All published articles of this journal are available on ScienceDirect.
Investigating the Efficiency of APRI, FIB-4, AAR and AARPRI as Noninvasive Markers for Predicting Hepatic Fibrosis in Chronic Hepatitis B Patients in Bangladesh
Abstract
Background and Aims:
Accurate, affordable non-invasive markers are highly needed for efficient diagnosis and management of liver fibrosis caused by chronic hepatitis B. This is the first study to investigate the diagnostic efficiency of Aspartate Transaminase to Platelet Ratio (APRI), Fibrosis Index (FIB-4), Aspartate transaminase to Alanine Transaminase Ratio (AAR) and AAR/Platelet ratio index (AARPRI) as non-invasive markers to predict hepatic fibrosis caused by Chronic Hepatitis B (CHB) in Bangladesh.
Methods:
In this study, a training cohort of 1041 CHB patients was recruited, whereas 104 and 109 CHB patients of matched ages were recruited as internal and external validation cohort groups respectively. Histological and hematological data were analyzed. METAVIR scoring system was used to classify liver fibrosis stages. Area Under Receiver Operating Curve (AUROC), correlations and cutoff values for the four diagnostic markers were calculated and assessed.
Results:
92%, 81% and 84% of the patients had liver fibrosis in the training cohort, internal and external cohort groups respectively. Among the four noninvasive panels, APRI showed the best area under ROC; (0.767, CI: 0.780-0.914; 0.775) for the training cohort, (0.775, CI: 0.693-0.857), and (0.847, CI: 0.780-0.914) for the internal and external cohorts respectively. Cut-off value of APRI was 0.512 with sensitivity/specificity of 84%/67% in training cohort, 81% / 66% in the internal cohort, and 88% / 66% in an external cohort. The odds ratio for APRI was 32.95 (95%CI: 4.746-228.862, p<0.001).
Conclusion:
Among all the four tested markers, APRI is the most accurate non-invasive test to predict major liver fibrosis (F2-3) in Bangladeshi CHB patients.
1. INTRODUCTION
Hepatitis B Virus (HBV) is the leading cause of liver diseases in Bangladesh as around 8 million people are reported to have the disease [1, 2]. The mortality of hepatitis B patients depends mainly on liver fibrosis, and hepatocellular carcinoma, however, in Bangladesh, HBV is estimated to cause 60% of liver cirrhosis and 65% of hepatocellular carcinoma cases [3]. Detecting and quantifying liver fibrosis stages are critical for better disease management and therapeutic monitoring of HBV patients. While liver biopsy is the main gold standard for assessing liver fibrosis, however, its high invasiveness, expensive cost, inconvenience for the patients together with the high risk of complications development are major limitations [4, 5]. (Two new references below). Consequently, noninvasive diagnostic blood and imaging markers are under continuous research to get promising screen tools for liver fibrosis in the affected patients. Several studies have proposed to use computer algorithm models for monitoring hepatic fibrosis [6-10]. Yet, they are of less use in clinical practice as they require sophisticated software programs [11, 12]. Meanwhile, multiple studies investigated the use of different hematological parameters as non-invasive models to help assess liver fibrosis [13]. Xiao et al. and Teshale et al. investigated using APRI and FIB-4 as noninvasive simple markers to monitor hepatic fibrosis in patients with hepatitis C [14, 15].
In 2015, The WHO encouraged to use the APRI score as a marker for assessing hepatic fibrosis in CHB [16]. However, its diagnostic performance and clinical applicability in patients care practice urgently need to be investigated among different populations with high variability, large cohorts and different fibrosis stages [17].
To the best of our knowledge, this is the first study to assess the diagnostic efficiency of Aspartate Transaminase to Platelet Ratio (APRI), Fibrosis Index (FIB-4), aspartate transaminase to Alanine Transaminase Ratio (AAR) and AAR/Platelet Ratio Index (AARPRI) as simple, affordable, noninvasive markers to predict liver fibrosis in CHB patients of Bangladeshi population.
2. METHODOLOGY
2.1. Study Design
From January 2017 to May 2018, this study was carried out in Dhaka Hospital, Bangladesh. All recruited patients were at that point beginning consistent follow-up visits in HBV out-patient center. Laboratory examinations and analytic liver biopsy were performed as a standard strategy for distinguishing proof of their fibrosis and consequent decision of antiviral treatment. Patients were chosen in view of biochemical examinations which were in accordance with biopsy results.
In this study, a total of 1254 CHB people were examined and confirmed by monitoring of HBsAg titer by ELISA technique and measuring of HBV DNA copies by using Smart Cycler II (HBV DNA PCR). 104 patients were grouped as an internal validation cohort. Also, to validate the overall performance of non-invasive biomarkers, we included 109 CHB patients as an external validation cohort from Rajshahi Medical Hospital, Bangladesh between September 2017 and April 2018.
Inclusion and exclusion criteria utilized were the same for all the recruited patients in the study.
2.2. Ethical Clearance
All recruited patients gave written consent for approval. The examination was performed in agreement with the latest version of Declaration of Helsinki for medical research and it was permitted by the Ethics Committee of Dhaka Medical College and Hospital and Rajshahi Medical College and Hospital, Bangladesh.
2.3. Inclusion Criteria
CHB was characterized as constant HBsAg positive for over a half year and it was confirmed by PCR. Naïve HBeAg-negative CHB patients were selected after assessing their liver biopsy results obtained from 2 principal histopathologists.
2.4. Exclusion Criteria
Patients who showed antagonistic activity to HBV infection were not included in the study in addition to the Immune tolerant patients characterized as HBeAg positive with relentless typical ALT levels.
2.5. Laboratory Tests
Hematological profiles of hepatitis B Patients including Hb, WBC, RBC, Red Distribution Width (RDW) and Platelet Count (PLT) were determined using the automated analyzer (ADVIA 2120i, Siemens Health care Diagnostics, Deerfield, IL). Biochemical parameters including ALT and AST levels were measured using ARCHITECT i2000SR (Abbott, IL). HBsAg was detected by ELISA and HBV DNA copy levels were measured using Smart Cycler II PCR equipment (USA, Detection limit: 300 IU/mL).
2.6. Liver Biopsy
Liver biopsy was executed using Ultrasound, Siemens Company, Germany. In this biopsy, a minimal of 1.6 cm of liver tissue was considered for diagnosis. Each specimen section was fixed, paraffin-embedded, and fixed in formalin with hematoxylin-eosin and reticular fiber staining. Histopathological examination was cross-checked by two pathologists.
Liver fibrosis stage was classified with METAVIR scoring system. Liver fibrosis stages are mentioned below:
F0: No Fibrosis.
F1: Portal Fibrosis without septa.
F2: Portal Fibrosis with few septa
F3: Numerous septa without cirrhosis.
F4: Cirrhosis.
2.7. Model Calculation
FIB-4, AAR, APRI, RPR, platelet count, were used to predict the liver fibrosis. The following formulas were used to calculate the investigated non-invasive markers.
FIB-4= (Age(Y) ×AST (U/L))/ (Platelet count (109/L) ×√ALT (U/L))
APRI= {AST (ULN)/Platelet count (109/L)} ᵡ100
AAR= (AST/ALT) ratio
AARPRI= AAR/ {Platelet count (109/L)/150}
| Features | Training (N=1041) |
External Cohort (N=109) |
p values | Internal Cohort (N=104) |
p values | |
|---|---|---|---|---|---|---|
| Male, n (%) | 831 (79.8) | 91 (83.5) | - | 81 (78.8) | - | |
| Age(Y) | 35.623 | 36.6513 | 0.707996 | 34.7885 | 0.307682 | |
| ALT(U/L) | 72.241 | 64.3578 | 0.663609 | 58.8365 | 0.890723 | |
| AST(U/L) | 71.042 | 62.2201 | 0.4885 | 58.6635 | 0.772197 | |
| HBV (IU/L) | 1.04837ᵡ107 | 1.250ᵡ107 | 0.766336 | 1388110 | 0.169515 | |
| HB(g/dl) | 13.996 | 14.0522 | 0.874116 | 14.0173 | 0.980537 | |
| WBC(10^3/µl) | 8.6665 | 8.66147 | 0.752923 | 8.48563 | 0.685867 | |
| PLT(10^3/µl) | 252.41 | 249.871 | 0.785595 | 256.807 | 0.619191 | |
| RBC(10^6/µl) | 4.8039 | 4.84358 | 0.71159 | 4.93374 | 0.349037 | |
| RDW (%) | 14.219 | 14.1339 | 0.955372 | 14.3529 | 0.155264 | |
| FIB-4 | 1.6141 | 1.64646 | 0.123258 | 1.25583 | 0.627637 | |
| APRI | .89545 | .768045 | 0.493051 | 0.73476 | 0.682005 | |
| AAR | .99662 | 1.01253 | 0.301893 | 0.94642 | 0.617333 | |
| AARPRI | .75893 | .809867 | 0.217583 | 0.68383 | 0.604218 | |
| Fibrosis, n (%) | ||||||
| F0 | 83(7.973103) | 18(16.5137) | - | 20(19.2307) | - | |
| F1 | 781(75.0240) | 77(70.6422) | - | 71(68.2692) | - | |
| F2 | 42(4.034582) | 3(2.752294) | - | 5(4.807692) | - | |
| F3 | 62(5.955812) | 5(4.587156) | - | 6(5.769231) | - | |
| F4 | 73(7.012488) | 6(5.504587) | - | 2(1.923077) | - | |
| Variables | Pearson Correlation, r | P value |
|---|---|---|
| Age | 0.313 | <0.001 |
| ALT(U/L) | 0.490 | <0.001 |
| AST(U/L) | 0.640 | <0.001 |
| HBV DNA(IU/L) | 0.066 | 0.115 |
| Hb(g/dl) | -0.242 | <0.001 |
| WBC(10^3/µl) | -0.255 | <0.001 |
| PLT(10^3/µl) | -0.448 | <0.001 |
| RDW (%) | 0.282 | <0.001 |
| RBC(10^6/µl) | -0.353 | <0.001 |
| FIB-4 | 0.764 | <0.001 |
| APRI | 0.850 | <0.001 |
| AAR | 0.397 | <0.001 |
| AARPRI | 0.628 | <0.001 |
2.8. Statistical Analysis
Normality tests were performed by Kolmogorov–Smirnov test. Mean ± standard deviation and percentages were used to represent continuous and categorical data. t-test and U-test were carried out for comparison of continuous parameters. The correlation between the four noninvasive fibrosis scores and METAVIR fibrosis scores was analyzed using Pearson’s test. The diagnostic ability of the four investigated fibrosis scores was assessed by ROC curve analysis and calculating the area under the curve using Z test. Cut off values were determined by Youden index (sensitivity + specificity − 1). All tests were 2-tailed and P value < 0.05 was considered statistically significant. Data analysis was performed using the SPSS measurable bundle, form 16.0 (SPSS, Chicago, IL, USA) or R, variant 3.0 (http://www.r-project.org/).
3. RESULTS
3.1. General Features of CHB Patients
General features of the recruited CHB patients are presented in Table 1. The results, as shown in Table 1 indicate that there were no significant differences of mean age, FIB-4, APRI, AAR, and AARPRI among the training (79.8% men), external cohort (83.5% men), and internal cohort (78.8% men). There were no significant differences between the training external and internal cohorts regarding fibrosis stages using METAVIR scoring.
3.2. Correlation of the APRI, FIB-4, AAR and AARPRI Scores with METAVIR Fibrosis Stages
The relationship between METAVIR fibrosis stages and the non-invasive fibrosis scores is shown in Table 2. The METAVIR scores were positively correlated with FIB-4 (r=0.764, p<0.001), AAR (r=0.397, p<0.001), and AARPRI (r=0.628, p<0.001).Interestingly, APRI score showed the highest correlation with METAVIR (r=0.850, p<0.001).
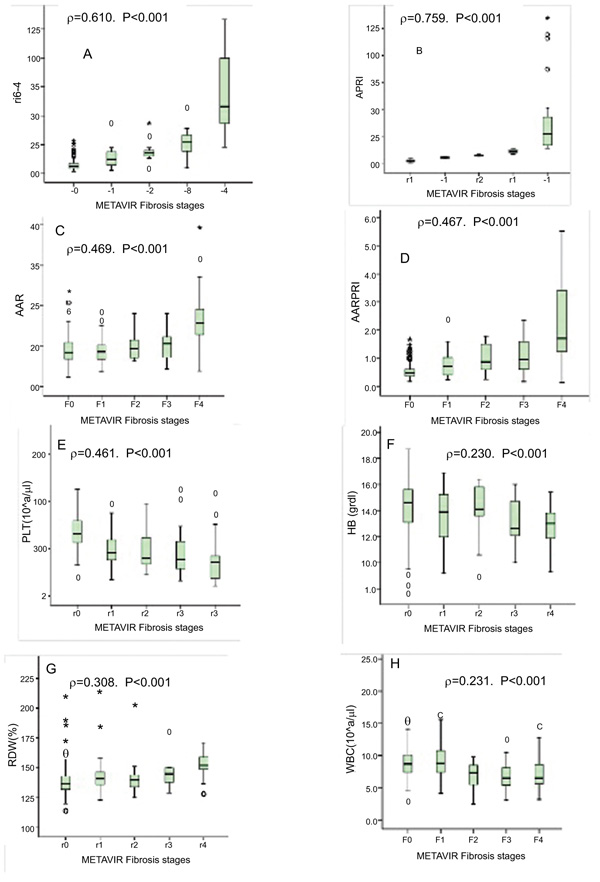
Fig. (1) shows in training cohort, the relationship between non-invasive markers and liver fibrosis stages of HBV people. The non-invasive markers of FIB-4(ρ=0.610,p<0.001) and APRI (ρ=0.759, p<0.001) score showed the strongest (positive) correlation with fibrosis stages according to METAVIR score.
3.3. Diagnostic Performances of FIB-4, APRI, AAR, AARPRI for Fibrosis
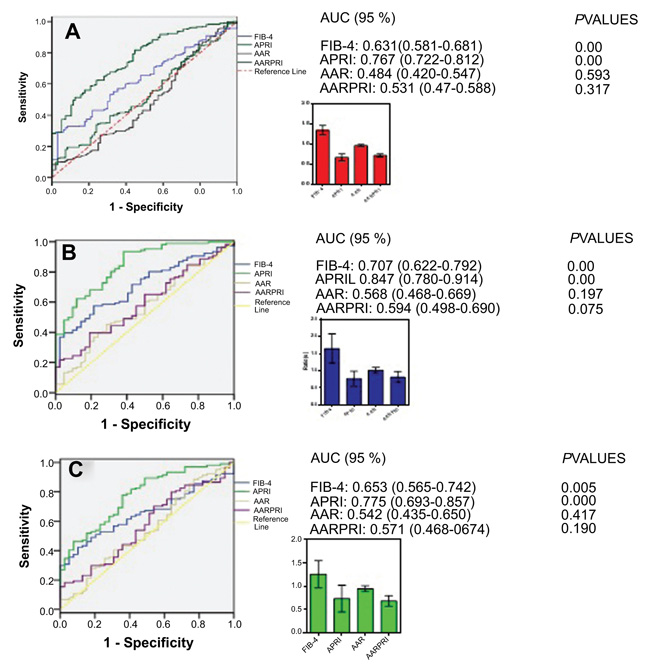
To assess the diagnostic performance of the blood biomarkers, we performed ROC analysis, results are shown in Fig. (2). In the training cohort Fig. (2A), the area under curve (AUC) of APRI was 0.767 (0.722-0.812), which was considerably higher than that of FIB-4 (AUC=0.631, 0.581-0.681, P<0.001) while those of AAR and AARPRI were (AUC=0.484, 0.420-0.547, P>0.05), AARPRI (AUC=0.531, 0.474-0.588, P>0.05) respectively. On the other hand, the external cohort Fig. (2B) and the internal cohort Fig. (2C) showed a similar array of results. Relevant to diagnostic performance, the AUC with 95% CI was 0.847 (0.780-0.914, P<0.001), 0.707 (0.622-0.792, P<0.001), 0.568 (0.468-0.669, P=0.197), 0.594 (0.498-0.690, P=0.075) for APRI, FIB-4, AAR, and AARPRI respectively in external cohort Fig. (2B). In internal cohort, the AUC (95% CI) for APRI, FIB-4, AAR, and AARPRI was 0.775 (0.693-0.857, P<0.001), 0.653 (0.565-0.742, P<0.001), 0.542 (0.435-0.650, P=0.417), and 0.571 (0.468-0.674, P=0.190) one-to-one Fig. (2C).
| - | FIB-4 | APRI | AAR | AARPRI |
|---|---|---|---|---|
| Training cohort | ||||
| Cut-off | 1.159 | 0.512 | 0.411 | 0.365 |
| Sensitivity | 83 | 84 | 63 | 83 |
| Specificity | 87 | 67 | 45 | 72 |
| External cohort | ||||
| Sensitivity | 82 | 88 | 72 | 81 |
| Specificity | 78 | 66 | 55 | 78 |
| Internal cohort | ||||
| Sensitivity | 86 | 81 | 66 | 88 |
| Specificity | 81 | 66 | 53 | 74 |
3.4. The Optimal Cut Off Values for FIB-4, APRI, AAR and AARPRI Scores
The optimal cut off values for the assessment of liver fibrosis in CHB are shown in Table 3. In the training cohort, an optimal FIB-4 cut-off value of 1.159 produced a sensitivity of 83%, a specificity of 87% while the optimum cut-off value of APRI was 0.512 with a sensitivity and specificity of 84% and 67% respectively. The optimum cut-off value of AAR and AARPRI was 0.411 and 0.365 respectively and the corresponding sensitivity and specificity were 63%, 45%, 83%, and 72% respectively.
4. DISCUSSION
Monitoring and quantifying liver fibrosis are critical for proper therapeutic intervention management in CHB. The major limitations combining liver biopsy procedure [18] and high expenses of fibro scan tests [18-21] urge the need for investigating more affordable and less complex noninvasive markers.
APRI and FIB-4 scores have been effectively used as noninvasive markers of fibrosis for hepatitis C patients [9, 14]. However, the exact diagnostic performance for APRI among other panels assessing liver fibrosis in CHB is not as settled as in hepatitis C, especially in large study groups.
The current study evaluated the diagnostic efficiency of FIB-4, APRI, AAR, and AARPRI for assessing liver fibrosis in a large cohort of the CHB patients of the Bangladeshi population. In this study, the diagnostic performances of AAR and AARPRI were comparable among the training and validation cohorts. Meanwhile, both APRI and FIB-4 scores were higher in CHB patients with significant fibrosis. This finding goes in line with previous small scale studies where APRI and FIB-4 scores were proved to more efficient in assessing huge fibrosis (F2-4) in CHB [18, 22].
FIB-4 scoring framework was more precise in patients with nil-to-direct fibrosis (FIB-4 score <1.45) versus incidences of more serious fibrosis. This finding agrees with previous reports, where Mallet et al., recommended using FIB-4 for the diagnosis of mild fibrosis and exclusion of significant fibrosis in CHB patients [9].
It is worth mentioning that, although increasing evidence highlights the prognostic value of individual CBC parameters to predict the clinical outcomes of other disorders [23, 24] to the best of our knowledge, our study is the first to report these correlations with liver fibrosis stage in the Bangladeshi population. We discovered that hematologic CBC parameters, namely hemoglobin and platelets, were independent predictors in the liver fibrosis stage CHB patients.
Interestingly in the current study, and concerning the AUROC, the overall diagnostic performance of APRI was significantly superior to that of FIB-4 in the training as well as the validation cohorts. In parallel with this finding, APRI was very recently found to be a more efficient diagnostic marker to assess advanced liver fibrosis in end-stage renal disease patients with CHB as compared to FIB-4 [25], where the research group suggested its use to decrease the need for liver biopsy in high-risk populations.
Additionally, in the current study, we assessed new cut-off values for APRI and FIB-4 particularly for CHB patients based on the performed ROC. The cutoff values proposed by the WHO guidelines have been derived from HCV studies [26]. We determined cut-off values of 0.512 and 1.159 for APRI and FIB-4 which had sensitivity/specificity of 84%/67% and 83%/87% respectively.
CONCLUSION
The current study included a large cohort group of CHB patients. Compared to FIB-4, AAR and AARPRI, APRI panel has been found to have the highest noninvasive diagnostic ability to assess liver fibrosis in CHB patients. The results of this study support the idea of the non-invasive markers, APRI is of supreme promising importance as an affordable, accessible tool to check fibrosis status of CHB in developing countries that can limit the need of liver biopsy.
Practically speaking, specialists using APRI should know about their impediments in HBV patients. Optimization of the diagnostic performance of APRI and FIB-4 tests can be further assessed by wider scale studies guaranteeing consistent sufficient liver tissue lengths for comparison to avoid any inaccuracy in evaluation.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Ethical approval was obtained from the review board of Dhaka Medical College and Hospital, and Rajshahi Medical College and Hospital, Bangladesh.
HUMAN AND ANIMAL RIGHTS
No Animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT OF PUBLICATION
Informed consent was obtained from all the participants.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


