All published articles of this journal are available on ScienceDirect.
An Overview on Resistivity, Diagnostic Challenges and Zoonotic Significance of: Mycobacterium avium ssp. paratuberculosis (MAP)
Abstract
Background:
Mycobacterium avium ssp. paratuberculosis (MAP) is a gram-positive, contagious, rod-shaped intracellular pathogen.
Methods:
MAP is the etiologic agent of Johne’s disease in cattle, and has tremendous economic effect in the ruminant industry. Simultaneously, the MAP has also been suspected as a cause of Crohn’s disease in humans.
Results:
There has been a challenge in the diagnosis of MAP due to its long incubation period, unknown pathogenesis & cross-reactivity among its closely related sub-species. The survival of MAP inside the host macrophages & monocytes, is still unclear. Resistivity & survival of MAP in the outside environment is also high.
Conclusion:
Thus, more research about its pathogenesis, control, and potential role as a zoonotic pathogen must be carried out in future.
1. INTRODUCTION
Mycobacterium avium ssp. paratuberculosis (MAP) belongs to order Actinomycetales, which includes diverse microorganisms. Genus Mycobacterium is differentiated based on their ability to produce mycolic acid. Earlier, species were differentiated based on phenotype [1]. It is gram-positive, acid-fast & intracellular pathogen [2]. Unlike other mycobacteria, paratuberculosis doesn’t produce any mycobactin, compound necessary for its growth. Hence it is dependent on external sources [3]. Based on the host of infection, cattle, and sheep, MAP was originally classified as “C type” and “S type”, respectively [4]. However, recently, with pulsed-field electrogelphoresis (PEGE), MAP is reclassified into subtype I, subtype II, and subtype III strains. The whole genome-based sequencing has shown that subtypes (I & III) belongs to S type and subtype II to C type [5].
MAP is a causative organism of Johne’s disease (JD) and a leading candidate behind the infectious theory of Crohn's disease [6]. JD is a chronic gastroenteritis disease of cattle characterized by cachexia, and in some cases, diarrhea. It primarily affects domestic & free-ranging domestic animals but also reported in primates, rabbits, stoats & foxes [7]. Crohn's disease is a chronic inflammatory disease of bowel with trans-mural inflammation and granuloma formation, as observed in JD [8].
2. WHY WE NEED TO KNOW ABOUT MAP
JD has a significant, negative economic impact on the ruminant industry and has been proposed as a pathogenic organism to humans [9]. Along with overwhelming expenses of examination & treatment, the economic impact of JD is early culling of an infected cow, reduced milk yield, and decreased slaughter value of the animal [10]. A total loss from JD has been assessed to 12-15% of the total value of economic production [7]. So, it is one of the insidious problems in the livestock industry, along with a potential role as a “zoonotic agent”.
3. WHAT MAKES MAP TOUGH?
3.1. Survival of MAP Inside the Host (Macrophages & Monocytes)
The hallmark of mycobacteria is their propensity to infect macrophages and exquisitely adapted to survive in host despite the aggressive immune response. The mechanism of interference of MAP in phagosome maturation is still unknown. The host immune system begins a series of attacks with the deployment of gamma delta cells, CD4+Tcells, and cytolytic CD8+ T cells. These cells interact with each other & infected cells, through a series of cytokines [11]. Genes involved in cholesterol homeostasis, calcium homeostasis, and antibacterial defense mechanism are down-regulated and the granulysin gene is upregulated. This possible mechanism might be in favor of the survival of MAP inside macrophages [12]. MAP subverts host immune response by failing to induce Janus kinase/signal transducers and activators of transcription (JAK/STAT) pathway, a hallmark of gamma interferon activation. Similarly, increased expression of negative receptors, suppressor of cytokine signaling 1 (SOCS1) & suppressor of cytokine signaling 3 (SOC3), and decreased expression of positive receptors chain 1 & 2, also supports the deactivation of gamma interferon during host-pathogen cellular signaling [13].
3.2. Survival of MAP Outside the Host
Environmental stress of biotic & abiotic origin resulted in elevated biofilm formation. This biofilm enhances the increased resistance of pathogens [14]. The survival of MAP in feces & soil is very high. In an experiment with cattle slurry, pig slurry, and an equally mixed slurry of both, the survival time of MAP was 98 days, 182 days, & 168 days respectively, in the anaerobic condition at 15° C. However at 5°C, for all of them, the survival time was 252 days [15]. Similarly, the MAP has high thermal tolerance, high resistance (ultra-violet light/ chlorine), and high survival in water [7]. Ghosh et al. [16], showed that MAP prevents phagosome maturation and reduces its acidification. At the transcriptional level, a large number of genes (n=300), were significantly and differentially regulated=, which includes sigma factor (sigH) genes that were responsible for the survival of MAP inside gamma interferon activated bovine macrophages [16].
4. WHAT MAKES MAP DIAGNOSIS DIFFICULT?
Currently, there are several methodologies for the detection of MAP, like serodiagnosis, Enzyme-Linked Immunosorbent Assay (ELISA), Polymerase Chain Reaction (PCR), amplification of MAP specific sequences, measurement of gamma interferon, and bacterial culture. However, still, some variables like antigen specificity & sensitivity, reproducibility, time for colony detection, PCR inhibitors in samples & cross-reactivity are the factors that limit its accurate diagnosis [17].
4.1. Early Diagnosis at the Prepatent Period (Subclinical Phase)
MAP has a prolonged prepatent period and is visible in later stages. However, shedding of the organism may take place before the disease is visible, as highlighted in the MAP lifecycle in the herd by Anne Lee, in Fig. (1) [18]. Earlier, the cellular mediated immune response is higher, and in the later stage, the humoral immune response becomes predominates [2]. Serological and cultural methods are popular in the livestock industry. Even with DNA/RNA detection methods, there is an issue of genetic relatedness to other mycobacteria. Currently, there are no gold standards to validate the findings of MAP [19]. The bacteriological cultural test is the most suitable at herd level. As infection of animals & effectiveness of test is more effective in later stages, a greater understanding of pathogenesis is critical for developing suitable diagnostic test and control of the disease earlier [20].
4.2. Close Relatedness of MAP & M.Avium Ssp.Avium (MAA)
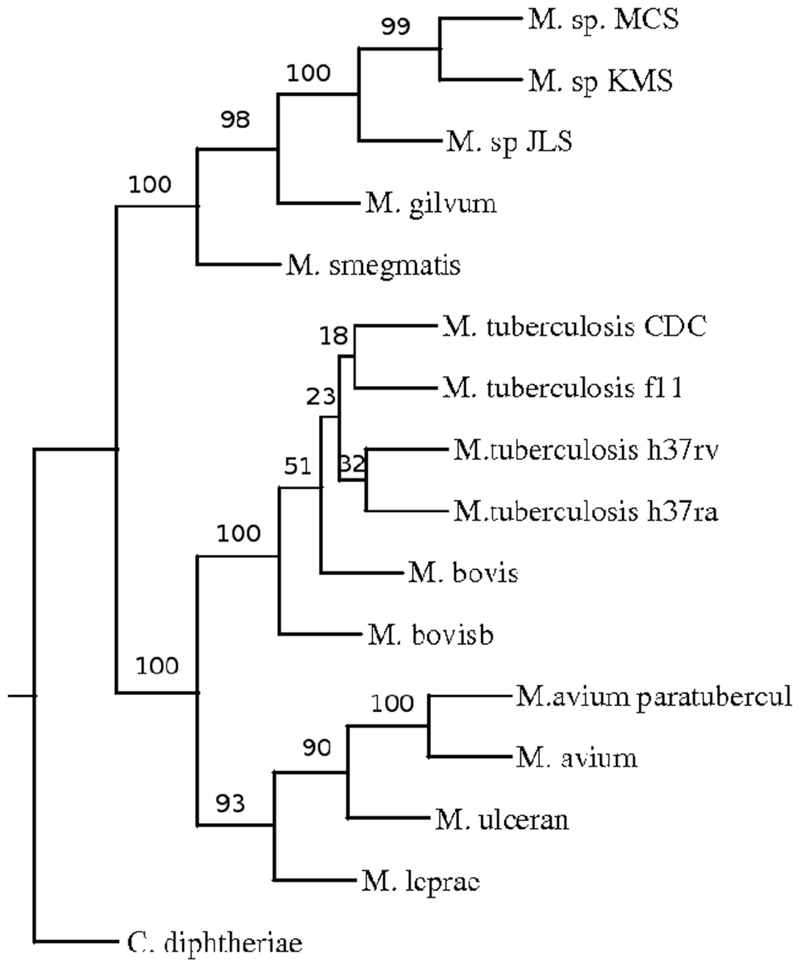
Analysis of the sequence of amplified 16S rRNA showed 99.9% identical between the M.avium ssp.avium (MAA) and M. Avium ssp.paratuberculosis (MAP) [21]. The relationship of different mycobacterial species can be better understood with a phylogenic tree based on 16S rRNA by Anchal Vishnoi, Rahul Roy et al. in Fig. (2) [22]. Apart from this, Anchal Vishnoi, Rahul Roy et al. had also presented the phylogenetic tree of the mycobacterial genome based on inter anchor distance as highlighted in Figs. (3) [22]. MAA and MAP have identical GRoES genes that differ from other mycobacterial species. This supports the current designation of Mycobacterium paratuberculosis as MAP [23]. Both MAA and MAP, intracellular pathogens, induce the secretion of Interleukin 10 (IL-10), low-level of tumor necrosis factor-alpha (TNFα) & Interleukin12 (IL-12), and downregulation of major histocompatibility complex (MHC) class I and class-II. Apoptosis with MAP was found slightly more than MAA, but the overall rate was found low with both subspecies. Both suppressed lipopolysaccharides (LPS)-induced macrophages apoptosis [24]. These all explains why both are kept under subspecies of Mycobacterium avium. (Figs. 2 and 3).
4.3. Mycobacterial Insertion Element IS900
Mycobacterial insertion element IS900 has been identified, and it would provide highly specific markers in the precise identification of MAP [25]. The insertion element IS900 has been used as the specific for MAP and has been used in diagnostic PCR. However, a copy of the sequence with 94% similar identity to IS900 has been isolated from Mycobacterium cookii. Thus alternative PCR system based on genes other than IS900 to confirm MAP is needed [26].
4.4. Modification in the Lab
MAP with a lab adapted strain k-10 and recent isolate from naturally infected cattle, strain 187, were undertaken Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) of cytosol and membrane, both showed marked differences in protein expression. This suggests that MAP might get modified in lab cultivation [27].

5. TRANSMISSION OF MAP
MAP in wildlife acts as potential reservoir and or vectors [28]. Calves get the infection through a contaminated environment. In utero transmission risk of MAP to fetuses was found to be 26.4% [29]. Rabbit can pass the MAP to calves or ruminants animals [30]. MAP was reported in feral cats from dairy farms infected with MAP [31].

6. ZOONOTIC SIGNIFICANCE OF MAP
Now, most believe that Crohn's disease is a syndrome caused by several agents. Mycobacterial theory & autoimmune theory has been proposed. But, both are complementary to each other. One deals with the aetiology of the disorder and second with its pathogenesis [6]. Major evidence that supports the link between MAP & Crohn's diseases is high detection rate of MAP in gut samples from Crohn's patient, serological response to MAP antigen from Crohn's patient, antibody & anti-MAP therapy resulting in improvement of Crohn's disease patient. However, there is a growing consensus that available evidence is insufficient to prove or disapprove MAP as a zoonotic agent [32]. Mycobacterial role in triggering autoimmune destruction of pancreatic cells in Type 1 Diabetes Mellitus might be due to high homology between Glutamic Acid Decarboxylase 65 K(GAD65)and MAP Heat shock protein 65 K (Hsp65) in antigenic peptide area [33]. MAP is postulated in the progression of HIV infection and other immune diseases like multiple sclerosis, sarcoidosis, Hashimoto's thyroiditis and Parkinson's disease [1]. The thermal resistance of MAP during pasteurization of milk led it as a potential foodborne pathogen. It is less susceptible to destruction by heat killing than most milk-borne zoonotic bacteria as Listeria & Mycobacterium bovis [34].
CONCLUSION
Initially, the MAP was supposed to affect domestic ruminants only. However, spilling of MAP to other ruminants & non-ruminants animal, even human, has grown its concern. MAP is very hardy, challenging to diagnose and had a severe economic effect in the ruminant industry, which makes a challenging need in its control. As MAP can transmit via faecal contaminated feed, water, milk & colostrum to any other species, Biosecurity & stringent sanitation could be one of the most cost-effective methods of control. In the coming days, experts need to do more applied research in understanding mycobacterial infection and its control methods.
ABBREVIATIONS
| MAP | = Mycobacterium avium ssp.paratuberculosis |
| MAA | = Mycobacterium avium ssp.avium |
| JD | = Johne's Disease |
| PCR | = Polymerase Chain Reaction |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIAL
The authors confirm that the data supporting the findings of this study are available within the article.
FUNDING
None.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial, or otherwise.
ACKNOWLEDGEMENTS
The author would like to thank reviewers for their constructive comments.


