All published articles of this journal are available on ScienceDirect.
Control of Early Blight of Tomato Caused by Alternaria Solani and Screening of Tomato Varieties against the Pathogen
Abstract
Introduction:
Early blight is a common disease of tomato, which is caused by Alternaria solani.
Objectives:
This work was accompanied to find an alternative to chemical fungicides and to screen tomato varieties against Alternaria solani.
Methods:
The infected leaves were collected from five tomato fields of Shere-e-Bangla Agricultural University, Dhaka and were cultured for the identification of the infectious fungus and The phytobiocidal role of six plants against Alternaria solani was evaluated in vitro model.
Results & Discussion:
Alternaria solani was identified as the infectious fungus. The growth of the test fungi Trichoderma spp. viz., Trichoderma viride, T. harzianum collected form NAMDEC and Trichoderma sp collected from field of BCSIR was monitored as optimum PH. All the selected Trichoderma spp. were antagonistic to A. solani. Antagonistic capacity of the Trichoderma spp. was tested by dual culture, volatile as well as non-volatile method. It was observed, T. viride was most effective in the reduction process of A. solani and T. harzianum. T. viride also showed highest inhibition in volatile and non-volatile trials. Six plant extracts viz., Adhatoda vasica (Nees), Azadirachta indica (A Juss). Ocimujm sanctum (L), Allium sativum (L), Datura metal (Linn) and Zingiber officinale (Rose) were selected to evaluate their in vitro efficacy of 5%, 10% and 20% concentration against the A. solani. Allium sativum was the most effective one against A. solani, followed by Azadirachta indica. The efficacy of five fungicides viz., Bavistin 50WP, Mancozeb 80WP, Indofil M-45, Sulcox 50WP and Tall 25EC were evaluated for their fungitoxicity against the A. solani at 100, 200,100, 600 and 800 ppm. Tall 25EC was the most effective fungicide against Alternaria solani followed by Mancozeb 80WP. After screening the five tomato varieties against A. solani, it was revealed that BARI Tomato-9 had the highest Percentage of Disease Index (PDI) and the leaf of BARI Tomato-7 had the lowest Percentage of Disease Index (PDI).
Conclusion:
The extract of Allium sativum was effective to control Alternaria solani at prescribed concentration. The highest PDI was found in BARI tomato-9 against Alternaria solani.
1. INTRODUCTION
Bangladeshi people earn their living from agriculture. Tomato is the most popular and the best vegetable in Bangladesh. It is a vegetable as well as fruit which succeeded popularity in the last century [1, 2]. Tomato is the primary ingredient to produce delicious food. It also includes a numbers of very tasty and nutritional food, such as tomato puree, ketchup, full peeled, juices, sauces as well as soups [3]. The main obstruction in our country in the production of tomato is a number of pathogens including fungi, virus as well as bacteria. Tomato Mosaic Virus (ToMV) the most important tomato virus follows by, Tomato Leaf Curl Virus (TLCV), Curly Top Virus (CTV), Tomato Yellows Top Virus (TYTV) and Cucumber Mosaic Virus (CMV). Theses viruses cause viral diseases which decrease plant vigor and reduce yield potential of the fruit to a considerable extent [4]. Tomato cultivation is also damaged by a number of other major factors after the primary rise till harvest. Among them, Aphids, flea beetles, leaf miners, and spider mites threaten young plant-bed tomatoes. In the field, flea beetles, aphids, leaf miners, stink bugs, and fruit worms cause minimal damage to the foliage. However, their fruit destruction as well as the expansion of the disease are some of the complications which can be very serious [5]. Among these type of diseases, early blight of tomato is one of the most significant and destructive diseases in the country. There are many types of diseases of tomato. Among them, the most common disease of tomato is early blight. The main reason of this disease is the necrotrophic fungus Alternaria solani Ellis and Martin is one of the very important foliar diseases of tomato. This disease is one of the most common tomato disease, which occurs nearly every season wherever tomatoes are grown, but it is quite eminent in the areas with bedew, and also depends on the rainfall and moisture ratio in the atmosphere. It affects leaves, fruits and stems and can be severely limiting the yield when susceptible cultivars are used and weather is favorable. Severe defoliation can occur and result in sunscald on the fruit [6]. Warm, humid (24-29°C/ 75-84°F) environmental conditions are conducive to infection. In the presence of free moisture and at an optimum of 28-30°C (82-86°F), conidia will germinate in approximately 40 min. Host colonization of early blight tomato is simplified by enzymes, facilitated by enzymes (cellulases, pectin methylgalacturonase) and alternaric acid damages the host cell wall, then destroys the host cell by a toxin as well as energize the pathogen taken from the nutrient of the dead cell [7]. Plant ruins as well as seed, and the fungus endure between crops as mycelia or conidia in soil [8]. Also, chlamydospores can distribute as survival composition [9, 10]. Therefore, the life cycle of A. solani includes, soil-, seed- as well as air-borne stages making the pathogen difficult to control by means of rotation and sanitation. The highest hosts of A. solani are solanaceous crops that include tomato, potato, eggplant as well as pepper [11, 12]. Early blight produces a wide range of symptoms at all stages of plant growth. It can cause damping-off, collar rot, stem cankers, leaf blight, and fruit rot. The classic symptoms occur on the leaves where circular lesions up to 1/2″ in diameter are produced. Within these lesions dark, concentric circles can be seen. The leaf blight phase usually begins on the lower, older leaves and progresses up the plant. Infected leaves eventually wither, die, and fall from the plant [13]. Seedlings infected by damping off rarely survive to produce a vigorous plant. Quite often a large section or the entire tray of seedlings is destroyed once the plants have mature leaves and a well-developed root system, they are able to naturally resist the fungus or mold that causes damping off. There is a critical period of growth between planting and maturity when special care needs to be taken to protect sensitive seedlings. A wide variety of vegetables and flowers can be affected by damping off. Young leaves, roots and stems of newly emerged seedlings are highly susceptible to infection. Under certain environmental conditions, damping off pathogens can cause root/crown rot in mature plants [14]. Trichoderma grows closely in all kinds of temperate as well as torrid soils, cultivation depends on many types of soil, for example, agriculture, forest, prairie, salt marsh and desert soils in all climatic zones. This Trichoderma is also found in colonizing roots, litter, decaying/decorticated wood, decaying bark as well as several plant materials at all climatic zones/latitudes. For example, Trichoderma spp. are ubiquitous ascomycetous fungi found commonly in agriculture, grassland, forest, saline, and desert soils, and are particularly dominant in the humic layer of hardwood forests where they represent up to 3% of total fungal propagules in forest soils and 1.5% in pasture soils in a wide range of crops [15-17]. Trichoderma separates were assessed counter to Lasiodiplodia theobromae by dual culture procedure. A 5 mm diameter mycelial disc from the margin of the 7 days-old culture of Trichoderma separates as well as the Lasiodiplodia theobromae were placed on the PDA media opposite to the plate at equal distance from the periphery [18]. In this study, we aimed to assess the antifungal activity of six plants against A. solani in vitro and to find an alternative to chemical fungicides and to screen tomato varieties against pathogens.
2. MATERIALS AND METHODS
2.1. Isolation of Fungus
Infected leaves (4X4 nm) were cut with a sterilized scalpel from the diseased leaves and preserved in a sterile Petri plate. The inocula were washed using sterilized distilled water. Then the surface was sterilized by dipping in 10% Chlorox solution for one minute. The inocula were then further washed with sterile water. The inocula were dried between two layers of sterilized filter paper to remove the excess water. Finally, the inocula were placed on plain agar medium in Petri plates. A total of 50 inocula were placed on 17 sterilized Petri plates containing PDA medium under laminar flow cabinet. Each Petri plate contained 20 mL of PDA medium with an addition of tetracycline HCl, used to check the bacterial growth and incubated at room temperature (25○C) for 7 days. The reading of the fungal colonies took 7 days of inoculation. This process was continued for two weeks depending on the medium.
2.2. Purification and Identification of the Fungus
The slide of agar block had to be sterilized because of microbial contamination from air, glassware, hands, etc. The sterilization process was 100% effective and guaranteed that the medium stay sterile unless exposed to contaminants by less than adequate aseptic technique and exposure to air. At first, a small block of solid agar medium was taken on the tip of sterile inoculating needle and touched to the surface of the culture gently to adhere to spores on the agar block. Then the slide of agar block was placed on a PDA plate (used 5-10 ml of medium). The spores were dispersed on the medium. The Petri dish was put under a stereoscopic microscope and collected the well separated spores by means of a needle containing a tiny piece of sterile dry filter paper. The filter paper was transferred to a PDA slant. The spores were absorbed on the filter paper and germinated on the medium. Fresh filter paper were used for each spore. Stock cultures were maintained on PDA slant and stored at 5 - 10○C in a refrigerator for future studies. Cultures were maintained by sub-culturing after four weeks of intervals.
2.3. Assessment of Pathogenicity of the Identified Fungus
Healthy matured leaves of tomato were thoroughly washed under running tap water and then disinfected in 10% Chlorox for one minute. Excessive Chlorox was removed by placing the leaves on two layers of sterile autoclaved filter paper on Petri plate. The leaves were inoculated with 5 mm actively growing mycelial block of test fungus and were incubated for 7 days. In control plates, 5 mm agar blocks were inocualed on the leaves. The fungus was tested to find out their pathogenic potentiality. Three replications were maintained in the fungus test and used as follows: T1 = Control (Unpricked), T2 = control (Pricked). T3= Unpricked dorsally inoculated leaves, T4= Pricked dorsally inoculated leaves T5= Unpricked ventrally inoculated leaves, T6= Pricked ventrally inoculated leaves. All Petri plates were incubated at 25○C for 5 days
2.4. Isolation and Identification of Trichoderma spp.
At first, 1 gm. soil was added with 99 mL of distilled water in a conical flask and mixed very well with a glass rod and marked as the mother suspension. Then five test tubes each containing 9 mL of distilled water were taken. One mL of mother suspension was added into the 1st test tube and made it 10 mL So, the first test tube in the mother suspension was diluted 10 times. After proper mixing, 1 mL of suspension from the 1st test tube was added to the 2nd test tube and made it 10 mL So, into the 2nd test tube the mother suspension was diluted 100 times. This process was performed for the rest of the test tubes and diluted the mother suspension for 10, 100, 1000, 10000 and 100000 times. For each dilution, 1mL of suspension was poured into a sterilized Petri plate and then about 20 mL of sterilized melted PDA medium (about 45○C) was added. The plate was moved in different directions gently on the laminar air flow cabinet to get a homogenous distribution of the suspension. Three replications were maintained for each dilution. All the Petri plates were incubated at 25○C. After three days of incubation, individual fungal colonies belonging to different genera including Trichoderma sp. were sub-cultured on PDA slants from the culture plates and the cultured slants were incubated at 25○C for 7 days and stored at 100.5○C refrigerator for future studies. Cultures were maintained by sub-culturing after four weeks of intervals. Identities of the soil fungus were determined following the standard literatures [19-21].
2.5. Dual Culture
Trichoderma separates were valued counter to test fungus by dual culture technique as described [22, 23]. A 5 mm diameter mycelial disc from the margin of the seven days old culture of Trichoderma separates as well as the test pathogen were placed on the PDA media at against of the plate at equivalent space from the periphery. In control plates, a sterile agar disc was placed at the centre of the plates. All the plates were incubated at room temperature (25○C). Data was collected on radial growth and inhibition of growth. The colony growth of the pathogen was measured at both sides. The radial growth was recorded after 7 days of incubation when the fungus in control plates covered the plates completely. The percentage Inhibition (PI) of the fungus over the control was calculated using the formula [24].
% inhibition C-T/C x 100
2.6. Effect of Volatile Substances of the Trichoderma on the Growth of the Test Fungus
Five mm diameter mycelial disc of 7 days old culture obtained from the margin of each the Trichoderma spp. isolates was placed at the centre of the PDA plates and was incubated in 25○C for 48h. In control plates, a 5 mm diameter of sterile PDA medium was placed in the Petri plate as performed above. Then A. solani was inoculated at the centre of PDA plates with 5 mm diameter mycelial plug and Trichoderma inoculated plates were inverted on the top of the test pathogen plates and held together by an adhesive tape. Three plates for each of the Trichoderma spp. were used. These sets were incubated at 25○C and after 7 days, colony diameter of the A. solani, in all sets was measured and the percentage inhibition in the colony diameter of the A. solani was calculated.
2.7. Effect of Culture Filtrates (Non-volatile Metabolites) of the Trichoderma spp . on the Growth of the Test Fungus
Three equal sized blocks of each Trichoderma spp. isolates were cut from the actively growing margins of 5 days old cultures and inoculated separately into 250 mL conical flasks containing 100 mL sterilized potato dextrose broth medium. After 10 days of incubation at 25○C the static cultures were filtered firstly through Whatman filter paper No.1 and finally through Seitz filter by vacuum filter to obtain cell free culture filtrates. Twenty mL metabolite of each fungus was added in 80 mL sterilized PDA medium separately and Twenty mL of supplemented medium was poured in a sterilized Petri plate and was allowed to solidify. Each Petri plate was inoculated centrally with a 5 mm agar disc, cut from the margin of actively growing culture of an A. solani. In the control set, Petri plate containing PDA medium without culture filtrates were injected with A. solani as mentioned above. In control set, an equal amount of sterilized water was added with the PDA medium instead of culture filtrate. Three replications of each treatment were maintained. All Petri plates were incubated at 25○C. The radial growth of the colonies was measured after 7 days of incubation. The percentage inhibition of A. solani was calculated.
2.8. In vitro Effect of Plant Extracts on the Growth of the Test Pathogen
Six plant parts were selected for evaluating their effect on the vegetative growth of Alternaria solani, the causal agents of early blight of tomato. Particulars of all these parts of the plants are given in Table 1.
2.9. Preparation of Aqueous Plant Extracts
The desired parts of each plant were thoroughly washed in tap water, air dried and used for fresh extract preparation. In case of leaves, bulbs, fruits and rhizomes, extracts were prepared by crushing known weight of fresh materials with distilled water at ratio of 1:1 (w/v). The pulverized mass of a plant part was squeezed through four folds of the fine cloth and the extracts were centrifuged at 3000 rpm for 20 minutes. The supernatants were filtered through Whatman filter paper and the filtrate was collected in 250 mL Erlenmeyer flasks. In the method, the requisite amount of the filtrate of each plant extract was mixed with a PDA medium to get 5, 10 and 20% concentrations and was sterilized in an autoclave at 121○C under 1kg pressure for 15 minutes.
2.10. Inoculation of the Test Pathogen
The medium was poured into sterilized Petri plates and was allowed to solidify. Each Petri plate was inoculated centrally with a 5 mm agar disc cut from the margin of actively growing culture of the test pathogen. In the control set, a Petri plate containing PDA medium with the requisite amount of distilled water instead of a plant extract was also inoculated with agar disc of the test pathogen in the same way as described above. Three replications were maintained for each experiment. The inoculated Petri plates were incubated at 25○C. The radial growth of the colonies was measured after 7 days of incubation.
2.11. Fungitoxicity of Fungicides Against A. solani
Five fungicides with different active ingredient(s) viz. Bavistin 50 WP, Mancozeb 80 WP, Indofil M-45, Salcox 50 WP and Tall 25 EC were collected from Bangladesh Agricultural Research Institute (BARI), Gazipur Table 2. First, the fungitoxicity in vitro of five fungicides at different concentrations was evaluated against the A. solani. For each fungicide, a stock solution having the concentration of 10,000 ppm was prepared. The calculated amount of the stock solution of a fungificide was supplemented with a sterilized PDA medium to get the concentration of 100, 200 and 400 ppm. Twenty mL of the supplemented medium of a particular concentration was poured in a sterilized Petri plate and was allowed to solidify. In the control set, the required amount of sterile water instead of fungicide solution was added to the PDA medium. Then it was inoculated at the center of the plate with a 5 mm mycelial agar disc cut from the margin of actively growing culture of A. solani. Three replications were maintained in each case. The inoculated plates were incubated at 25○C. The radial growth of the colonies was measured at 5 days of incubation.
2.12. Methods of Sterilization and Incubation
Five different varieties of tomato leaves were washed with sterile water and surface were sterilized by dipping in 10% Chlorox solution for 1 minute. The leaves were washed with sterile water to remove chemicals. Finally, the leaves were placed inside the folds of sterile blotting paper to remove excess water. The three leaves of each tomato variety were separately inoculated on 9 cm Petri plates. A 5 mm diameter mycelial disc was cut off by sterile cork borer from the margin of the 5 days old culture of test fungus. A 5 mm mycelial block was inoculated on each leaf. Three plates for each treatment were maintained. In the control plates, leaves of each variety were inoculated only with sterilized PDA disc. All Petri plates were incubated at (25○C) for 7 days.
| SI. No. | Plant Species | Native Name | Family | Plant Parts Used |
|---|---|---|---|---|
| 1. | Adhatoda vasica Nees | Basak | Acanthaceae | Leaf |
| 2. | Azadirachta indica A Juss | Neem | Meliaceae | Leaf |
| 3. | Ocimum sanctum L. | Tulsi | Labiatae | Leaf |
| 4. | Allium sativum L. | Rasun | Liliaceae | Bulb |
| 5. | Datura metal Lin. | Datura | Solanaceae | Fruit |
| 6. | Zingiber officinale Rosc. | Ada | Zingiberaceae | Rhizome |
| SI.No. | Fungicides | Active Ingredient(s) | Manufacturer |
|---|---|---|---|
| 1. | Bavistin 50 WP | 50% Carbendazim (methyl benzibidazal 1-2 yl carbamate) | BASF, Germany |
| 2. | Mancozeb 80 WP | Zinc ion co-ordination product with managanese ethylene-1, 2-bisdithio carbamate polymer | Sundat S., Singaur |
| 3. | Indofil M-45 | 80% Mancozeb (Zinc-ion co-ordination product with manganese ethylene bisdithio carbamate polymer) | Mumbai, India |
| 4. | Salcox 50 WP | Copper oxycholoride | Chemiski production gesellschaft, German |
| 5. | Tall 25 EC | Propiconazole | Sarda worldwide exports private limited, India |
| Scale | Description |
|---|---|
| 0 | No symptom spot on the leaf |
| 1 | 1-20% leaf area infected and covered by spot |
| 2 | 21-40% leaf area infected and covered by spot |
| 3 | 41-60% leaf area infected and covered by spot |
| 4 | 61-80% leaf area infected and covered by spot |
| 5 | 80% leaf area infected and covered by spot |
| Features | Description |
|---|---|
| Colony | Initially greenish and becoming black with age |
| Mycelia | Smooth mycelial growth with circular and irregular margin |
| Conidiophore | Singly or in groups, straight or flexuous brown to olivaceous brown |
| Conidia | Solitary straight or slightly flexuous oblong or muriform or ellipsoidal tapering to beak, pale or olivaceous brown, in the broadest part with 5-9 transverse and 0 or a few longitudinal septa. |
| Beaks | Flexous, pale and sometimes blanched |
2.13. Data Collection
The degree of leaf infection was studied by visual observation, based on the extent of development of lesion of the leaves up to 7 days after inoculation. The Percent Disease Index (PDI) of leaf infection was rated following 0 to 5 scales Table (3).
3. RESULTS AND DISCUSSIONS
3.1. Isolation, Purification and Identification of the Fungus
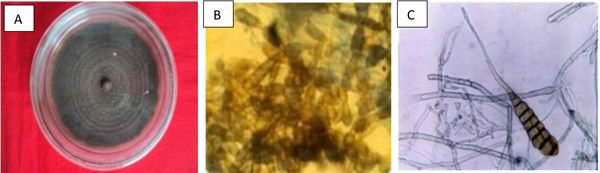
Standard tissue isolation technique was used to obtain Alternaria solani from the tomato leaves with early blight symptoms. Isolation was repeated several times to obtain pure culture. Culture and microscopic characteristic of A. solani are presented in Table 4 and Fig. (1).
In this investigation, Alternaria solani growth on PDA medium was greenish black, circular and smooth [25]. The conidiophores arose singly or in small groups, usually simple, straight or flexuous, almost cylindrical, septate, rather pale brown or yellowish brown, smooth while the conidia were solitary straight or slightly flexuous oblong or muriform. The broadest part had 5-9 transverse and 0-4 longitudinal septa [26-28]. A. solani exhibited the same characteristics.
3.2. Pathogenicity Test
The results revealed that A. solani was capable of producing symptoms on tomato leaves (Fig. 2). In the present study, unpricked leaflet (T1, control) and pricked leaflet (T2, control) did not show any symptoms because, only agar blocks were placed on T1 and T2 plate. In the treatments, leaflets (T3, T4, T5 and T6) showed symptoms for early blight. The microscopic observation further confirms the presence of A. solani.
3.3. Effect of pH on the growth of Alternaria solani
Mycelium growth of A. solani was observed within a range of pH level (pH 5.5 to 10.0). A highly significant difference in the growth of A. solani was obtained at different pH levels Table 5. Maximum growth (41.0 mm) was recorded at pH 8.0 followed by 7.5, 6.5, 7.0, 6.0, 9.0, 9.5, 5.5 and least growth was obtained (26.5) at pH 10.0. The optimum pH for the growth of A. solani was found to be in the range of 6.5-8.0.
So, A. solani showed the highest growth at pH 8.0 that means alkaline soil is suitable for its growth. It was observed that the growth of A. solani decreased after pH 8.0.
3.4. Cultural and Morphological Characteristics
Three Trichoderma isolates grown in PDA media showed variation in cultural characteristics as mentioned in Table 6. The isolates showed differences in their mycelia features and colony characters. Mycelia of all Trichoderma spp. were hyaline. The colony colour of Trichoderma spp. turned to green.
During the present investigation, various distinct symptoms were recorded after the observation of mature colony of Trichoderma spp. on PDA. Trichoderma produced grass with green and yellow pigmentation [29, 30].
T. harzianum was characterized according to [31]. Other species of Trichoderma in the present study was characterized as described [32].
3.5. Effect of Dual Culture, Volatile and Non-Volatile Metabolites of Three Trichoderma Isolates on Alternaria solani
The results of the effect of dual culture, volatile and non-volatile metabolites of the Trichoderma spp. on the growth of A. solani are shown in Table 7. The maximum inhibition of radial growth of A. solani by dual was observed in case of T. viride (67.77%) followed by Trichoderma sp (65.00%) after 7th days of incubation at 25. The least inhibition was found in the case of T. harzianum (64.44%). The maximum inhibition of radial growth of the A. solani was observed with the volatiles metabolites of T. viride (69.44%) followed by T. harzianum (61.11%). Trichoderma sp. showed the least effect in the growth of the pathogen (58.72%). The present study confirmed that the volatile metabolites had a fungistatic property. Volatile metabolites secreted by Trichoderma spp. showed significant in controlling A. solani. The maximum inhibition of radial growth of the same pathogen by culture filtrates was observed in case of T. viride (90%) followed by T. harizianum (87.22%). Least inhibition was noted in case of Trichoderma spp. (86.11%). In the present study, the percentage of inhibition of A. solani due to non-volatile metabolites of Trichoderma spp. ranged from 90-86.11%, where T. viride showed maximum inhibition. The findings supported Fusarium oxysporum, and reported the inhibitory effects of Trichoderma sp. the results are quite similar to Colletotrichum capsici finding which reported that T. viride was the most effective one in case of both non-volatile metabolites and dual culture tests, whereas, T. harzianum showed a maximum inhibition [24].

| pH | Radial growth (mm) |
|---|---|
| 5.5 | 29.5 |
| 6.0 | 33.2 |
| 6.5 | 35.0 |
| 7.0 | 34.5 |
| 7.5 | 36.3 |
| 8 | 41.0 |
| 8.5 | 28.5 |
| 9.0 | 32.5 |
| 9.5 | 30.0 |
| 10.0 | 26.5 |
3.6. In vitro Effect of Plant Extracts on the Growth of the Alternaria solani
Result of plant extracts on the radial growth of A. solani is presented in Table 8. Data in Table 8 showed that plant extracts had varied degree of growth inhibition of the A. solani at different concentration. Out of six plants extracts, Allium sativum showed 74.07% radial growth inhibition of the test pathogen at 20% concentration which was followed by Zingiber officinale (70.05%), Adhatoda vasica (64.50%), Ocimum sanctum (58.67%), Azadirachta indica (36.50%).
The least inhibition was noted in case of Datura metal (29.50%). The order of effectiveness against A. solani at 10% concentration was Allium sativum (55.18%), Zingiber officinale (53.10%), Azadirachta indica (34.80%), Adhatoda vasica (33.50%), Datura metal (27.2%), Ocimum sanctum (26.99%). The order of effectiveness against test fungus at 5% concentration was Allium sativum (35.0%), Azadirachta indica (32.50%) Zingiber officinale (31.85%), Adhatoda vasica (30.11%), Ocimum sanctum (28.45%)> Datura metal (25.11%). So, it was showed that the inhibition of the A. solani increases with the increase of the concentration of the plant extracts in culture. It is also cleared that Allium sativum extract has the most effective inhibition in the radial growth of A. solani (74.07%). This result is similar to A. solani; Rhizoctonia sloani; Alternaria > spp. Alternaria carthami
[33-38].
3.7. Fungitoxicity Against the Alternaria solani
Table 9 showed that the increasing concentrations tested of fungicides decreased the fungus radial growth. The maximum inhibition of the radial growth of A. solani by fungicides at 100 ppm was observed in case of Tall EC (100%) followed by Moncozeb 80 WP (78.57%), Indofil M-45 (62.09%) and Salcox 50 WP (55.64%). The least inhibition was found in case of Bavistin 50 WP (53.96%). The maximum inhibition of radial growth of A. solani by fungicides at 200ppm was observed by Tall 25 EC (100%), followed by Mancozeb 80 WP (80.95%), Indofil M-45 (70.16%) and Salcox 50 WP (61.29%). Least inhibition was noted in case of Bavistin 50 WP (57.14%). The maximum inhibition of radial growth of the same pathogen at 400 ppm was observed in case of Tall 25 EC (100%) followed by Mancazeb 80 WP (85.71%), Indofil M-45 (79.03%) and Salcox 50 WP (68.54%). Bavistin 50 WP showed the least effect when checked to the growth of the A. solani (60.31%). The result in Table 9 also indicates that the reduction percentage of radial growth was increased by increasing the concentrations of fungicides tested. Tall 25 EC showed full inhibition of A. solani even at the low concentrations of 100 ppm. On the other hand, Mancozeb 80 WP also inhibited A. solani by 85.11% at 400 ppm concentration [39-44].
| Cultural Characters | T. harzianum | T. viride | Trichoderma spp |
|---|---|---|---|
| Mycelium | Hyaline | Mycelium not visibe to at the naked eye | Hyaline |
| Colony colour | White and scattered greenish patches became visible as the conidia formed | Dark green | Grass green |
| Colony propagule | Luxuriant with regular cottony growth | First growing conidia spreads vigorously | First and vigorous growth |
| Pigmentation | No | No | Yellow pigmentation observed |
| Isolates Trichoderma | Percent of Growth Inhibition of Alternaria solani | ||
|---|---|---|---|
| Dual Culture | Volatile | Non-Volatile | |
| T. viride | 67.77 | 69.44 | 90 |
| T. harzianum | 64.44 | 61.11 | 87.22 |
| Trichoderma sp | 65.00 | 58.72 | 86.11 |
| Plant | Percent Growth Inhibition at Different Concentrations | ||
|---|---|---|---|
| 5% | 10% | 20% | |
| Adhatoda vasica | 30.11 | 33.50 | 64.50 |
| Azadirachta indica | 32.50 | 34.80 | 36.50 |
| Ocimum sanctum | 28.45 | 26.99 | 58.70 |
| Allium sativum | 35.0 | 55.18 | 74.07 |
| Datura metal | 25.11 | 27.20 | 29.50 |
| Zingiber officinale | 31.85 | 53.70 | 70.05 |
| Fungicides | Percent growth inhibition of A. solani at different concentration (ppm) | ||
|---|---|---|---|
| 100 | 200 | 400 | |
| Bavistin 50 WP | 53.96 | 57.14 | 60.31 |
| Mancozeb 80 WP | 78.57 | 80.95 | 85.11 |
| Indofil M-45 | 62.09 | 70.16 | 79.03 |
| Salcox 50 WP | 55.64 | 61.29 | 68.54 |
| Tall 25 EC | 100 | 100 | 100 |
| Tomato Varieties | Range of PDI | PDI |
|---|---|---|
| BARI Tomato -2 | 30-60% | 55.55% |
| BARI Tomato -3 | 25-50% | 40.55% |
| BARI Tomato -7 | 5-40% | 31.11% |
| BARI Tomato -9 | 40-60% | 57.77% |
| BARI Tomato -15 | 20-40% | 35.55% |
3.8. Screening of Tomato Varieties Against the Alternaria solani.
The PDI value indicates the highest percentage of disease incidence (57.77%) was recorded in BARI Tomato-9, followed by BARI Tomato-2 (55.55%), BARI Tomato-3 (40.55%), BARI Tomato-15 (35.55%) and BARI Tomato-7 (31.11%). In every case, disease severity was moderate. Results are shown in Table 10.
In vitro screening experiment indicated that none of the tested tomato varieties was resistant to A. solani. Although, lowest PDI was observed in BARI Tomato-7 with the PDI range 5-40% and average PDI 31.11%. The findings were partially supported by observing twenty four diseases of vegetable and fruit crops in the Chittagong region and the highest incidence of leaf blight disease was (43%) as recorded from the early blight of tomato. These findings also supported the study [45] that researched on fifteen tomato advanced lines including Manik and BARI Tomato-10 which were evaluated on the basis of late blight and early blight. They found the highest disease incidence of early blight of tomato at V-259 and the lowest disease incidence at V-215. On the basis of early blight disease intensity, none was found to be highly resistant or immune against the disease.
CONCLUSION
The purpose of this study was to screen tomato varieties against pathogens and in vitro control of early blight of tomato. The isolated pathogen was identified as Alternaria solani. Growth of A. solani was tested at ten different levels of pH and the maximum radial growth was observed at a pH range of 6.5-8.0 and least at pH 10. The antagonistic capacity of the Trichoderma spp. was tested by dual culture method. The finding showed that all the Trichoderma spp. reduced radial growth of A. solani. Trichoderma viride was the most effective one on decreasing the radial growth of A. solani and the effects of volatile and non-volatile metabolites of selected Trichoderma spp . against the tested fungi. Among five fungicides evaluated in the in vitro conditions against A. solani, Tall 25EC gave the highest inhibition of the radial. Among the plant extracts, Allium sativum was the most effective plant extract to decrease radial growth of test pathogen followed by Zingiber officiniale, while Datura metal was the least effective. All the five selected fungicides reduced the radial growth of A. solani. Tall 25 EC was the most effective one on inhibition of the radial growth of A. solani followed by Mancozeb 80 WP, while Bavistin 50 WP was the lowest effective one. We have revealed the highest percentage of disease index in BARI Tomato-9 followed by BARI Tomato-7. None of the tested varieties were found to be immune to the test pathogen. The comparision between bio-control and chemical control showed that bio-control agents is the best solution for controlling the early blight disease of tomato.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


