All published articles of this journal are available on ScienceDirect.
Molecular Detection and PCR-RFLP Analysis of Mucoviscosity-Associated Gene A (magA) in Clinical Isolates of Multidrug-Resistant Klebsiella pneumoniae in Bangladesh
Abstract
Background and Objective:
The mucoviscosity associated gene A (magA) in the hypermucoviscous variants of K. pneumoniae is reported to be associated with invasive infections and considered a virulence factor. We sought to analyze the magA genes in K. pneumoniae isolates in the clinical specimen collected from Bangladesh.
Methods:
We established a multicenter cohort of patients with Klebsiella infection hospitalized at 05 different hospitals between September 2016 and April 2017. We collected 313 K. pneumoniae isolates from patients who consented to participate in the study. The isolates were evaluated for harboring the magA genes using a single-tube multiplexed polymerase chain reaction. The magA genes were analyzed by PCR-RFLP technique using two enzymes, namely PciI and SmaI. Antibiogram assay using 12 commercially available antibiotic discs was performed on all the isolates.
Results:
The presence of K. pneumoniae specific gene (ureD) was confirmed in all the isolates. The percentage of isolates harboring the magA gene was 7.34%(23 isolates), the majority of which was collected from the patients admitted in intensive care units (16 isolates, 69.6%), and infectious diseases wards (5 isolates, 21.7%). PCR-RFLP analysis revealed that for 7 out of 23 isolates, where Sma1 could not cleave the magA gene. All the isolates showed resistance to ampicillin, carbenicillin cefradine, chloramphenicol, erythromycin, kanamycin, and sulphamethoxazole, though the extent was varying. However, imipenem showed 100% sensitivity to all the tested isolates.
Conclusion:
This study demonstrates the presence of the magA gene in multidrug-resistant clinical isolates of K. pneumoniae collected from Bangladesh.
1. INTRODUCTION
Klebsiella pneumoniae is an encapsulated gram-negative bacterium of the Enterobacteriaceae family [1]. As a component of the human gut microbiota, this bacterium is usually harmless to the body. It turns into a threat only if
transmitted into other parts of the body. Nevertheless, infections are infrequent as healthy immune systems of men can resist the bacterium effectively. However, in immunocompromised patients, it causes severe diseases, e.g., septicemia, pneumonia, urinary tract infections (UTI), liver abscess, chronic lung disorders, and nosocomial infections [1, 2].
K. pneumoniae are intrinsically resistant to ampicillin. Besides, they ubiquitously possess an inducible chromosomally encoded ß-lactamase (cephalosporinase), making them resistant to first and second-generation cephalosporins [3, 4]. In addition, clinical reports indicate that they are increasingly becoming resistant toward tigecycline, colistins, and polymyxin antibiotics, turning out to be a pan drug-resistant bacteria, or in other words, a ‘superbug’ [5-8]. Consequently, once the modest bacterium, has now turned into a serious threat to public health [9, 10].
Among the Gram-negative bacteria, K. pneumoniae are the second-most frequent hospital-acquired and nosocomial pathogens [11, 12]. According to a recent report, these bacteria are the primary reason for 3 – 15% of all hospital-acquired infections, and the carrier rate can be as high as 77% among hospitalized patients [13, 14]. The constant unregulated use of antibiotics and improper hygiene managements are among the foremost reasons behind this spread of multi-drug resistant strains [13]. Proper diagnosis and transmission management have been the key to control the spread of this pan drug-resistant bacterium [15-18]. In clinical settings, K. pneumoniae infections are usually diagnosed by examining samples of the infected tissue, e.g., sputum, urine, or blood by culture-based methods [15, 19]. Depending on the site of infection, ultrasounds, X-rays, and computerized tomography (CT) can also be useful [20]. However, these diagnosis techniques have several limitations, including turnaround time, narrow application, low specificity, and sensitivity [15, 19]. Cutting-edge technologies, e.g., Sanger and next-generation sequencing, have gained a foothold in health care for diagnosis of infection at the individual level, outbreak detection, transmission mapping, and source tracing [21, 22]. However, high-throughput screening through targeted sequencing is expensive, labor extensive, and requires expertise. Simple molecular techniques, e.g., PCR, PCR-RFLP assays are, in contrast, rapid and easy to perform, but have considerable sensitivity, specificity, and wide-range of application in infection detection [15, 23].
Several virulence factors, e.g., adhesion, siderophore, O antigen, and capsule attribute to its pathogenesis [24]. Among the 77 forms of capsular antigens, K1 and K2 are mostly studied. Between these two, K2 is the most common type isolated from patients with pneumonia, bacteremia, and UTI. Previous genetic studies identified that the genomic map of K. pneumoniae capsule contains a gene cluster subsuming the cps (capsular polysaccharide synthesis), rmpA, rmpA1 and rmpA2 (regulator of the mucoid phenotype A, A1, and A2, respectively), wb (the wb gene cluster codes for O-specific polysaccharide), and magA (mucoviscosity associated gene A) [25, 26]. Functional aspects of these genes include the synthesis of capsular polysaccharide, the regulation of the extracellular polysaccharide capsule's synthesis, and the production of lipopolysaccharide, respectively. In most of the isolates of K. pneumoniae, these four genes remain conserved. Among the genes mentioned above, magA is a novel virulent gene that causes acute infection of Klebsiella, e.g., septicemia, bacteremia, pneumonia, and lung and liver abscesses [27, 28]. It spans about 1 kb region and involves in biosynthesis, transfer, and glycosylation of lipopolysaccharide [27]. The presence of chromosomal magA gene confers a hyper-viscous phenotype, which can be characterized by a positive string test (formation of a mucoviscous string of 5 mm diameter during passing loop through a colony) and resistance to phagocytosis. Among 77 characterized capsular serotypes (K), a major portion belongs to K1 and K2 serotypes. Based on these observations, the genetic locus containing magA can be targeted as a new pathogenicity island responsible for increasing the virulence of K. pneumoniae strains [29].
In the present study, we sought to investigate the presence of magA gene in isolates of K. pneumoniae obtained from patients from different wards of Bangladeshi hospitals.
2. MATERIALS AND METHODS
2.1. Samples
From September 2016 to April 2017, we collected 313 clinical specimens from patients, including urine, blood, trachea, and wounds. Authorized personnel collected the specimen at the bedside from the patients (one isolate per patient) admitted in 05 hospitals of 05 different regions, namely Sylhet, Dhaka, Mymensingh, Rajshahi, and Khulna districts of Bangladesh. All isolates were subjected to standard confirmatory tests, which included microscopic evaluation, Gram staining, oxidase test, catalase test, and growth on SIM (sulfide, indole, motility), lysine iron agar, phenylalanine agar, urea agar, malonate, blood agar, and MacConkey agar. Bacterial isolates were stored in tryptic soy broth (TSB) supplemented with 20% glycerol at -20°C.
2.2. Design of Primers
Primer3Plus tool was utilized to design the specific primers for PCR-based identification of magA and ureD genes in K. pneumoniae isolates [30]. The ureD (urease D, an accessory gene in the urease gene cluster) was coamplified as an internal control, as it is a specific gene for K. pneumoniae [31]. Since we took advantage of a single tube multiplexed PCR for this study, careful optimization of Ta of the primers was critically important. We used Primer3Plus, OligoAnalyzer 3.1, and Multiple Primer Analyzer tools to optimize the annealing temperature (Ta) of the primers (Table 1) [30, 32-34]. The designed primers were commercially purchased from Integrated DNA Technologies.
| Target | Primer | Tm o(C) | Ta o(C) | Product size (bp) |
| ureD | Forward: ATCGTCACTGAGGAAACGC | 54.4 | 49 | 590 |
| Reverse: ACCCGGAAGAAGAGACCT | 54.3 | |||
| magA | Forward: TGGCTTAGACCGATGGTTG | 53.9 | 1057 | |
| Reverse: ACATTGCCGCTACTACAGG | 53.6 |
2.3. Genomic DNA Extraction
We extracted genomic DNA from the obtained isolates using a commercially available bacterial DNA isolation kit pursuing the manufacturer's instructions (GenElute™ Bacterial Genomic DNA Kit, Sigma-Aldrich, Cat.: NA2120). The DNA quality was examined by electrophoresis on 0.7% agarose gel (Ultra-pure Agarose, Invitrogen, Cat. No.: 6500-25).
2.4. Single Tube Multiplex PCR Amplification of ureD and magA Genes
The single-tube multiplexed PCR was carried out with 3.0 µL of template DNA, 1 µL of each primer (10 μM), 12.5 µL of master mix (Promega Hot Start, Cat No.: M7122) and 5.5 µL of nuclease-free Water (Promega, USA) in a total volume of 25 µL. The magA and ureD genes were amplified simultaneously using the ProFlex™ 2 x 96-well PCR System (Applied Biosystems, Cat No.: 4484076). The PCRs were initially heated to 96°C for 7 minutes, followed by 35 cycles of denaturation (96°C, 1 minute), annealing (49°C, 1 minute), and extension (72°C, 1 minute). A final extension was carried at 72°C for 7 minutes. PCR products were evaluated by electrophoresis on 1.2% agarose gels ((Ultra-pure Agarose, Invitrogen, Cat. No.: 6500-25). 1-kb DNA ladder (Promega, Cat. No.: G5711) was used to compare DNA band sizes.
Using commercially available DNA purification kits, we purified 15 randomly selected PCR products of both genes from the TBE buffered agarose gels (GeneJET Gel Extraction Kit, Cat.: K0692). Purified PCR products were sent to Macrogen Corp. (Geumcheon-gu, Seoul, Rep. of Korea) for Sanger sequencing. The sequence outcomes were aligned and compared with the reference sequences of the genes using the BLAST program at the web interface of the National Center for Biotechnology Information (http://blast.ncbi.nlm.nih.gov/Blast.cgi) [35]. The confirmed samples were used as the control for the rest of the analysis.
2.5. Restriction Digestion
10 µL of the PCR products, which produced positive outcomes for both the genes, were transferred to a separate 0.5 µL-Eppendorf vial and added with 17 µL of nuclease-free water. Then 1.5 µL of each of PciI (Thermo Fisher Scientific, Cat: ER1871) and SmaI (Thermo Fisher Scientific, Cat: ER0661) restriction enzymes were added and mixed carefully to the solution and incubated at 32°C for an overnight period (Table 2). The incubation temperature was set at the mean temperature of the optimum incubation temperatures of the enzymes mentioned above. Using UV illumination, the restriction fragments were compared against a 1-kb plus DNA ladder (Thermo Scientific, Cat. No.: FERSM1331) on 1.5% agarose gels. This step was repeated multiple times to confirm that the restriction digestion was not incidental.
2.6. Antibiogram Assay of the Isolates
A disc diffusion method was adopted to investigate antibiotic profiling of K. pneuomoniae isolates using 11 commercially available discs [36, 37]. High-quality absorbent paper discs impregnated with the following antibiotics were used: ampicillin (10 µg/disc), carbenicillin (10 µg/disc), cefradine (25 µg/disc), chloramphenicol (30 µg/disc), ciprofloxacin (30 µg/disc), erythromycin (15 µg/disc), gentamycin (10 µg/disc), imipenem (10 µg/disc), kanamycin (30 µg/disc), levofloxacin (5 µg/disc), streptomycin (10 µg/disc) and sulphamethoxazole (25 µg/disc). A fresh bacterial culture inoculated on Tryptocasein Soy Agar plates (Sigma-Aldrich, Cat.: 22091) by spreading. An antibiotic disc and a blank disc (as negative control) were placed carefully onto each culture plates and incubated overnight at 37°. After the incubation period, the zone of inhibition due to the activity of antibiotic-containing discs was measured following the protocol described by Sharmeen et al. [38].
3. RESULTS
3.1. Patient Characteristics and the Isolates Obtained
In the current study, 313 isolates of K. pneumoniae were obtained from the intensive care units (ICU), internal medicine and infectious diseases wards of the five hospitals (Table 3). Out of these 313 samples, 192 isolates (61.3%) were collected from females and the rest (n = 121, 38.7%) were from males. The age of the patients ranged from 13 to 84 years, with a mean of 49.50 ± 18.24 years.
| Restriction enzyme | Target | Cutting site | Cutting positions | Fragment sizes (bp) |
| SmaI | PCR amplified ureD gene of K. pneumoniae | CCC↓GGG | 453/454 | 453, 372 |
| PciI | PCR amplified magA gene of K. pneumoniae | A↓CATGT | 958/962 | 958, 269 |
| Features | Values |
|---|---|
| Total no. of isolates (n) | 313 |
| Age (years) of the patients (mean ± SD, range) | 49.50 ± 18.24, 13 - 84 |
| Female: male | 1.0: 0.63 (192: 121) |
| Regions from where specimen were collected (n, %) | - |
| Sylhet | 94, 30.03 |
| Dhaka | 69, 22.04 |
| Mymensingh | 57, 18.21 |
| Rajshahi | 52, 16.61 |
| Khulna | 41, 13.1 |
| Hospital units from where specimen was collected (n, %) | - |
| Intensive care unit (ICU) | 144, 46 |
| Internal medicine unit | 94, 30 |
| Infectious diseases wards | 75, 24 |
| Type of specimen from where isolates were collected (n, %) | - |
| Urine | 159, 50.7 |
| Trachea aspirates | 64, 20.4 |
| Blood | 49, 15.6 |
| Wound | 41, 13.1 |
3.2. Multiplex PCR Outcomes
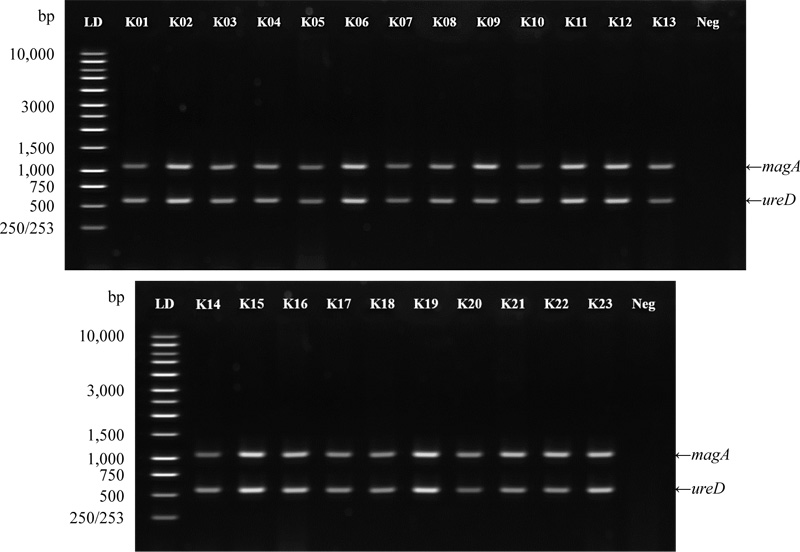
All the isolates carried the ureD gene (served as an internal control), and 23 isolates harbored the magA gene (Fig. 1). Out of these 23 isolates, 07 were collected from Dhaka (10.14%, n = 69). Among the rest 16 isolates, 05 were from Mymensingh (8.77%, n = 57), 08 were from Sylhet (8.51%, n = 94) and 03 were from Rajshahi (5.77%, n = 52). None of the isolates collected from the Khulna region harbored the magA gene. Hence, the overall prevalence of the magA gene in our studied subjects was 7.34% (n = 313). The isolates harboring magA were mostly isolated from the patients hospitalized in the intensive care unit (ICU) (Table 4). Twenty-three out of the 313 isolates harbored the magA gene, and most of these isolates were collected from the patients admitted in intensive care units (16 isolates, 69.6%) and infectious diseases wards (05 isolates, 21.7%).
| Features | No (%) |
| Total | 23 (7.34) |
| Hospital units from where specimen was collected (n, %) | - |
| Intensive care unit (ICU) | 16 (69.6) |
| Infectious diseases wards | 5 (21.7) |
| Internal medicine unit | 2 (8.7) |
| Type of specimen from where isolates were collected (n, %) | - |
| Urine | 8 (34.8) |
| Trachea aspirates | 6 (26.1) |
| Blood | 5 (21.7) |
| Wound | 4 (17.4) |

3.3. PCR-RFLP Analysis
After overnight incubation, PciI could digest the magA gene into two fragments (958 and 269 bp) in 16 out of the 23 isolates. On the contrary, SmaI digested the ureD gene into the desired fragments (453 and 372 bp) in all 23 isolates (Fig. 2).
3.4. Antibiotic Sensitivity of K. pneumoniae Isolates
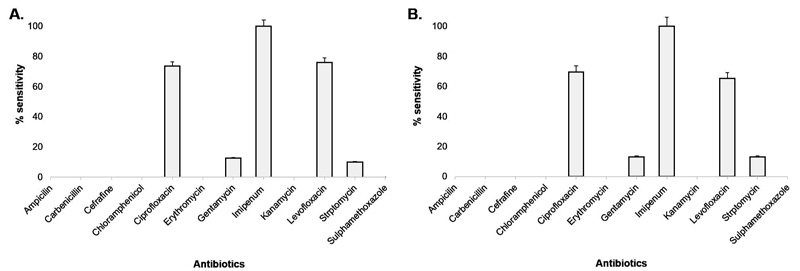
All the isolates exhibited resistance to ampicillin, carbenicillin, cefradine, chloramphenicol, erythromycin, kanamycin, and sulphamethoxazole. Ciprofloxacin and levofloxacin showed ~75% sensitivity, whereas gentamycin and streptomycin exhibited ~12% sensitivity. Two isolates, both collected from Mymensingh district, showed a 10% sensitivity for streptomycin. Imipenem was the only antibiotic used, which showed 100% sensitivity to all the tested isolates (Fig. 3). Among the 23 isolates that contained the magA gene, imipenem, ciprofloxacin, levofloxacin showed 100%, 69.6%, and 65.28% sensitivity, respectively. The sensitivity of both gentamycin and streptomycin was 13.04% (Fig. 3).
4. DISCUSSION
K. pneumoniae are responsible for several nosocomial infections among the debilitated elderly and children [39]. They cause significant morbidity following urinary tract infections, septicemia, and pneumonia due to high resistance to extensively administered antibiotics [9, 40].
The magA gene, which plays a critical role in polysaccharide capsule production of the bacterium, is one of the critical factors in K. pneumoniae associated virulence [41]. Previous studies reported that the secondary acquired infections, e.g., eye infection, bacteremia, sepsis, and meningitis, are magA related diseases [42, 43]. In the present study, over 7% isolates were positive for the presence of magA gene (Table 3). Amrai et al., studied 173 K. pneumoniae isolates collected from different clinical samples taken from Iran and reported 2.3% prevalence of the magA gene [44]. The prevalence of the gene in Bangladeshi subjects was significantly higher (p = 0.0203; χ2= 5.386). Previous studies on clinical samples of K. pneumoniae, invasive and non-invasive metastatic infections reported that the prevalence of the magA gene was 08 – 100% [45-50]. Another remarkable observation was found studying isolates collected from the liver abscess that none of the samples were positive for the presence of the magA gene.
Previously, the magA gene was considered one of the primary reasons for the prevalence of these bacteria in East and South Asian countries [51]. Several reports have suggested the magA gene as a root among geographical location, genetic predisposition, and susceptibility to the infections [51-54]. Struve et al., sorted out that only K1 serotype of Klebsiella spp. isolated from patients with liver abscesses carries the magA gene [29]. In the current study, most of the species containing magA gene were isolated from patients in ICUs, and trachea and urine samples. It was noticeable that long-time hospitalization and the use of invasive medical tools enhanced the susceptibility of patients towards a Klebsiella infection. Our study suggests that the evaluation of magA could be considered a suitable index for rapid molecular identification and diagnosis of these bacteria. Besides, it can be used for the timely treatment of infections caused by K. pneumoniae.
This study describes a PCR-RFLP based technique for precise detection and analysis of magA gene in K. pneumoniae isolates collected from clinical specimens. Our analysis revealed that all the isolates were uniformly cleaved by Pci1. Nearly 1 in every 3 isolates (30.43%) produced negative results for Sma1. This variation by restriction endonuclease digestion of PCR products exerted significant diversity in the magA gene. What it translates into is, the magA gene in K. pneumoniae is significantly evolving in clinical isolates of Bangladesh.
Previous studies affirmed that K. pneumoniae are resistant to most of the antibiotics and thus, they are termed as multidrug-resistant. Our study revealed that K. pneumoniae isolates collected from clinical samples are 100% resistant to several antibiotics, including ampicillin, cefradine, chloramphenicol, erythromycin, kanamycin, and sulphamethoxazole. Only imipenem was found fully effective against all the isolates. These outcomes support previous reports from Dhaka and Sylhet regions of Bangladesh [23, 55, 56]. Our study suggests that multi-drug resistant strains of K. pneumoniae are prevalent across the country.
The antibiotic-resistant pattern has continuously been increasing at an alarming rate, primarily due to the overdose and unregulated use of antibiotics. Nowadays, powerful antibiotics are prescribed in the treatment of most of the diseases, contributing to the spread of multidrug-resistant bacteria [36]. For effective treatment, it is critically important to evaluate the antibiotic-sensitivity status of the causative agents [36, 37].
CONCLUSION
Infection with multi-drug resistant K. pneumoniae is now a global public health concern. Our study suggests that magA producing K. pneumoniae strains are prevalent in the clinical samples of Bangladesh. It also points out that this gene continues to evolve in K. pneumoniae strains in this country. We suggest that the magA gene, coupled with the ureD gene (as an internal control) in the single-tube multiplexed PCR, can be used as a reliable genetic marker for the rapid diagnosis of multi-drug K. pneumoniae infections. Considering the presence of the magA gene among clinical isolates of K. pneumoniae, especially in the isolates collected from ICUs, it is necessary to adopt suitable infection controlling tools and appropriate therapeutic approaches to prevent further spread of this virulent organism in our hospital settings.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Ethical and Animal Care Committee of the Shahjalal University of Science and Technology.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Written informed consent was obtained from all the participants.
AVAILABILITY OF DATA AND MATERIALS
The authors confirm that the data supporting the findings of this research are available within the article.
FUNDING
The authors received no formal funding for this study.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors wholeheartedly praise the cooperation by the hospital authorities who helped in sample collection for this investigation. We also appreciate the wholehearted cooperation of Prof. Md. Faruque Miah and his lab at Shahjalal University of Science and Technology.


