All published articles of this journal are available on ScienceDirect.
A Nanoparticles based Microbiological Study on the Effect of Rosemary and Ginger Essential Oils against Klebsiella pneumoniae.
Abstract
Background:
Klebsiella pneumoniae is a nosocomial pathogen in outbreaks of hospital infections. It is one of the major factors for morbidity and mortality in hospitalized patients especially those infected with colistin-resistant pathogens. Many plant essential oils have antimicrobial activities and have been investigated as natural sources to combat multiple antibiotic resistances. Moreover, recent advances in phytonanotechnology have created exciting opportunities for the management of many infections.
Objective:
This study aims at investigating the antimicrobial and antibiofilm effect of rosemary and ginger essential oil-based nano-sized formulations on colistin resistant K. pneumonia clinical isolates.
Methods:
Isolation and identification of 30 K. pneumonia isolates from different human samples were done followed by antibiotic susceptibility testing and detection of biofilm gene (mrkD). Examination of the activity of the tested essential oils and their chitosan nanoparticle formulations against the selected isolates was made by determination of their MICs using broth microdilution method followed by biofilm inhibition test and quantitative real-time PCR for the expression of mrkD gene in the presence of the oils and nanoparticles formulations compared to untreated bacterial isolates.
Results:
Our results showed that the minimum inhibitory concentration of rosemary and ginger oils was 1250 μg/ml, that of nanostructured lipid carrier-rosemary oil and nanostructured lipid carrier-ginger oil was 625 μg/ml and rosemary oil loaded chitosan nanoparticles and ginger oil loaded chitosan nanoparticles possessed minimum inhibitory concentration of 156 μg/ml. Results also revealed complete (100%) inhibition for mrkD gene expression when compared to untreated K. pneumonia.
Conclusion:
Oil loaded chitosan nanoparticles showed the highest antimicrobial and antibiofilm activity.
1. INTRODUCTION
Infections caused by Gram negative bacteria are of particular concern; these organisms are highly efficient in the regulation or acquisition of genes encoding antibiotic resistance mechanisms, particularly in the presence of antibiotic selection pressure [1]. In addition, they use multiple mechanisms against the same antibiotic or a single mechanism to affect multiple antibiotics [2].
Klebsiella pneumonia is one of the most common opportunistic pathogens from either nosocomial and community- acquired infections. It causes a wide array of infectious diseases including pneumonia, septicemia, and urinary tract infections UTI [3]. In fact, K. pneumonia is second to Escherichia coli as an etiologic agent of community-acquired UTI [4, 5]. Unfortunately, the ability of K. pneumonia to acquire multiple drug resistance, particularly extended-spectrum β-lactamases (ESBLs), can worsen the condition [6].
In Egypt, K. pneumonia infections rates have gone up dramatically. Molecular typing and virulence analysis are powerful tools that can shed light on K. pneumonia infections, with an increasing prevalence of multidrug-resistant (MDR) patterns [7].
Virulence factors comprise mechanisms allowing pathogenic bacteria to cause infections like siderophores, capsular polysaccharides, aggregative adhesion, and both types 1 and 3 fimbriae play an important role in infection severity [8]. In type 3 fimbriate bacteria, bacterial binding that leads to colonization and biofilm formation on plastic devices such as endotracheal tubes may be more effective. Type 3 fimbriae of K. pneumoniae is chromosomally encoded by the mrkABCDF operon [9], and it consists of the major fimbrial subunit mrkA and the adhesion mrkD that have previously been shown to mediate collagen binding. The MrkA polypeptide contains the major structural component of type 3 fimbriae and is polymerized to form the fimbrial shaft. The MrkA polypeptide might facilitate adsorption to abiotic polymers of recently implanted devices. The MrkD protein functions as the type 3 fimbrial adhesin and mediates binding to extracellular matrix (ECM) proteins such as collagen molecules. The formation of biofilms by K. pneumonia on plastic surfaces is independent of the presence of the MrkD adhesin, but is facilitated by the presence of the fimbrial shaft on the surface of the bacteria [10].
The use of Essential Oils to fight Multidrug Resistance is a generous source of a number of compounds with potential application for the treatment of several infectious diseases. Many investigated essential oils, derived from local botanical plants are promising candidates that might be used against MDR bacteria. Yet there is still a vast flora that could provide additional antimicrobial leads and drugs once systemically explored [11].
Recent advances in phytonanotechnology have provided exciting opportunities for the management of many diseases, including infections. Nanoparticles are colloidal systems with a compact structure in which the therapeutic agent is coated on the particle surface by adsorption or conjugation. These nanocarriers can interact with and penetrate into bacteria, providing unique bactericidal mechanisms. Many nanoparticles can provide sustained, controlled drug release because of their compact nature. Furthermore, the biodistribution profile of nanocarriers can be controlled by modifying their surface physicochemical properties to target the tissue of interest. Therefore, nanotechnology is promising for more efficient drug delivery to target cells, improving the therapeutic management of infectious diseases [12].
There are very few studies focusing on the investigation of antimicrobial activities of medicinal plants nanotechnology against clinically isolated antibiotic-resistant pathogens. Hence the aim of this work is to investigate the antimicrobial effect of Rosemary and Ginger nanostructured lipid carriers and chitosan nanoparticles dosage form on clinically isolated strains of colistin-resistant K. pneumonia.
2. MATERIALS AND METHODS
2.1. Bacterial Strains
Thirty K. pneumonia isolates were obtained from sputum specimens from patients in Alexandria Fever Hospital. For the identification of isolates, VITEK 2 automated system (Bio-Merieux, l’Etoile, France) was used.
2.2. Antimicrobial Susceptibility
Antimicrobial susceptibility testing was performed using the disc diffusion method; antibiotic discs tested were Ampicillin (AMP, 10μg), Ampicillin/sulbactam (SAM, 10/10μg), Cefazolin (KZ, 30μg), Cefepime (FEP, 30μg), Ceftriaxone (CRO, 30μg), Cefuroxime (CXM, 30μg), Imipenem (IPM, 10μg), Gentamicin (CN, 10μg), Tetracycline (TE, 30μg), Levofloxacin (LEV, 5μg), Trimetho- prime/Sulphamethoxazole (TMP/SMX, 1.25/23.75 μg), Chloramphenicol (C, 30μg), Amikacin (AMK, 30μg) (Oxoid, UK) and colistin (CL, 10 μg) (Himedia) on all isolates. Obtained values were interpreted according to the Clinical and Laboratory Standards Institute (CLSI (2019) into sensitive, intermediate and resistant categories.
2.3. Biofilm Formation Assay
The biofilm-forming ability of all K. pneumonia isolates was tested using the microtiter plate method [13]. Overnight, nutrient broth cultures were diluted to 1:100 in fresh nutrient broth; then 200 μL of the freshly inoculated medium was dispensed into wells of a 96-well polystyrene microtiter plate. Negative control wells contained broth only. After 24 h of incubation at 37ºC, the content of each well was aspirated, and each well was washed with 250 μL of sterile physiological saline. Then plates were stained for 5 min with 0.2 ml of 2% crystal violet. Excess stain was rinsed off under running tap water. After the plates were air-dried, the dye bound to the adherent cells was eluted with 160 µl of 95% (v/v) ethanol. The Optical Density (OD) was measured at 630 nm by using an automated reader. Assays were performed in triplicate and the average was taken. The cut-off OD (ODC) was defined as three standard deviations above the mean OD of the negative control; the isolates were classified as follows: O.D ≤ O.Dc, non adherent; O.Dc<O.D ≤ 2 ×O.Dc weakly adherent; 2 ×O.Dc<O.D ≤ 4 ×O.Dc moderately adherent and 4 ×O.Dc<O.D strongly adherent.
2.4. PCR for Detection of the Adhesion mrkD Gene
The oligonucleotide primers synthesized by Thermo Fisher Scientific company (USA) 5'- CCACCAACTATT- CCCTCGAA -3' and 5'- ATGGAACCCACATCGACATT -3' were used to amplify mrkD gene [14]. Genomic DNA from K. pneumonia isolates was extracted by boiling [15]. A 25 μl reaction volume was used for all PCRs with mixtures that contained 12.5 μl of PCR Master Mix reagents (One PCR Ultra Supermix w Fluorescent Dye, GeneDireX - MB208-0100, USA), 50 pmol of the forward and reverse primers and 5 μL of the DNA template. The PCR cycling parameters were: Initial denaturation for 3 minutes at 94°C, denaturation for 40 seconds at 94°C, annealing for 1 min at 55ᴼC, extension for 2 min at 72°C, and final extension for 5 min at 72°C. DNA fragments were detected on 1% agarose gel (GeneDireX - MB755-0100, USA) with ethidium bromide staining.
2.5. Essential oils (EOs) Isolation and Gas Chromatograph/Mass Spectroscopy (GC-MS) Analysis
Ginger and Rosemary were ground into a fine powder and then 200 gm of each was used to prepare the oil by Cleavenger hydrodistillation method [16]. The distilled oil in the graduated tube of 1 ml and 6 ml respectively, was collected and dried over anhydrous sodium sulphate and preserved in a dark glass bottle and then kept in the refrigerator (4-8°C) till use. The prepared oils were analyzed using GC/MS (Thermo Fisher Scientific, Germany). The relative amount of individual components of the total oil is expressed as a percentage peak area relative to the total peak area. Qualitative identification of the different constituents was performed by comparison of their relative retention times and mass spectra with those of Retention Indices (RI) and mass spectra.
2.6. Preparation of Nanostructured Lipid Carrier-EO Nanoparticles
2.6.1. Preparation of Nanoparticles
We prepared two types of nanoparticles for this study. Nanostructured lipid carrier nanoparticles (NLCs) were prepared using stearic acid, Labrafacand Tween 80. Rosemary or ginger oil was incorporated into the lipid phase. Both lipid and aqueous surfactant phases were heated separately to 60oC. The melted lipid phase was then added to the aqueous solution dropwise under high shear homogenizer following sonification [17].
Essential oil-loaded chitosan nanoparticles (Np-CS) were prepared with two concentrations, 1% (w/v) and 0.5% (w/v). Medium molecular weight chitosan solution was first mixed with surfactant and rosemary/ginger oil. The mixture was then cross-linked with tripolyphosphate and stirred until opalescent colloid appeared [18].
2.6.2. Particle Size, Zeta Potential, Polydispersity Index and Entrapment Efficiency of Nanoparticles
Particle size, zeta potential and PDI of both prepared NLCs and chitosan nanoparticles were measured by Dynamic Light Scattering (DLS) using Zetasizer Nano ZS. Samples were diluted several times with deionized water before measuring to avoid multiple scattering.
The entrapment efficiency of rosemary or ginger oils in nanoparticles was determined indirectly by the difference between the total amount of rosemary/ginger oils used in the formulation and the amount of unentrapped oils. The amount of unentrapped oils was determined using a Spin-Pure eppendorf filter tube by centrifugation. The filtrate concentration was determined by UV/VIS spectrophotometer. All the measurements were performed in triplicates [17].
2.6.3. Transmission Electron Microscope (TEM)
The morphology of representing CS-Rosemary nanoparticles was examined by transmission electron microscopy (JEOL, Japan). The average size was calculated using the IMAGEJ software [17].
2.7. Determination of Minimal Inhibitory Concentration (MIC)
The rosemary oil, ginger oil, NSLC-rosemary, NSLC-ginger, Np-CS-rosemary and Np-CS-ginger (2.5 mg/ml) were 2 fold serially diluted in sterile nutrient broth and then 200 µl of formulations were distributed in each well of a 96-well polystyrene microtitre plate. Each well received 10 µl of diluted overnight broth cultures of the tested microorganisms to reach a final inoculum of about 0.5 mcfarland in the reaction wells. The plate was incubated at 37°C for 24 hrs and then visually examined for microbial growth to determine the MIC values. Uninoculated wells containing the oils and their nanoformulations were included in the experiment as negative controls (CLSI (2019)).
2.8. Biofilm Inhibition Assay
The effect of ¾, ½ and ¼ MIC of rosemary oil, ginger oil and their chitosan formulations on biofilm formation was detected by using the microtitre plate method. Different concentrations of antimicrobial agents prepared in 100 μl of TSB were added to microtiter wells containing 100 μl of the inoculum prepared as described above. Quantitation of biofilms was performed after 24 h of incubation at 37°C, as described for the biofilm formation assay. Drug-free medium was used in control wells [19].
2.9. RNA Isolation and Quantitative Real-time RT-PCR
For transcriptional analysis, K. pneumonia was inoculated into 25 ml of nutrient broth in 250 ml shake flasks at a starting OD 600 of 0.05, and then cultured at 37 °C for 5 h with 250 rpm agitation in the presence or absence of ½ MIC of rosemary oil, ginger oil and their chitosan formulation. Extracted RNAs were evaluated by Nanodrop instrument (Thomas Scientific, US). Total RNA was isolated using a RNA-spin™ Total RNA Extraction Kit (iNtRON - 17211, South Korea). The transcription levels of type 3 adhesin fimbriae gene (mrkD) were determined by qRT-PCR, with or without rosemary oil, ginger oil and their chitosan formulation; rpoB [20] was used as housekeeping gene 1 and rrsA [21] as housekeeping gene 2 as control and qRT-PCR data were normalized by rpoB and rrsA transcriptions. qRT-PCR was performed using a TOPreal™ One-step RT qPCR Kit (SYBR Green with low ROX) (enzynomics - RT432S, South Korea) and an ABI StepOne Real-Time PCR System (Applied Biosystems) [22].
3. RESULTS
3.1. Antimicrobial Susceptibility
The antibiotic susceptibility profile for all strains is shown in Table 1. All K. pneumonia isolates were β-lactam resistant with varied resistance degrees for the rest of the tested antibiotics where aminoglycosides and colistin showed the lowest resistance patterns.
3.2. Biofilm Formation
The results showed that 100% (30) of the tested K. pneumonia isolates were strong biofilm formers. The 13 colistin resistant, strong biofilm forming K. pneumonia isolates were selected to continue the study.
3.3. Detection of the Adhesion mrkD Gene by PCR
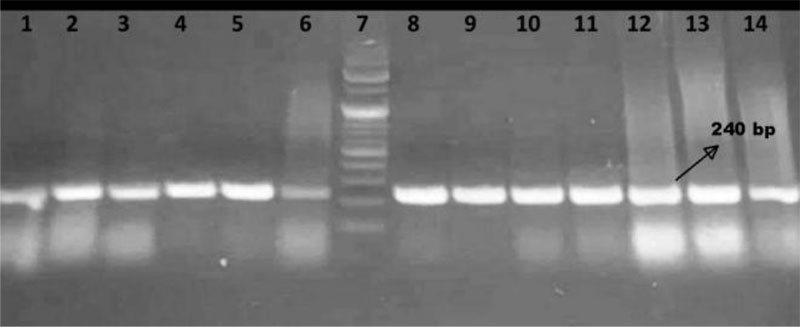
Upon amplifying mrkD gene using the conventional PCR technique, all the 13 selected isolates gave positive results as shown in Fig. (1), where a 240 bp amplification product was visualized in gel electrophoresis analysis.
3.4. Particle Size, Polydispersity Index, Zeta Potential, and Entrapment Efficiency of Nanoparticles
Table 2 presents the Particle Size (PS), Zeta Potential (ZP), Polydispersion Distribution Index (PDI) and Entrapment Efficiency (EE%) of the nano-formulations. NLCs had much larger particle sizes than nanoparticles prepared by chitosan. When increasing the concentration of chitosan from 0.5% to 1%, the nanoparticle sizes also increased. Chitosan is an unbranched cationic biopolymer, therefore, all prepared chitosan nanoparticles carried positive charges. In contrast, nanostructured lipid carriers were negatively charged. However, zeta potential indicated more stable formulation in NLC because the particles expel each other due to stronger negative charges. NLCs had more monodispersion of nanoparticles than CS which had larger pdi indicating more heterogenic dispersed particles. When chitosan concentration increased, the mixture became more heterogeneous as both particle size and PDI significantly increased.
Both NLC and CS formulations showed high entrapment efficiency (> 90%) with the highest value of 99.25% in 0.5% CS-Ginger. In general, CS had higher EE% than NLC.
| Antibiotic |
Resistance (n = 30) |
|
|---|---|---|
| No. | % | |
| Ampicillin | 30 | 100 |
| Ampicillin/sulbactam | 30 | 100 |
| Cefazolin | 30 | 100 |
| Cefriaxone | 30 | 100 |
| Cefepime | 30 | 100 |
| Cefuroxime | 30 | 100 |
| Imipenem | 30 | 100 |
| Amikacin | 11 | 36 |
| Gentamicin | 13 | 43 |
| Trimethoprim/sulfamethoxazole | 30 | 100 |
| Tetracycline | 18 | 60 |
| Levofloxacin | 10 | 33 |
| Chloramphenicol | 24 | 80 |
| Colistin | 13 | 43 |
| Nano-Formulation | PS (nm) | ZP (mV) | PDI | EE% | |
|---|---|---|---|---|---|
| Nanostructured Lipid Carrier (NLC) |
NLC-Ginger | 461.7±8.584 | -25.9 | 0.356 | 92.53% |
| NLC-Rosemary | 476.7±12.3 | -32.7 | 0.45 | 93.47% | |
| Chitosan Nanoparticle (CS) |
0.5% CS-Ginger | 23.3±61.42 | 0.261 | 0.722 | 99.25% |
| 0.5% CS-Rosemary | 22.04± 2.923 | 1.44 | 0.670 | 92.12% | |
| 1% CS-Ginger | 45.77±194.4 | 0.144 | 0.984 | 98.91% | |
| 1% CS-Rosemary | 33.59±4.001 | 0.270 | 0.690 | 94.51% | |
| Tested oils and their Formulations | Concentrations of oils and their formulations | |||||
|---|---|---|---|---|---|---|
| 3/4 MIC | 1/2 MIC | 1/4 MIC | ||||
| Biofilm Classification | % inhibition | Biofilm Classification | % inhibition | Biofilm Classification | % inhibition | |
| Rosemary oil | None | 60 | None | 73 | None | 77 |
| Np-CS* rosemary | None | 95 | None | 97 | None | 98 |
| Ginger oil | None | 60 | None | 73 | None | 87 |
| Np-CS ginger | None | 95 | None | 97 | None | 98 |
3.5. Transmission Electron Microscope (TEM)
Fig. (2) shows the TEM image of 0.5% CS-Rosemary nanoparticles indicating spherical shapes with an average size of around 55 nm. These nanoparticles were well distributed and few agglomerations were observed.

3.6. Determination of MICs
For the selected thirteen isolates, the MIC of rosemary oil and ginger oil was found to be 1250 μg/ml, the MIC of NSLC-Rosemary oil and NSLC-Ginger oil was 625 μg/ml and the MIC value of rosemary oil Np-CS loaded chitosan nanoparticles - and ginger oil Np-CS loaded chitosan nanoparticles was 156 μg/ml. There was a statistically significant difference between the MIC rosemary oil and ginger oil with their NSLC and Np-CS chitosan formulation. (Z = -3.606; p<0.001) Using Wilcoxon Signed Ranks test and also Mann-Whitney test indicated that the MIC was lesser for chitosan formulation (median=156) than for NSLC (median=625), (U=84.5; p<0.01), so chitosan nanoparticles were chosen to test biofilm inhibition assay and RT PCR.
3.7. Biofilm inhibition
Table 3 shows the inhibitory effect of the oils and their chitosan-nanoparticle formulations used on the selected 13 isolates.
3.8. Quantitative Real-time RT-PCR for mrkD, the Adhesin of Type 3 Fimbriae of K. pneumonia
The ability of ½ MIC of the oils and their chitosan nanoparticle formulation to inhibit the expression of adhesion gene mrkD in K. pneumonia treated with oils and their chitosan formulation was assessed using qPCR and using two housekeeping genes rpoB and rrsA as a reference standard. Our results revealed a 100% complete inhibition for mrkD gene expression when compared to untreated K. pneumonia.
4. DISCUSSION
Klebsiella pneumonia is an opportunistic pathogen and a frequent cause of infections. The increased resistance of this pathogen is leading to limited therapeutic options. In the present study, all our tested isolates were resistant to all β-lactam antibiotics; these results were in accordance with the results of Caneiras et al., where 93.5% and 87.1% of their isolates were resistant to ceftazidime and cefotaxime respectively [23]. Similarly, Montso et al. found that 66.7-100% of their isolates were resistant to β-lactams [24]. Moreover, our tested isolates showed 100% resistance to imipenim; in contrast, other studies showed that imipenim was one of the most effective antibiotics [25].
We found that 80% of our isolates were chloramphenicol resistant; also Wu et al. reported that 68.75% of their tested K. pneumonia were chloramphenicol resistant [26]. In contrast to our results, Caneiras et al. found that 83.9% of their isolates were resistant to gentamycin [23], while in our study, aminoglycosides showed better efficacy where the resistance ranged from 36-43%.
Colistin is effective against many Gram-negative bacteria and is considered as one of the last-resort antibiotics for the treatment of human infections caused by multidrug-resistant Gram-negative bacteria. In this study, we reported high colistin resistance (43%) which was also reported by Falagas et al. [27].
Klebsiella pneumonia can form a thick layer of biofilm as one of its important virulence factors, allowing the bacteria to attach to biotic or abiotic surfaces that contribute to drug resistance. Resistance of biofilm-mediated infections to effective chemotherapy has many adverse effects on patient outcomes and survival [28]. All our tested isolates (100%) were strong biofilm-forming; this percentage is much higher than the results found by Seif et al. [29] where only 33%, 52.1%, 8.5% and 6.4% of their isolates were strong, moderate, weak and non-biofilm formers.
Rosemary EO expressed important antibacterial activities on many bacterial species [28]. Ginger EO is widely used as an ingredient in food, pharmaceutical, cosmetics, other industries and antimicrobial activities. These oils are identified as monoterpenes in the literature and are indicated to affect the cell membrane as a toxic agent deforming the structure and function of the microbial cell membrane [30]. Many studies have reported an enhancement of the antimicrobial and physical properties of EOs-loaded nanoemulsions as compared to their conventional emulsions [31].
In this study, the MIC of rosemary and ginger oils against 13 isolates was found to be 1250 μg/ml, whereas the MIC of these oils formulated on Nanostructured lipid carrier nanoparticles was 625 μg/ml, and the lowest MIC was found with the use of Essential oil-loaded chitosan nanoparticles formulation which was 156 μg/ml. This shows that there was an 8-fold increase in the antibacterial effect of these essential oils against K. pneumonia isolates which can be attributed to the synergetic effect of the chitosan and essential oil when being combined in NPs. Tavassoli et al. indicated that rosemary EO showed higher inhibitory effects on bacteria (MICs: 0.5-1.5 mg/mL) tested than on yeasts [32]. Several studies have demonstrated that nanoparticles (NPs) functionalized with essential oils have a significant antimicrobial effect against MDR pathogens due to increased solubility and chemical stability, decreased rapid evaporation and minimized degradation of active essential oil components [31]. Many essential oils have excellent antimicrobial activity against pathogenic microorganisms. However, their utilization is very limited due to low water solubility and less stability due to environmental factors including heat, moisture, and oxygen. Nano-sized formulations have emerged as a viable solution in order to improve water dispersion and protect EOs from degradation [32, 33]. They may also prevent evaporation of volatile components and offer controlled and targeted release [33].
Because the formation of biofilms was one of the most important problems affecting the treatment of infectious diseases particularly in MDR strains, new essential oils with high bacteriostatic or bactericidal activity grouped at phytoalexins were being used in the treatment of different infectious diseases. Being produced to protect against microbial infections, it was not surprising to see their inhibition effects on microbial growth or antibiofilm effects on different microorganisms, hence leading to new treatment strategies. Various researches have been investigated with different essential oils and it has been observed that these oils have antibiofilm effects on various bacterial and fungal species [34].
Different Sub MIC concentrations ranging from ¾ to ¼ MIC were used to inhibit biofilm formation. Table 3 shows biofilm inhibition of the oil alone and its Chitosan formulation where a drastic decrease in biofilm formation was noticed with Chitosan formulation.
In vitro, fimbriae from a variety of bacterial species were shown to facilitate biofilm formation. The type 3 fimbriae of K. pneumonia are comprised of the major fimbrial subunit (MrkA) and the adhesin (MrkD) that has been shown to mediate binding to collagen. The presence of the MrkD adhesin was necessary for growth on collagen-coated surfaces, unlike biofilm formation on abiotic plastic surfaces. Fimbriate strains lacking the MrkD adhesin did not efficiently grow on and adhere to these surfaces. The MrkD adhesin could enable bacterial adhesion and growth on polymers coated with host-derived proteins. Our results showed a 100% total inhibition of mrkD gene expression upon treatment of K. pneumonia with ½ MIC of the oils and their chitosan nanoparticle formulation relative to untreated cultures in qPCR because the addition of carboxymethyl groups enhances the overall positive charge on chitosan molecules, leading to greater contact with negatively charged components as lipopolysaccharides and proteins present in the bacterial membrane, leading to degradation of the membrane and the release of large intracellular material outside of the cells [35, 36].
CONCLUSION
In conclusion, oil loaded chitosan nanoparticles show the highest antimicrobial and antibiofilm activity which can be used as antibiotic alternatives to promote health and reduce the emergence of antibioticresistance.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Faculty of Pharmacy, Damanhour University, Egypt, with Ref.no. (318PM3).
HUMAN AND ANIMAL RIGHTS
No human or animals were used in the study.
CONSENT FOR PUBLICATION
All patients participated on a voluntary basis and gave their informed consent.
AVAILABILITY OF DATA AND MATERIALS
The data that support the findings of this study are available from the corresponding author [R.A], upon reasonable request.
FUNDING
None.
CONFLICT OF INTEREST
The authors whose names are listed certify that they have no affiliations with or involvement in any organization or entity with any financial interest, or non-financial interest in the subject matter or materials discussed in this manuscript.
ACKNOWLEDGEMENTS
I would like to thank the Faculty of Pharmacy, Pharos University, and the Faculty of Pharmacy. Damanhour University, for providing material and moral support.


