All published articles of this journal are available on ScienceDirect.
Isolation and Culturing Axenic Microalgae: Mini–Review
Abstract
Microalgae have several applications in nutraceuticals, pharmaceuticals, cosmetics, biofuel production, and bioremediation, among other fields. Isolation and purification are extremely important for obtaining axenic cultures of microalgae from different environments and crucial for their biotechnological applications, but it is not an easy task. In view of the above, it is fundamental to know the classical and advanced techniques and examples of how scientists from around the globe have applied such methods to isolate several genera and the impact of each step on successful algal purification. This review provides a brief and simple explanation of the methodology for sampling, growth, obtention of unialgal, and posterior axenic culture, which will facilitate the development of novel microalgae-related discoveries and applications for new researchers.
1. INTRODUCTION
Microalgae are constituted by a vast cluster of eukaryotic and prokaryotic photoautotrophic microorganisms, which are typically from diverse phylogenetic positions, with extremely different morphology, physiology, and biochemistry. However, microalgae generate around 50% of the oxygen and contribute to nearly 30% of CO2 fixation in the atmosphere. They are primary producers of the ocean and play the most relevant role in the marine food web. In addition, they have applications in nutraceuticals, pharmaceuticals, cosmetics, biofuel production, and bioremediation. Due to the above, isolation and purification of microalgae are crucial steps for their biotechnological applications. The objective of algae isolation and purification is to obtain axenic cultures, which means a viable culture of a single species without contaminants. Under sterile conditions, these cultures will remain pure. Many methods of basic algae culturing were developed around the 1900s, so novel isolation and identification protocols are essential for identifying specific characteristics like the lipid content or bioactive compounds. Some algae species are easily cultivated, whereas others are nearly impossible to grow. Therefore, for successful isolation, the natural environmental conditions should be understood to replicate them in lab conditions. Elimination of contaminants like bacteria, fungi, or other algae is fundamental to obtain axenic microalgae cultures [1-4].
The aim of working with microalgal axenic cultures in research is to ensure that the results obtained from a study came directly from the microalgae species studied and not from a contaminant microorganism. If the goal of the microalgal culture is to produce and commercialize biomass, an extract, or a compound from a specific species, axenic culturing is critical for quality control. Any contaminant can have an important economic impact, result in the loss of a batch, or severely modify the composition of the final product.
2. PRE-ISOLATION TECHNIQUES
These techniques are applied prior to isolation. Collection, proliferation, separation in smaller groups, and remoting of coarse contaminants will only facilitate the posterior isolation process and obtention of axenic cultures.
2.1. Collection
The collection method is crucial for a successful establishment of an axenic culture. Damage of algal cells during collection leads to failure. When sampling an unknown environment or target organism, multiple collection methods should be addressed. Sampling techniques may include syringe sampling, scraping, brushing, and the inverted Petri dish method. However, the most reported sampling method is simply water collection from a water body. If the target species is usually found in-depth, a Niskin bottle or rosette sampler can be used. When sampling, it is fundamental to record factors such as light, water temperature, dissolved O2 and CO2, nutrient concentration, pH, and salinity to mimic these conditions at the laboratory and establish the location coordinates to replicate sampling if needed [2, 3, 5].
The sampling conditions will not play the main role in their posterior axenicity as long as the aseptic technique is applied. However, if the genera or species to be isolated is targeted or previously known, the conditions of the collection should be adjusted to favour it, so the axenic culturing can be achieved more easily.
2.2. Enrichment of Samples
Once a sample is taken to the lab, adding nutrients to the natural sample will potentiate microalgal growth. Organic substances should be added in small amounts because they can cause undesirable bacterial proliferation. The enriched culture is incubated and examined every few days for the growth of the target species, and then individual cells are isolated. Sometimes the target species grow successfully after isolation but die after several transfers, indicating that the culture medium lacks an element or compound or that the organism accumulates wastes that poison their environment [2]. Enrichment of samples should be targeted for different kinds of microalgae; for example, delicate algae and flagellates could be favored by enriching samples with ES enriched seawater; chrysophytes, chlorophytes, diatoms, and cryptophytes grow well under the addition of F/2 media to water samples. On the other hand, cyanobacteria growth is facilitated by adding BG11SW media [6].
This nutrient addition process will facilitate the growth of the targeted genera or species, easing the posterior purification techniques by favoring their growth. This will allow the number of microalgal cells to overthrow the number of fungi or bacterial cells, or even the cells of other microalgal species that are not of interest to the researcher.
2.3. Remotion Methods
Microalgae usually secrete a heterogeneous matrix called extracellular polymeric substances (EPS), which mainly consists of polysaccharides, proteins, nucleic acids, and lipids. This way, algal cells interact with each other, mediate their adhesion to surfaces, and generate cell protection and energy storage reservoirs. Bacteria frequently attach and feed on this EPS layer. Vortexing, sonication, and surfactant treatment prior to isolation methods assist in detaching bacteria and increasing the separation efficiency [7, 8].
Detaching the bacteria from the EPS will increase the effectiveness of techniques such as filtration, centrifugation, and antimicrobial treatment, by allowing the proper separation of cells by their difference in size and density and making the bacteria more susceptible to the chemical agents added.
2.4. Filtration
Essentially, samples are separated into two portions mainly based on particle size differences [7]. Recently, Haoujar (2020) applied this method by filtering samples from Moroccan Mediterranean seawater through a series of membranes decreasing mesh (33, 20, and 0.45 μm) then placing membranes on agar plates, thus achieving the isolation of 32 microalgae (Mostly coccoid) [9]. This method allows the separation of microorganisms by their cell size only, but by itself, it is usually not enough to obtain axenic or unialgal cultures since different organisms can have similar cell sizes. However, this technique can be applied in such a way that the microalgae cells, which are usually bigger than bacteria, can be retained in a membrane or filter to be posteriorly cultivated in a media in which most parts of the bacterial cells were removed.
2.5. Centrifugation
Centrifugation works by separating organisms by cell size using gravity. Low-density cells, such as bacteria, tend to stay in the supernatant fraction and are discarded, while microalgae and higher-density cells normally remain in the pellet fraction. After several centrifuging cycles, a higher concentration of a desired microalgal strain will be achieved. Using a silica or percol density gradient is useful for separating the different cells into different bands. The separation efficiency of centrifugation depends on speed and time; therefore, these parameters should be optimized for a given species. Also, excessive centrifugal force can sometimes damage fragile cells [5, 7, 10]. Lee (2021) applied density gradient centrifugation to dinoflagellate species Karenia mikimotoi and Alexandrium tamarense using a 90%, 90–50%, 90–50–30%, and 90–50–30–10% Percoll solution and harvesting the algal cells, reducing the bacterial population from 5.79 ± 0.22 log10 CFU/mL to 1.13 ± 0.07 log10 CFU/mL [11]. As seen in several studies, centrifugation is helpful in concentrating algal cells and reducing the bacterial population. However, this technique does not generate an axenic algal culture by itself, but it will facilitate posterior techniques that will ensure the purity of cultures.
3. ISOLATION TECHNIQUES AND UNIALGAL CULTURES
In most cases, these techniques will allow the establishment of a unialgal culture, free from other microalgae species, but that may or may not be contaminated with bacteria. Therefore, these methods do not usually ensure the axenicity of the microalgal cultures.
3.1. Single-Cell Isolation
This method can be performed with a micropipette, a pasteur pipette, or a glass capillary. The objective is to collect a single cell from the sample, deposit it in a sterile droplet of culture medium, pick up the cell again, and transfer it to a second sterile droplet. This process is repeated until a single algal cell can be placed into the final isolation vessel [2]. Novel techniques for single-cell isolation include micromanipulation by employing stereomicroscope, microcapillary tubes, and optical tweezers, which greatly reduces labor and cell damage. However, they require expensive equipment and skilled lab personnel. These techniques perform cell separation with a high level of accuracy, making it an ideal tool for screening and isolation. Microcapillaries are usually made of glass with an outer diameter of 1 mm. On the other hand, optical tweezers (OT) is a non-invasive method for physical manipulation that uses a highly focused laser beam to trap and manipulate microscopic objects using only the properties of light, applying usually infrared (IR) region wavelengths between 700 and 1300 nm that generates two kinds of forces, the scattering force produced by the photons striking the cell along their propagation direction and the gradient force produced by a gradient of field intensity. These forces act as optical tweezers and depend on the wavelength of the laser beam and the particle size. This allows for the micromanipulation of a single object that ranges in size from nanometres to micrometres, including living cells. Such optical forces can stop and drag biological swimmers, such as microalgae. Application of OT for single-cell manipulation may be based on fluorescence activation or cells’ shape, size, and refractive index difference, providing an opportunity to sort cells to a compartment without any pre-treatment. However, very few studies have reported on this specific technique [5, 12-16]. Pilat et al. concluded that non-invasive optical manipulation of living cells of photosynthetic microorganisms could be performed using laser wavelengths longer than 935 nm. They also observed that a 1064 nm wavelength did not cause any damage to the microalgae, and it is sufficient for reliable 3D manipulation of cells. Lim (2012) applied the serial dilution and posterior micromanipulator technique to obtain pure cultures of Nannochloropsis sp., Dunaniella salina, Chlorella sp., and Tetraselmis sp [17]. Ota (2019) designed a glass microfluidic device to perform isolation, cultivation, and time-course observation of Euglena gracilis by using semi-closed microchannels. Then the cells were identified by Raman microscopy, a vibrational spectroscopic technique that relies on the inelastic scattering of monochromatic light produced by a laser that interacts with the sample, which causes a shift in the energy of the photon to a higher or lower frequency that can be detected by the equipment. Biomolecules in microalgae have their own specific functional groups and show characteristic peak frequencies that appear in Raman spectra, which allows researchers to identify specific biomolecules for specific microalgae species. In this case, they manage to identify the formation of paramylon, a unique metabolite of E. gracilis [16, 18].
3.2. Isolation in Agar
In order to achieve successful isolation using this technique, the algae must be able to grow on solid media. Coccoid cells and most diatoms grow well on agar, some flagellates do not grow on agar, and dinoflagellates rarely grow on agar. After agar culturing, a micropipette tip is driven into the agar to pick up the selected cells and then discharged into a liquid culture medium. This technique can also be used to maintain axenic cultures by perpetual transfer with repeated use of the agar pour technique [2]. It is common to use more than one method for microalgae isolation. Jahn (2014) isolated 24 unialgal cultures by filtering samples through a 100 μm filter, enriching the samples with f/2-medium and streaking the enriched microalgal communities on f/2-medium solidified plates with agar, then picking colonies from the plates and transferring them to a liquid f/2-medium in 12 well plates [3]. It is also common for researchers to use several medium cultures to obtain different genera of microalgae. Thao (2017) isolated oleaginous microalgae. After sampling, they used BG11 for fresh, f/2B for brackish, and f/2 for marine water samples. Then, for all three media, a solid or semi-solid medium was prepared by adding 10 g/L or 5 g/L agarose, respectively. Two methods of isolation were applied: 1) Streaking on solid agarose plates; 2) Adding 0.1 mL of enriched cultures to 5 mL of semi-solid medium, vortexed and poured over the solid medium to form a semi-solid overlay, incubated, and repeated until pure isolates were obtained [19]. Certain features of microalgae, such as phototaxis, can be combined with agar cultivation to maximize results. Banerjee (2012) isolated Chlamydomonas sp. from Chlorella sp. mixtures by passing light (6,000 lux for 7 h) through agar, given that Chlamydomonas has flagella and the rhodopsin pigment is attracted towards the light [20]. Isolation in agar by itself may lead to an axenic microalgae culture, as long as the targeted microalgae to be isolated does not grow on consortia with other microorganisms and does not have bacterial cells attached to the EPS. Posterior tests and microscopic observation may be required to ensure the axenicity of the culture. If, after isolation in agar, the targeted microalgae remain contaminated, further techniques should be applied. However, this technique allows the separation of targeted microalgae from most of the microorganisms from a sample and drastically reduces the contaminants.
3.3. Dilution Techniques
This is done by transferring only one cell into a vessel by diluting a sample or culture. This technique is usually performed by preparing and filling test tubes with 9 mL of sterile saline solution or adequate culture media. Then, 1 mL of microalgae culture or an enriched sample is transferred into the first test tube and mixed, obtaining a 1:10 dilution. Then, 1 mL from the first dilution is transferred into the second tube and repeated five to six times. It is important to consider inoculating many tubes from the last dilution because some single cells will die and some inocula will contain two or more species or no cells at all. Enrichment substrates can be added to isolation tubes to specifically grow a target species or incubate under certain specific conditions. Axenic isolates are not often obtained with the dilution technique because bacteria are commonly more abundant than algae. The dilution technique is often used when attempting to culture random algal species from field samples with the goal of discovering and studying new species [2, 21]. Dilution techniques will allow obtaining cultures which are mostly composed of some microalgal species but do not ensure the axenicity of the culture. Contamination tests and microscopy observation is needed to detect other microorganisms present in the culture. Further techniques will likely have to be applied in order to generate the axenic microalgal culture.
4. PURIFICATION METHODS FOR MICROALGAE AND OBTENTION OF AXENIC CULTURES
Once a target species has been grown and isolated from other microalgae species, it is time to generate an axenic culture by eliminating any traces of bacteria or fungi that are commonly associated with microalgal culture contamination. The use of the sterile technique and pre sterilization of any material used during any of the steps of isolation, purification, sub-culture, or escalation of microalgae, minimizes the risk of contamination and increases the success of axenic microalgae culture achievement. A laminar flow hood is recommended, but the sterile technique can still be performed in a laboratory room without one if precautions are taken. The sterility of materials should be checked, and if there is any doubt, the material should be discarded. During purification, the main issue is to avoid killing the target species since algae are vulnerable to adverse environments. For microalgae, the addition of purification antibiotics or other selective agents is common [2].
4.1. Antibiotic Treatment
The main objective of antibiotic treatment is to reduce bacterial contamination of an algal culture to a number that will allow a small inoculum to transfer enough algal cells without viable bacteria. The efficacy and toxicity of the antibiotic treatment depend on the concentration and period of exposure and vary depending on the microalgae and bacteria species. Viable bacteria tend to decline drastically after 48 h of exposure to the microalgal culture to the antibiotic treatment. Then a small portion can be transferred to an adequate medium without antibiotics. Antibiotics will work in one of two ways: bactericidal (e.g., penicillin, vancomycin) will kill bacteria by interfering with the formation of the bacterial cell wall, or bacteriostatic (e.g., chloramphenicol, tetracycline), which interferes with the bacterial metabolism but does not necessarily kill them. Theoretically, antibiotics will destroy all the contaminants without harming algal cells, but this is rarely achieved in practice. A well-studied and basic antibiotic treatment is 100/25/25 mg/L of penicillin, streptomycin, and gentamycin which is usually tolerated by most algae. Purification may also be achieved by the sequential transfer of the algal culture through a series of different antibiotics. This method has lower toxicity to the algae. Also, one antibiotic mix may be fatal to some bacteria but only suppress the growth of others, while using sequential antibiotics may kill the surviving bacteria [2, 7, 22].
Many combinations and techniques for purification with antibiotics can be applied, from simple antibiotic addition like Bonett (2020), who managed to obtain axenic cultures from Chlorella sp., C. sorokiniana and Desmodesmus sp using only a BG11 medium added with ampicillin in concentrations of 50, 70, 80, 90 – 100 mg/ L [23], to using combinations such as the one reported by Wu (2013) in which he obtained axenic cultures of Desmodesmus sp. by continual sub-culturing in BG11 agar plates supplemented with ampicillin and kanamycin [24]. More complex antibiotic combinations can be used when bacterial contamination persists. Han (2016) obtained purified strains of Nannochloropsis sp., Cylindrotheca sp., Tetraselmis sp., and Amphikrikos sp after treating them with a mix of ampicillin, gentamycin, kanamycin, neomycin, and streptomycin (600 mg/L each) for 3 days and then transferring them to an antibiotic-free medium for 5 days to posterior spread on solid f/2 media to isolate colonies. They also reported that microalgae treated with antibiotics at high concentrations resume their growth fast when transferred to an antibiotics-free medium [25]. Lee (2021) generated axenic cultures for both armored and unarmored dinoflagellate species Karenia mikimotoi and Alexandrium tamarense. After gradient centrifugation they used an antibiotic treatment by spreading 0.1 mL xenic cultures on agar containing: penicillin (100 U), streptomycin (100 μg/mL), gentamicin (100 μg/mL) and tetracycline (1 μg/mL). After 14 days of incubation, no signs of bacterial colonies were obtained [11].
Regarding antibiotic toxicity to microalgae, Youn and Hur found that chloramphenicol and penicillin G showed lethal effects on microalgae, being higher for chloramphenicol and that the optimum quantity for antibiotic treatments without microalgal toxicity was 2,000 ppm of dihydrostreptomycin for Chlorella ellipsoidea, neomycin 500 ppm for Isochrysis galbana and Heterosigma ahashiwo, chloramphenicol 500 ppm for Cyclotella didymus, and dihydrostreptomycin sulphate and neomycin 6,000 ppm for Thalassiosira allenii [20]. Bashir and Cho (2016) also studied the effect of antibiotics on microalgae, comparing the growth and protein synthesis of chlorophytes Dictyosphaerium pulchellum and Micractinium pusillum in the presence of kanamycin and tetracycline and reported a significant effect of both antibiotics in the measured parameters [26].
Antibiotics are usually applied once the sample has been enriched and previous isolation techniques have been applied to the culture, so a unialgal culture has been obtained. This treatment will allow the elimination of bacteria and/or fungi present in the culture or reduce it in a way that will ease the obtention of the axenic culture. Antibiotic treatment is one of the most used techniques by researchers for obtaining axenic microalgal cultures. It is usually economical, and most laboratories have the facilities and equipment to carry out the methodology.
4.2. Enzymatic Treatment
Obtaining axenic cyanobacteria culture using an antibiotic treatment usually has certain limitations and sometimes requires an alternative. Microalgae tend to be more resistant to lysozyme digestion than bacteria. Another option for generating an axenic culture is using a lysozyme base. Axenic cyanobacterial cultures have previously been obtained by treating cultures with the minimum lethal concentration for up to 90 min [7]. Also, combinations of enzymatic treatment and antibiotics can lead to promising results. Tale (2014) used a combination of lysozyme (20 μg/mL) and antibiotic mixture (cefotaxime-500 μg/mL and tetracycline 50 μg/mL) to obtain axenic cultures of the genera Chlorella and Monoraphidium [27].
4.3. Antimicrobials
Sodium hypochlorite, sulfonamides, potassium tellurite, detergents, pesticides, salts, aldehydes, and peroxides have also been used to obtain axenic cultures. However, these compounds tend to have higher toxicity on microalgae. Recently the triiodide ion (-3 I) has been applied with promising results. This is adsorbed onto a resin surface and then filled into a column. The contaminated unialgal cell suspension is usually run through this column several times for axenization. This technique was applied to a Nannochloropsis gaditana culture contaminated with bacteria. Observations lead to the conclusion that the treatment remarkably reduced the number of viable bacteria and had few effects on the microalgae. There are a few reports on the use of natural agents in algae cultures for contamination control, such as cinnamaldehyde, geraniol, carvacrol, and cinnamic acid, that were evaluated to eliminate fungi. Natural chemical agents are generally less polluting, more easily degradable, and may comply with disposal regulations. Also, the resistance of biological contaminants to currently used chemical agents enhances the importance of finding new alternatives. Still, these natural substances need research and regulation in order to be used [7, 26-28].
For the optimal chemical agent control of contamination and purification of microalgae cultures, the screening of antibiotics, chemicals, or enzymes which inhibit biological pollutants without damaging the target microalgae is the preferred route [29, 30].
5. AUTOMATED ISOLATION TECHNIQUES FOR MICROALGAE
Flow cytometry is the most common automated technique being exploited and used for microalgae cell sorting, with the advantage of autofluorescence of their natural intracellular pigments, allowing to distinguish different species or distinguish microalgae from other microorganisms. Based on the properties of Chlorophyll Autofluorescence (CAF) and Green Autofluorescence (GAF), algae species such as diatoms, dinoflagellates, or prokaryotic phytoplankton can be distinguished. Fluorescence-Activated Cell Sorting (FACS) cytometry is gaining relevance and is based on light scatter and fluorescence, resulting from the passing of a fluid stream through a laser beam, in which cells scatter or absorb the laser beam and emit fluorescence, which provides information such as cell size, integrity, and photosynthetic characteristics. Due to the high amounts of chlorophyll, microalgae cells are naturally fluorescence-active. After excitation of chlorophyll via a laser, an intense red fluorescence signal is emitted and can be used as a trigger for automated cell sorting, which allows microalgae isolation and the removal of bacteria. The efficiency of sorting is based on the abundance, cell size, shape, and hardiness of the algae being sorted and can be increased by enrichment and sonication of the culture prior to sorting [1, 2, 5, 31, 32]. Examples of FACS application include Bui (2013), who, after obtaining unialgal cultures by classical methods, used FACS to obtain their axenic cultures, for which DNA analysis showed no bacterial ribosomal DNA sequences present [33]. Fistarol also reported using FACS and managed to isolate microalgae from the genera Scenedesmus, Actinotaenium, Cosmarium, Haplotaenium, Staurastrum, Aulacoseira, Navicula, Thalassiosira, and Protoperidinium. However, in their work, they also mention that not all species of microalgae are suitable for sorting by FACS, either because they are too large to fit the capillary lines of the instruments or too sensitive to endure the pressures applied [4]. The use of FACS in combination with fluorescent dyes such as Bodipy or Nile red generates a great method for screening of organisms production of metabolites of interest. This combination was applied by Pereira (2013), who isolated strains from the genera Picochlorum, Nannochloris, and Desmochloris with lipid contents ranging from 20% to 25% of the biomass dry weight [34]
6. MAINTENANCE OF MICROALGAE AXENIC CULTURES
Once the axenic culture has been achieved, the next goal is usually to maintain it in the long term. Microalgae are highly sensitive to changes in environmental conditions, which affects their morphological and physiological characteristics. Microalgae are generally maintained by periodic transfer, freeze-drying, and cryopreservation. The method of choice depends largely on the capability of the laboratory, type of culture, cost of materials, and labor. There is no universal method for the preservation of all species of microalgae. Different strains exhibit different responses to stress. Some strains are successfully freeze-dried and cryopreserved, while others are not. It is recommended to have at least two methods of preservation to minimize losses.
6.1. Periodical Sub-Culture
Microalgae are traditionally maintained by periodical sub-culture due to the feasibility of the procedure. Conventional sub-culturing is made by moving an inoculum from a late log/stationary phase into a pre-sterilized, fresh culture medium using the aseptic technique. The transfer frequency is determined by the growth characteristics of the strain. Continuous maintenance over long periods of time is labor-intensive. However, it is often the only procedure available in some laboratories or the only way for the preservation of microalgae recalcitrant to cryopreservation or freeze-drying. A principal limitation of perpetual transferis the media and incubation regimens, that in contrast to the natural conditions may cause morphological and physiological changes in some species. Other concerns include the possibility of contamination or handling mistakes [35].
6.2. Freeze Drying
Freeze-drying requires the elimination of water from a frozen algal suspension by sublimation under reduced pressure for which specialized equipment is needed. Freeze-dried microalgae maintain their original shape and texture, and it has been found to be successful in preserving mostly cyanobacteria. This procedure minimizes the hazard of genetic drift, contamination, and accidental mislabeling. Also, the cultures require smaller storage space and reduced labor. It is also the best way of shipping cultures. However, freeze-drying has not been found to be a successful maintenance method for many microalgae, which exhibit very low levels of viability after the freeze-drying process [35, 36].
6.3. Cryopreservation
Cryopreservation may be defined as the storage of an organism at an ultralow temperature, so it remains capable of surviving the defrosting process. Many microalgae can be cryopreserved but typically with lower post-thaw viability levels. Cryopreservation is still largely an empirical science process. Despite this, hundreds of species of cyanobacteria and eukaryotic microalgae have been successfully cryopreserved by commonly applying methanol (MeOH), dimethylsulfoxide (DMSO), or glycerol as cryoprotective additives (CPAs). This type of methodology reduces the routine costs for the culture collection, ensuring the stability and purity of samples and reducing the risks of contamination during the handling of samples. The specific procedure and CPA used for a particular species or even a strain may vary. Some trial and error may be required for new isolates. Cryopreservation often includes placing 1.0 ml of the culture (preferably actively growing under suitable conditions) at the desired temperature of choice, either a -20°C, -80 °C, or -170 °C (vapor phase of liquid nitrogen) with the chosen CPA. The usage of a two-step protocol may be necessary for some strains. This requires the addition of the CPA to the culture and cooling it to a specified subzero temperature to facilitate dehydration, then cooling rapidly to the final storage temperature. For the revival of algal cultures, the cryogenic vials are usually placed in a 35 °C water bath for 5 minutes and then transferred into 50 ml of an appropriate growing medium and incubate at appropriate conditions. Generally, strains that display specific cell characteristics such as unicellular, small, spherical, with no spines, and without vacuoles are usually highly viable after thawing. This cryopreservation methodology is currently applied by The Culture Collection of Freshwater Microalgae based at Universidade Federal de São Carlos, which currently has the largest microalgae collection in Brazil, holding around 700 strains of freshwater microalgae. Some disadvantages are that cryopreservation requires specialized equipment, special consumables, and training. Errors resulting from improper thawing under uncontrolled conditions, even briefly, may lead to the death of culture [35-39].
After cryopreservation, viability should be determined by detecting the metabolic activity and/or reproduction capacity of the cells. Vital staining employing fluorescene diacetate or re-growth of cells are the most practicable approaches. Usually, a minimum acceptable viability level is around 30%. In addition, there should be no significant differences in genotype or phenotype between control and post-thaw cultures. These conditions are currently problematic to achieve and may not be technically feasible for some taxa and for non-axenic material [40].
7. CONTAMINATION CONTROL AND SCALING OF MICROALGAE AXENIC CULTURES
Despite using the sterile technique, contamination may occur. Therefore, the evaluation of contamination in any step of algal culture is essential for quality control. When a culture is presumably contaminated, several methods can help detect it. The simplest technique is microscopy, which allows to directly perceive the growth of contaminant microorganisms. This can be combined with the use of dyes that highlight undesired microorganisms. One of the disadvantages of microscopy is that it requires an experienced operator in order to obtain good results. Sterility tests can be performed by inoculating with the microalgae culture a 0.1% peptone agar or broth and incubating at a suitable temperature according to the suspected contaminant. This is usually satisfactory to observe bacterial or fungal growth. Malt extract is an alternative if fungi contamination is suspected. To test for air-borne contamination, a series of the test medium is exposed over specific periods, incubated, and observed. Variations of these simple tests can be used to test culture medium, vitamins, stock solutions, pipettes, etc. Flow cytometry may also be used to detect contamination in cultures. However, as mentioned above, it is not cheap. Molecular techniques based on the polymerase chain reaction (PCR) have the advantage of detecting contamination even in low concentrations, and there are other new molecular alternatives to identify biological contaminants such as metagenomics, metatranscriptomics, and metaproteomics. Nonetheless, they are far more expensive than microscopy or sterility tests. These methods, with nearly endless modifications, can be performed to help evaluate sterile conditions. The tests must be carried out under conditions that show positive results when contamination exists and should not be considered as an absolute measure of sterility but as indicators of contamination. Different protocols and modifications are often required according to available materials and the goal of microalgae culture [2, 29, 30].
As mentioned above, microalgae have applications in nutraceuticals, pharmaceuticals, cosmetics, biofuel production, etc. However, a bulk volume of microalgal biomass is needed to achieve its commercialization. Most methods for production are based on open ponds (which increase the chances of contamination from the environment) and closed photobioreactors. Open pond production has been successful only for a limited number of microalgae by applying very high salinity or high pH. Mass production of microalgae depends on large volumes of water. Therefore, it is unrealistic to adopt heat sterilization. Although water can be processed with bleach or filtered before mass cultivations, some pollutants usually remain. Closed photobioreactors reduce exposure of culture to the environment, giving better protection against pollutants. However, aeration is needed. Filtration with a microporous membrane is generally adopted for air sterilization, but it is impractical to apply it to large volumes of air during mass cultivation. Complete sterility is very difficult to achieve, even in closed photobioreactors. Therefore, it is essential to develop techniques for the timely identification of contaminants and achieve significant control [29, 30].
Temperature is a variable that could help control contamination. Incubating outside the temperature range of contaminant microorganisms may help reduce the risk of their growth, keeping in mind the optimal temperature for microalgal growth. Another factor that can be manipulated for the benefit of culture control is using pH values that may suppress contaminant growth but maintain the viable microalgae. Similarly, salinity can inhibit the development of contamination. Chemical agents can be applied to control contaminants, but this requires knowing the undesired microorganism, the minimum inhibitory concentration of the agent to use, and the tolerance of the cultivated microalgae. Commonly used chemical agents are antibiotics, fungicides, pesticides, salts, aldehydes, peroxides, and ammonia or combinations. However, this alternative for contamination control is generally applied in the initial stages of cultivation (axenization processes). Another alternative for contamination control is the use of pulsed electric fields to selectively damage cells in the culture. One of the problems is that there is not enough information for biological control. Mechanisms known so far for contamination control have deficiencies and are not always effective as contamination may persist and lead to lower productivity, poor microbiological quality, or deviance from the intended composition. An alternative is the use of controlled closed and smaller systems. Although they have a higher economic impact on production, they allow for better control of any source of contamination and are recommended for feedstuffs or nutraceutical applications of biomass. Strain selection with resistance to biological pollutants is also an important factor to consider when mass production is intended. A greater understanding of how these biological pollutants interact with host microalgae and reasonable cultivation technology may lead to the development of effective control methods [29, 30].
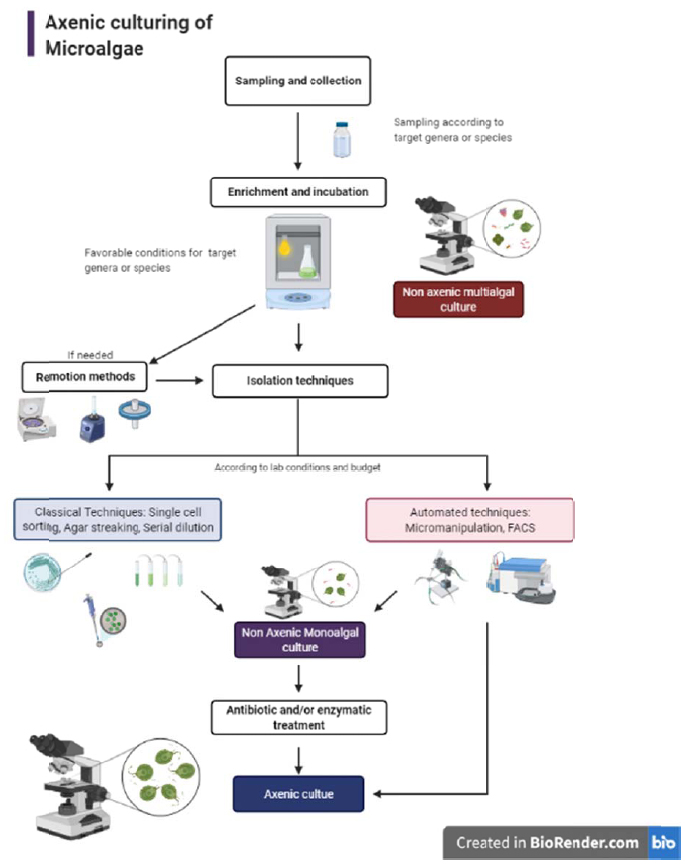
CONCLUSION
Based on the methodology reviewed, the best approach for microalgal isolation, obtention of axenic cultures, and maintenance depends on various factors. First, researchers must consider if there is a target species, in which case the methodology should be emphasized on sampling the correct environment, provide species requirements, and be aware of their weaknesses by using the appropriate media and avoiding purification and maintenance techniques that could be harmful. If the objective of isolation is to discover or study several species in a certain geographic location or sample, the approach will be completely different. Several sampling methods, media culture, and growth conditions should be applied. Once several genera have been identified, the criteria for isolating each must be applied. Another issue to be taken into account in microalgae isolation is the accessibility, resources, and lab facilities of the research institution. The researcher will evaluate if their lab has access to techniques like FACS, micromanipulation, freeze-drying, or cryopreservation. Otherwise, classical techniques should be applied. However, if the researcher is limited to resources for applying classical techniques, successful isolation, axenic culture establishment, and maintenance can be achieved without a major obstacle beyond the time consumed. Once the available techniques have been determined, a serial combination of them is recommended in order to achieve better results in the axenic microalgae culture, as shown in Fig. (1). If the main goal is to commercialize biomass or produce it at a large scale, culture contamination control should be taken into account. However, this is not an easy task, as strain selection, mass culture conditions, and periodical tests for contaminant microorganisms should be addressed previously to mass cultivation.
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


