODS
Clinical Specimens
Overall criteria for diagnosis UTI was growth of ≥104 CFU/mL of a urinary tract pathogen) in patients who did not have another potential source for their fever, a history of UTI, malodorous urine or hematuria, appeared "ill", abdominal or suprapubic tenderness on examination, or fever ≥39°C [25]. Patients who had received antibiotic treatment within the preceding 2 weeks, and those with known congenital anomalies of urinary tract or central nervous system-associated anomalies were excluded.
In this "prospective study" we collected 12572 urine samples from Children with UTI in Mofid children hospital Tehran, Iran /The age of patients ranged between 1-12 years old. Samples were collected in sterile plastic containers. The samples were inoculated in the Blood agar and EMB. After 24 hours of incubation at 37°C significant isolated microorganisms were identified by using Gram stain, catalase, oxidase, and other conventional tests. Each colonies which have β-hemolysis and Sorbitol fermentation negative on SMAC agar, suspected to EHEC so used Reverse passive Latex Agglutination Method for Detecting and Characterizing Verotoxins (Shiga Toxins) in Escherichia coli.
VTEC-RPLA-SEIKEN
A reverse passive latex agglutination was performed according to the Manufacturer’s instructions (No: 230553).
Collection of Samples and Preparation of Template DNA for PCR
A total of 378 E.coli were collected over the periods from 1 October 2008 to 31 June 2009. All bacteria plated directly on both blood agar and SMAC agar (Oxoid, Wesel, Germany) and incubated overnight at 37°C. All colonies suspended to
VTEC were diluted in 1 ml of Nutrient broth with 15%glycerol. The suspension was diluted 1:20 in PCR-grade H2O (Appli- Chem, Darmstadt, Germany); 1/10 was taken from this mixture for PCR with either a conventional instrument [21].
Extraction of DNA and Detection of STEC Strains by (M-PCR)
One mL of bacterial material was suspended in sterile water and heated at 100 °C during 10 min to release DNA. PCR was directly performed using consensus primers amplifying the toxin vtx genes Table 1. For each PCR-positive sample, a maximum of 20 colonies obtained on the MacConkey agar plate was tested separately in order to isolate VTEC strains.
Oligonucleotide Primers Sequences
| Amplification Product Size (bp) | Reference | Sequence (5΄-3΄) | Primer |
|---|---|---|---|
| 180 | Paton and Paton (1998) modified by Fitzmaurice J. (2003) | ATAAATCGCCATTCGTTGACTACAGAACGCCCACTGAGATCATC | VT1F VT1R |
| 255 | Paton and Paton (1998) modified by Fitzmaurice J (2003) | GGCACTGTCTGAAACTGCTCCTCGCCAGTTATCTGACATTCTG | VT2F VT2R |
Products seen on gel electrophoresis after multiplex PCR .lane M: ladder, lane 1, 2, 3: vtx-1 lane 6, 7, 8: vtx-2.
Positive vtx consensus PCR isolated colonies were subsequently identified through biochemical tests (indole production, Klieger test, ß-glucuronidase activity).The vtx1 and vtx2 genes were finally detected in sample isolates by the Multiplex-PCR procedure. Each of the primers was used at 20 pM, with 200 mM each deoxynucleoside triphosphates (Boehringer Mannheim, Meyher, France), 2.510X PCR buffer, 2.5 mM MgCl2, and 2 U of Taq DNA polymerase (Denmark). Then, 3 min in 95 °C, 35 cycles including, denaturation for 45s at 94 °C, primer annealing for 90s at 58 °C, and extension for 90s at 72 °C and, finally, 7 min incubation at 72 °C in a Corbett Research DNA thermal cycler (Corbett, Australia) were applied. The reaction products were then analyzed by electrophoresis on 1.2% agarose gels with 1% ethidium bromide (Fermentas, Germany). DNA from the reference strain, E. coli reference VTEC E. coli O157:H7 (ATCC; 43895), and a reagent blank, which contained all components except the template DNA(NTC), were included as positive and negative controls, respectively [22].
RESULTS
Fig. (1) shows the pattern and distribution of some pathogens caused UTI. E. coli with 47.2%isolate had the highest frequency followed with Klebsiella oxytoca with 147 (18.3%) isolate. Staphylococcus epidermidis (14.2%), Pseudomonas aeruginosa (8.2%), Proteus mirabilis (6.4%) and Staphylococcus aureus (4.3%), respectively. Approximately 20.9% strains of E. coli which isolated from urine were β-hemolytic E. coli, 87.67% were SMAC positive and 3% were β-hemolytic and SMAC positive E. coli were ETEC.In diagram, as you see, we found only 2.3 % of E. coli isolated from urine children with UTI was Enterohemorrhagic. (Figs. 2 and 3). The result electrophoresis Products seen on gel electrophoresis after multiplex PCR. lane M: ladder, lane 1, 2, 3: vtx-1 lane 6, 7, 8: vtx-2 (Fig. 4).
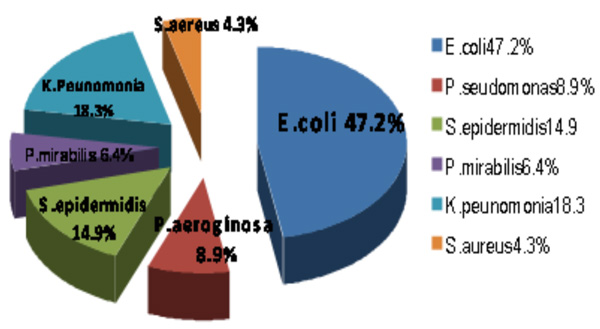 |
Fig. (1). The prevalence and pattern of bacteria in urine children with UTI. |
 |
Fig. (2). The prevalence of Enterohemorrhagic E.coli isolated from urine children with UTI. |
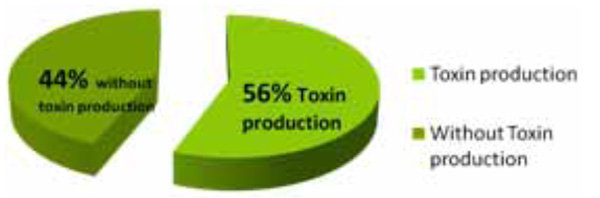 |
Fig. (3). The prevalence of Enterohemorrhagic E.coli isolated from urine children with UTI by VTEC _RPLA. |
 |
Fig. (4). The prevalence of verotoxin genes in Verotoxigenic E.coli isolated from urine children with UTI. |
DISCUSSION
This study confirms E.coli O157:H7 as one of the major causative agent of severe UTI in children. Studies have shown that VTEC strain is commonly isolated in hemolytic colitis and hemolytic uremic syndrome belonging to serogroup O157 [23,24,26]. Following this assertion, VTEC is vtx on the increase in UTI subjects with 16.7%, while chronic PID of 100% -1 suggest an absolute prevalence and also gynaecological condition given an inference of 100% vtx -1 & 2. Then if, vtx -1 and vtx -1&2 could give such an increasing prevalence, then many of these patients could as well be suffering from undiagnosed life threatening hemorrhagic colitis (HC) and hemolytic uremic syndrome [23]. Very scanty documentation of E.coli O157:H7 prevalence in UTI and other renal related diseases could not be obtained. Comparation of data was not possible from other part of the country due to poor documentation.
In our study only 2.3 % of E.coli that isolated from children's urine with UTI was Enterohemorrhagic. The prevalence of EHEC in Aslani and et al., studies (Institute Pasteur of Iran, Tehran 2008 in stool) and Nazek Al-Gallas (Tunisia,2007 in stool samples) was 44.5%, 11.1% respectively because EHEC is more dominant in stool than urine.
Since the detection of VT represents a rational, serotype independent strategy for the diagnosis of VTEC infections, simple and reliable procedures that allow the detection of VT.
For this purpose, we evaluated the performance of a VTEC-RPLA-SEIKEN assay(An reverse passive latex agglutination Vero Toxigenic E. coli, for the detection and characterization of VTx s in E. coli culture filtrates. In this study, 44% of E.coli was non-verotoxigenic and 56% was verotoxigene. Mohamed Karmali and et al., in 1998 Evaluated a Microplate Latex Agglutination Method (Verotox-F Assay) for Detecting and Characterizing of Verotoxins (Shiga Toxins) in Escherichia coli. They examined 68 VT-positive E. coli strains and 104 VT-negative strains E. coli and conclude that the Verotox-F assay is highly sensitive and specific for the detection and characterization of VTs in culture filtrates of human E. coli isolates. The test is rapid, reliable, and easy to perform; its results are easy to interpret; and it should allow testing for VT to become more widely performed. We also used a different method of latex agglutination but the same conclusion [27].
In 1983–2002 in Atlanta, Georgia Brooks and et al., considered Non-O157 Shiga Toxin–Producing Escherichia coli infections. They found 61% stx1, 22% stx2 and 17% both stx1 and stx2 while, we had 17/3 % vtx1, 52/45% vtx2 and 30/3 both vtx1 and vtx2.This significant different percent is that in our study was on O157 Shiga Toxin–Producing Escherichia coli urinary tract Infections [28]. Thus we suggest rapid method for identification of this bacteria and proper treatment to inhibition of unwanted complications.
CONFLICT OF INTEREST
None declared.
ACKNOWLEDGEMENTS
The authors would like to thank Professor Fatemeh Fallah for helpful comments and discussions. They would also like to special thanks to pediatric infection research center personnel in Mofid Children Hospital.







