All published articles of this journal are available on ScienceDirect.
Prevalence of Human Papillomavirus Associated with Head and Neck Squamous Cell Carcinoma in Jordanian Patients
Abstract
Background:
In addition to smoking and alcohol consumption, human papillomavirus (HPV) is a leading etiology for Head and Neck Squamous Cell Carcinoma (HNSCC). However, this causal association is still understudied in Middle Eastern populations.
Objective:
The aim of this study was to determine the prevalence of HPV-associated infection in the Jordanian HNSCC patients and the associated HPV genotypes.
Methods:
Formalin-Fixed Paraffin-Embedded (FFPE) squamous cell carcinoma samples of the head and neck were collected from two referral centers in Amman, Jordan to determine the existence of HPV DNA. After DNA extraction HPV infection and genotyping were identified using real-time PCR.
Results:
HPV DNA was detected in 19 out of 61 (31.1%) HNSCC samples. Despite screening for 28 different genotypes, HPV 16 was the only genotype identified in all examined samples. Most HPV-positive samples were obtained from the oropharynx (41.7%), oral cavity (37%), and larynx (18.2%). No significant association between HPV 16 genotype and age, sex, tobacco use, anatomical location, or tumor grade was noticed.
Conclusion:
This study reported a high association between HPV 16 genotype and HNSCC in Jordanian patients. These data should facilitate the implementation of appropriate HPV awareness campaigns, and activate selective prophylactic measures against HPV infection.
1. INTRODUCTION
Head and neck cancers are a group of malignancies originating from the epithelium of the upper respiratory-digestive tracts [1]. Histologically, most of these cancers are of epithelial origin, with more than 90% are squamous cell carcinoma [1]. HNSCCs are attributed in most cases to environmental exposure to tobacco, alcohol, poor oral hygiene, and chemical irritation [2]. However, 10%-20% of HNSCCs occur irrespective of the previous factors. Recently, Human Papillomavirus (HPV) infection of the oropharynx has been implicated as a major contributing etiology for HNSCC [3, 4]. Most of the high-risk HPV infections in HNSCC patients are directly related to infection with HPV 16 and to a lesser extent with other HPV genotypes [5].
The global incidence of head and neck cancers is estimated to be around 650,000 new cases annually, and it is ranked as the 9th most common cancer worldwide [6]. The number of HNSCC that are associated with HPV infection varies globally, but a recent meta-analysis study has shown it to be around 32% [7]. These variations may be attributed to the geographical location of habitat, or variation in the sensitivity and specificity of the detection methods for HPV infection. The incidence of HNSCC in Jordan is stable at about 100 new cases each year, representing almost 2% of the total number of new cancer cases locally [8].
HPV-positive HNSCCs were shown by multiple studies to possess better survival outcomes compared to HPV-negative patients [9, 10]. Previous studies showed that the three-year survival rate is about 80% for HPV positive tumors but has declined to ~60% for HPV-negative tumors [11]. In Jordan, only a single study on the incidence of HPV-related HNSCC was reported, which was conducted on samples taken from a single medical laboratory [12]. In our study, samples were obtained from two central referral hospitals in Amman (King Hussein Cancer Center and King Hussein Medical Center) which represent patients from the different Jordanian governorates and thus, would capture the majority of HNSCC patients in the country. The aim of this retrospective study was to determine the prevalence of HPV-related HNSCC and their different genotypes in Jordan and reflect the results by proposing preventive control measures.
2. MATERIALS AND METHODS
2.1. Clinical Samples
A retrospective screening of the medical records of HNSCC patients in King Hussein Cancer Center and King Hussein Medical Center, diagnosed between 2013 and 2018, was performed. The patient eligibility criteria for inclusion were: patient with a histologically confirmed diagnosis of squamous cell carcinoma from an HPV-driven anatomical site, and being a first-time HNSCC patient who has not received radio- or chemotherapy before tumor excision. After exclusion of the inappropriate samples for our study, only 65 patients with oral cavity and oropharyngeal tumors were fit for inclusion. After the histopathological study of samples, only 61 were finally included in this study. Fig. (1) represents the subject disposition flow chart which illustrates the inclusion and exclusion criteria for HNSCC patients in this study. Patients with HNSCC subtypes with no established etiological connection to HPV (namely, nasopharyngeal, tracheal and salivary glands), with an inoperable disease (lacking a surgical tumor sample) and with insufficient tumor sample mass were excluded from the study.
2.2. Study Population
The patient data was extracted from the patient files, where the information included age, tobacco use, tumor stage, treatment protocol, and follow-up information. The demographic and clinical findings of this study are summarized in Table 1 and showed a mean age of 57 years (range 18-83) with a predominance of male patients. The stage of the HNSCC tumor varied from T1N0M0 to T4aN2bM0 with a slight majority of patients (56%) presented with late-stage tumors (T3 and T4) compared to 44% of the patients who presented with an early stage (T1 and T2) tumors. Distant metastasis, especially to the lungs, was reported in 10 (16.4%) patients, also all patients had their tumors removed surgically and 60-70% of the patients were treated with chemo- and radiotherapy. Regarding the level of differentiation of HNSCC: 55.7% of the tumors were moderately differentiated, 23% well-differentiated, and 21.3% poorly differentiated squamous cell carcinomas. The patients’ social history of alcohol consumption was unavailable. This study was approved by the Institutional Review Board (IRB) of the Hashemite University, King Hussein Cancer Center, and King Hussein Medical Center.
2.3. DNA Extraction
Formalin-Fixed Paraffin-Embedded (FFPE) tissue blocks for each patient were obtained from the pathology departments in both centers. For each patient, 5-10 sections of 5 μm thickness were obtained and stored in 2ml tubes ready for DNA extraction using the QIAamp DNA tissue kit (Qiagen, Germany) according to the manufacturer’s instructions. Briefly, the FFPE were deparaffinized by washing twice for 3 minutes in xylene (100%) with heating, then twice with ethanol (100%), to remove residual xylene.
Samples were covered with ATL lysis buffer and 20 μl proteinase K (Roche, Germany) and incubated at 56 ᵒC for 3 hours. Then, 4 μl of RNAse (100 mg/ml) was added and incubated at room temperature for 2 min. After that, AL buffer and ethanol were added and mixed thoroughly, followed by the binding and washing steps, then DNA was eluted in 200 μl AE buffer and stored at -20ºC for subsequent processing [13]. DNA concentration was measured using a Qubit dsDNA HS Kit (Thermo Fisher Scientific, USA) in Qubit 3.0 Fluorometer (Thermo Fisher Scientific, USA) as detailed by the manufacturer.
2.4. HPV Detection and Genotyping
HPV detection and genotype identification were carried out using Real-time PCR (RT-PCR) thermal cycler (Bioer, China). REALQUALITY RQ-Multi HPV detection is an in vitro diagnostic kit (AB ANALITICA, Italy) which is capable of identifying 28 HPV types including 14 high-risk (16,18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, 68), 6 potential high-risk (26, 53, 67, 70, 73, 82), and 8 low-risk (6, 11, 40, 42, 43, 44, 55, 83) genotypes [14]. The kit amplifies a fragment in the region of E6 and E7 in the HPV genome and detects the different genotypes as a pool except for HPV 16 and HPV 18 which are detected individually. The kit includes an internal control (human beta-globin) to monitor for the quality of DNA extraction and amplification process, and also provides a negative control (nuclease-free water) and a positive control (plasmid DNA contain fragments of HPV 16, 18, and 33 for HR-HPV, and fragments of HPV 6, 11, and 26 genotypes for LR-HPV).
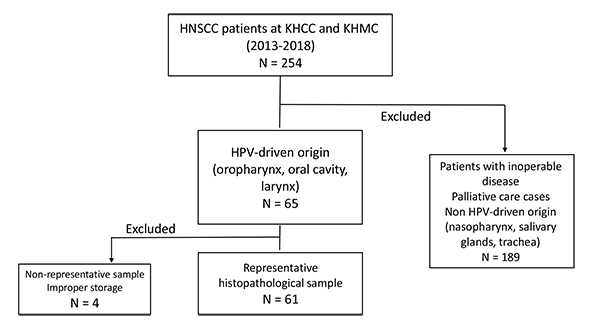
The flow chart explains that a total of 250 Head and Neck Squamous Cell Carcinoma (HNSCC) cases at King Hussein Cancer Center (KHCC) and King Hussein Medical Center (KHMC) between the years 2013-2018 were reviewed initially, and only 65 cases were included in the study based on the established association between HPV and oral, laryngeal and oropharyngeal cancers. Patients with salivary glandular, tracheal and nasopharyngeal tumors were excluded. Moreover, patients with inoperable late-stage disease where no tumor samples were collected were also excluded from the study. Lastly, 4 out of 65 samples were excluded from the analysis because the paraffin-embedded tumor samples did not contain sufficient tumor mass necessary for genetic testing.
| Parameters | Values |
|---|---|
| Mean Age at Diagnosis (SD) | 56.85 (13.23) |
| Sex n (%) | - |
| Male | 47 (77) |
| Female | 14 (23) |
| Tumor Stage n (%) | - |
| T1 | 14 (23) |
| T2 | 13 (21.3) |
| T3 | 27 (44.3) |
| T4 | 7 (11.4) |
| Nodal Category n (%) | - |
| N0 | 22 (36) |
| N1 | 17 (28) |
| N2 | 22 (36) |
| Metastasis n (%) | - |
| M0 | 51 (83.6) |
| M1 | 10 (16.4) |
| Primary Treatment n (%) | - |
| Surgery | 61 (100) |
| Chemotherapy | 37 (60.7) |
| Radiotherapy | 44 (72.1) |
3. RESULTS
This study represents a retrospective analysis of 61 HNCSS patients from 2 central referral hospitals in Amman, Jordan over a period of 5 years (2013 to 2018). There were 47 males and 14 females diagnosed with HNSCC with a distribution ratio of 3.5:1 across our study population Fig. (2). Although the mean age of males (60 years) was higher than that of females (47 years), it was not statistically significant. Tobacco smokers represented 65.6% of the studied sample where 57.1% of the females and 68% of the males were smokers. Cancers of the oral cavity (44.3%), and the larynx (36%) represented the majority of cases included in this study, while oropharyngeal cancer was seen in a small number of patients (19.7%) as shown in Table 2. Patients were divided into 3 groups based on the anatomical origin of the HNSCC lesions. Cancers from the buccal cavity (3.3%), the floor of the mouth (4.9%), the tongue (32.9%) and the gingiva and hard palate (others) (3.3%) were all considered as oral cavity cancers, while cancers from the tonsils (4.9%), the soft palate (4.9%), and the base of tongue (9.8%) were categorized as oropharyngeal cancers. The remaining cancer cases originated from the larynx (36%).
Next, the existence of HPV DNA in the collected samples was assessed. Real-time PCR analysis showed that among the 61 HNSCC patients, 19 cases were HPV positive, and the overall prevalence rate of HPV was 31.1%. The most prevalent and only genotype detected was HPV 16 observed in all 19 cases. These were detected in four females and fifteen males with a mean age of 57.4 years (range 42-83); however, no significant statistical difference was noted between HPV positive and HPV negative patients regarding the mean age of patients or their gender (p = 0.856 and 0.813 respectively) (Table 2). HPV positive smokers were 30% (12/40) compared to 33.3% (7/21) in nonsmokers, but no significant statistical differences were seen between both groups (p = 0.789). By anatomical location, 37% (10/27) of the oral cavity, 41.7% (5/12) of the oropharyngeal, and 18.2% (4/22) of the laryngeal cancers were positive for HPV infection. There was no significant difference among the different anatomical location of cancers between HPV positive and HPV negative cases (p = 0.249). All HPV-positive oral cavity carcinoma was seen in the tongue 50% (10/20), and none was detected in the buccal cavity, floor of the mouth, gingiva or hard palate Fig. (3). The highest HPV infection was seen in the oropharyngeal cancers (41.7%) which is in agreement with the previously reported global estimates (34-46%). HPV-positive cases in the oropharynx were observed to be 33.3% (1/3) in the tonsils, 33.3% (1/3) in the soft palate, and 50% (3/6) at the base of tongue Fig. (3). In laryngeal cancer patients, the detection rate of HPV infection was 18.2% (4/22). HPV 16 genotype was found in 21.4%, 35.3%, and 30.8% of well-differentiated, moderately differentiated, and poorly differentiated cases respectively, and there was no statistically significant association between HPV-positivity and tumor differentiation (p =0.641). Also, there was no association between tumor stage and HPV-infectivity in the studied population (p =0.742) (Table 2).
4. DISCUSSION
Head and neck cancers are closely associated with the use of tobacco and alcohol. Besides, increasing reports have identified a contributing role of HPV infection in the pathogenesis of HNSCC [15]. In this study, HNSCC cases from two central referral hospitals in Amman, Jordan, which fit the inclusion criteria, were recruited. The sample had a targeted preference for oropharyngeal, oral, and laryngeal cancers due to their close association with HPV [16]. HPV prevalence in HNSCC in this study was 31.1% which is in agreement with other regional [17], and global studies [18]. Due to the paucity of local reports investigating the association between HPV and HNSCC, it was challenging to further interpret our results. In one report, the HPV infection rate in association with HNSCC was 14.8%, which is significantly lower than the HPV prevalence obtained in our analysis [12]. However, in comparison with our study, samples in Abu Qatouseh et al. were collected from a single, local private laboratory, but the sample size was higher. These differences in sample representation might account for the significant difference in the association rate between HPV infections and HNSCC. Moreover, while we utilized quantitative real-time PCR to determine the presence of HPV genotypes, Abu Qatouseh et al. only examined the samples using conventional PCR, which might have been less sensitive in the detection of viral DNA, accounting for the lower observed prevalence of HPV in HNSCC samples [19].
In neighboring areas, like Saudi Arabia and Turkey, the prevalence rate of HPV infection in HNSCC in two recent studies was found to be 10% and 2%, respectively, representing a lower HPV infection rate compared to our findings [20, 21]. At the same time, a Syrian study has reported a 43% HPV prevalence in HNSCC patients [22]. Other studies in the region have also shown variable HPV prevalence rates of 85% and 0% in Lebanese and Saudi populations, respectively. In this regard, it is important to mention that the sample size in our study is similar to that in the previously mentioned evidence. For example, Maroun et al., 2020 used a sample of 34 patients diagnosed with HNSCC to estimate the prevalence of HPV infection in a Lebanese subpopulation, while, Jaber et al., 2019, studied a sample of 45 for the same purpose in Saudi Arabia [23, 24]. Accordingly, it is more likely that this variation in the reported HPV prevalence in these studies is attributed to differences in sociocultural behavior, the sensitivity of detection methods, and genetic predisposing factors.
HPV infection as a cause of HNSCC has recently gained great attention and multiple studies have shown a close association between HNSCC and high-risk HPV infection with multiple genotypes, mostly HPV 16, but also with HPV 18, 11, 35, 53 and 82 [25-27]. In our study, despite screening for 28 genotypes of HPV in HNSCC, the only detected genotype was HPV 16, which goes in agreement with some previous studies [12, 21]. Furthermore, the highest rate of HPV 16 infection was seen in the oropharynx, compared to the oral cavity, and the larynx. The use of tobacco in oropharyngeal cancer patients was 66.7% (8/12), while it was 72.7% (16/22), and 59.3% (16/27) in both laryngeal and oral cancer patients, respectively. While smoking rates were high in patients with different types of HNSCC, there was no significant association between tobacco use and the status of HPV infection. However, our data indicates that both factors independently are strong risk factors for the development of HNSCC amongst Jordanians.
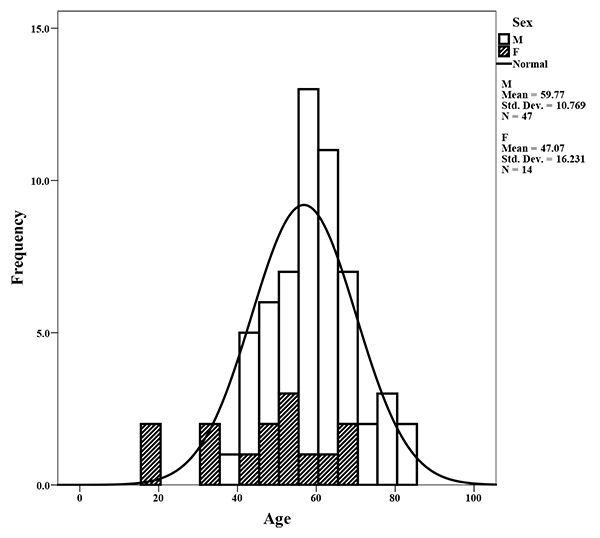
Mean age of males, and females is 60 and 47 years, respectively. Seventy-seven percent of the cases were males compared with 23% females.
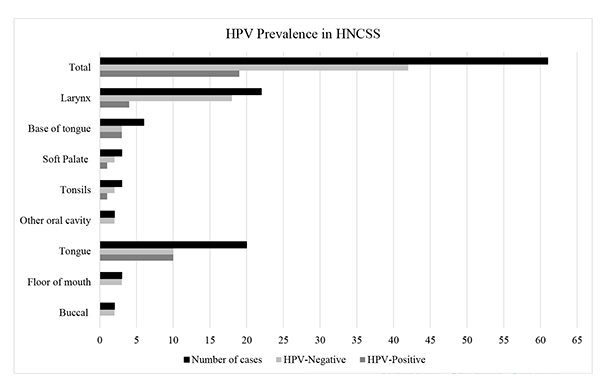
The highest HPV-associated prevalence was seen in the tongue and base of tongue at 50%, followed by the tonsils and soft palate at 33%, and finally the larynx at 18%.
| Characteristics | Total | HPV+ | HPV- | X2 | P value | |||
|---|---|---|---|---|---|---|---|---|
| N | % | N | % | N | % | - | - | |
| Age (years) | - | - | - | 0.033 | 0.856 | |||
| 18-64 | 44 | 72.1 | 14 | 31.8 | 30 | 68.2 | - | - |
| ≥ 65 | 17 | 27.9 | 5 | 29.4 | 12 | 70.6 | - | - |
| Gender | - | - | - | 0.056 | 0.813 | |||
| Male | 47 | 77 | 15 | 31.9 | 32 | 68.1 | - | - |
| Female | 14 | 23 | 4 | 28.6 | 10 | 71.4 | - | - |
| Tobacco | - | - | - | - | - | - | 0.071 | 0.789 |
| Yes | 40 | 65.6 | 12 | 30 | 28 | 70 | - | - |
| No | 21 | 34.4 | 7 | 33.3 | 14 | 66.7 | - | - |
| Anatomical location | - | - | - | 2.780 | 0.249 | |||
| Oral cavity | 27 | 44.3 | 10 | 37 | 17 | 63 | - | - |
| Oropharynx | 12 | 19.7 | 5 | 41.7 | 7 | 58.3 | - | - |
| Larynx | 22 | 36 | 4 | 18.2 | 18 | 81.8 | - | - |
| Tumor stage | - | - | - | - | - | - | 0.108 | 0.742 |
| Early (T1 and T2) | 27 | 44.3 | 9 | 33.3 | 18 | 66.7 | - | - |
| Late (T3 and T4) | 34 | 55.7 | 10 | 29.4 | 24 | 70.6 | - | - |
| Differentiation level | - | - | - | 0.890 | 0.641 | |||
| Well-differentiated | 14 | 23 | 3 | 21.4 | 11 | 78.6 | - | - |
| Moderately-differentiated | 34 | 55.7 | 12 | 35.3 | 22 | 64.7 | - | - |
| Poorly-differentiated | 13 | 21.3 | 4 | 30.8 | 9 | 69.2 | - | - |
The association of HPV infection with laryngeal squamous cell carcinoma is not well-described as with oropharyngeal and oral cancers. A study by Fusconi et al. has shown that despite having a positive HPV DNA in laryngeal cancer specimens, the level of E6/E7 mRNA which is a more specific marker for HPV involvement in laryngeal cancer than p16 was less than 5% [28]. Also, another study by Kreimer et al. has shown that the rate of infection with HPV in laryngeal cancer tissue was 24%, which is in agreement with our results, but also showing that a 19% HPV infection rate was seen in normal laryngeal mucosa as well [27]. In accordance with other studies, our results have shown 18.2% HPV-driven laryngeal cancer [26, 29], compared to studies that showed a lower association [30, 31].
HNSCCs can also be classified into keratinized and non-keratinized. The first type is seen more commonly in elderly men, and it is closely associated with smoking and alcohol consumption. On the other hand, the non-keratinized subtype is associated more often with HPV infection and seen in middle-aged men (40-50 years) with minimal exposure to alcohol and tobacco [32]. In our study, we could not establish an association between the age of patients and the rate of HPV infection. Additionally, no significant statistical association between patients’ gender and HPV status was perceived. While earlier reports showed male predominance in HPV-driven HNSCC [33], recent reports have linked increased women’s exposure to tobacco and alcohol to the increased number of cases diagnosed with HNSCC, especially oropharyngeal cancers [34].
Multiple studies have shown that the TNM classification is important in predicting the patients’ outcome for HPV-negative HNSCC, but has a minimal role in the HPV-positive cases. Klozar et al. proposed that the level of regional lymph node metastasis is a poor predictive factor in the HPV-positive HNSCC patients outcome [35]. Other studies have shown that the N stage, the extracapsular spread, and the early T stages (T1-T3) were of minimal prognostic value in HPV-related HNSCC, and the only poor prognostic indicator was having a stage T4 tumor [36]. In our study, T4 staged patients represented a minority of the recruited patients, and no statistically significant correlation was observed between the stage presentation and HPV positivity.
Most cases of squamous cell carcinoma present as moderate to poorly differentiated tumors. The moderately differentiated and keratinized are usually seen in HPV-negative HNSCCs, while the poorly differentiated and non-keratinized are more common in the HPV-positive HNSCC [37]. Although some studies have shown a statistically significant correlation between the level of tumor differentiation and the HPV 16 infection in HNSCC, our finding failed to show this correlation [12].
Results in this study represent a robust source of information to policymakers in Jordan to consider launching HPV awareness campaigns on the close association between HPV infection and head and neck cancers. Also, these data are alarming in regards to certain social behaviors that are associated with increased risk of HNSCC such as tobacco use, excessive alcohol consumption, and unprotected sexual intercourse. The source of strength in this study includes having representative samples collected from two referral centers and using the real-time as a sensitive detection method. The small sample size represents a limitation to this study.
CONCLUSION
Our study provides the most recent evidence on the prevalence of HPV infection among HNSCC patients in Jordan. Our data indicates that HPV infection rate in Jordan is on the rise when compared to previous reports on the prevalence of HPV-associated HNSCC in the country. This study has also identified HPV genotype 16 as the circulating genotypes in the local community. These findings suggest the implementation of appropriate HPV awareness campaigns and recommend the use of the HPV bivalent vaccine as HPV 16 was the most common subtype identified in HNSCC patients in Jordan.
LIST OF ABBREVIATIONS
| HPV | = Human Papillomavirus |
| HNSCC | = Head and Neck Squamous Cell Carcinoma |
| PCR | = Polymerase Chain Reaction |
| FFPE | = Formalin-Fixed Paraffin-Embedded |
| IRB | = Institutional Review Board |
| HR-HPV | = High Risk-Human Papillomavirus |
| LR-HPV | = Low Risk-Human Papillomavirus |
| E6/E7 | = Early Protein 6 and 7 of Human Papillomavirus |
ETHICAL APPROVAL AND CONSENT TO PARTICIPATE
Ethical approval was obtained from the Institutional Review Board of the Hashemite University (Approval No. 7/2/2015/2016), King Hussein Cancer Center (Approval No. 17 KHCC 106), and King Hussein Medical Center (Approval No. 36/11/2016)
HUMAN AND ANIMAL RIGHTS
No Animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
AVAILABILITY OF DATA & MATERIALS
The authors confirm that the data supporting the findings of this research are available within the article.
FUNDING
This project was funded by a grant provided by the scientific research deanship (Grant No. AM/1601100/10 /13/16), The Hashemite University, Zarqa, Jordan.
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors also would like to thank Dr. George Sahyoun from the MedLabs consultancy group for providing HPV positive controls.


