All published articles of this journal are available on ScienceDirect.
Assessment of the Antimicrobial Activity of Few Saudi Arabian Snake Venoms
Abstract
Background
Venoms of two cobras, four vipers, a standard antibiotic and an antimycotic, were evaluated comparatively, as antimicrobials.
Methods:
Six venom concentrations and three of the standard antibiotic and the antimycotic were run in micro-dilution and diffusion plates against the microorganisms.
RESULTS:
Echis pyramidum, Echis coloratus and Cerastes cerastes gasperettii highest venom concentrations gave significant growth inhibition zones (GIZ) with respect to a negative control, except Bitis arietans, whose concentrations were significant. The cobra Walterinnesia aegyptia had significant venom concentrations more than Naja haje arabica. The Staphylococcus aureus Methicillin Resistant (MRSA) bacterium was the most susceptible, with a highly (P < 0.001) significant GIZ mean difference followed by the Gram positive Staphylococcus aureus, (P < 0.001), Escherichia coli (P < 0.001), Enterococcus faecalis (P < 0.001) and Pseudomonas aeruginosa which, had the least significance (P < 0.05). The fungus Candida albicans was resistant to both viper and cobra venoms (P > 0.05). The antibiotic Vancomycin was more effective than snake venoms though, they were more efficient in inhibiting growth of the resistant Pseudomonas aeruginosa. This antibiotic was also inactive against the fungus, whilst its specific antifungal Fungizone was highly efficient with no antibacterial activity.
Conclusions:
These findings showed that snake venoms had antibacterial activity comparable to antibiotics, with a directly proportional relationship of venom concentration and GIZ, though, they were more efficient in combatting resistant types of bacteria. Both venoms and the standard antibiotic, showed no antifungal benefits.
INTRODUCTION
Bacterial infections involving the multidrug resistant strains are among the top leading causes of death throughout the world. Healthcare system across the globe has been suffering from an extra-ordinary burden in terms of looking for the new and more potent antimicrobial compounds [1]. The majority of bacteria such as Pseudomonas, Klebsiella, Enterobacter, Acinetobacter, Salmonella, Staphylococcus, Methicillin Resistant S. aureus (MRSA), Enterococcus and penicillin-resistant Streptococcus pneumoniae (PRSP) vancomycin-resistant enterococci have developed several ways to resist antibiotics. Such bacteria are becoming a serious clinical problem throughout the world [2-6].
Natural products are important sources of medicinal compounds. A wide variety of organisms produce such bioactive compounds and some of these natural substances have been shown to be able to kill bacteria [6-10]. Snake venoms contain a great variety of biologically active proteins responsible for various pathological effects. Venoms include toxins which are highly potent compounds with selective and specific activities. They can be useful and valuable as pharmacological tools in drug research, as potential drug design templates and as therapeutic agents [11, 12]. In recent years, venoms and venom components from different venomous animals have shown potential antibacterial activity. This includes snake [13-15] and scorpion venoms [16-19].
To date, only a few studies have been made on the antimicrobial activities of snake venoms [9]. In 1948, Glaser investigated antibacterial activity of Crotalus venom [20] and then in 1968 Aloof Hirsch and his colleagues reported an antibacterial lytic factor from the venom of the cobra Hemachatus haemachatus [21]. Now it is fully documented that the snake venoms have a number of cytotoxic factors along with potent killing effect on bacteria as well as viruses [22-25]. Rideiro et al., [15] reported the presence of L-amino acid oxidase present in snake venom and displayed many important biological properties that included the bactericidal and virucidal activities. A new antibiotic vejovine has been discovered from the Vaejovis mexicanus scorpion venom and this agent has been proved to be highly effective on pathogenic bacteria [19]. Captopril, anti-hypertensive drug was designed based on the peptide inhibitor of angiotensin-converting enzyme from the venom of Bothrops jararaca snakes [26]. Echistatin and Ecarin are two bioactive molecules, isolated from Echis carinatus snake venom. Echistatin is one of the most potent disintegrin polypeptide which has platelet aggregation inhibitor activity and used as an anticoagulant while Ecarin is an enzyme used in the ecarin clotting time (ECT) test to monitor anticoagulation during treatment with hirudin [27-30].
In a recent study it was demonstrated that the venom from one of the world's most venomous snakes Mamba could be the key to a new breed of painkillers, may be more potent then morphine [31]. Several naturally occurring peptides have shown their promise towards the antimicrobial activity however the family Viperidae snake venoms have not been explored thoroughly, although they are a major source of so many active peptides [13, 32]. The antimicrobial peptides are electrically attracted to the negative charged groups of the cell surface, where they develop an α- helical conformation and these charged groups get accumulated on the membrane. This can result in the formation of transient pores, membrane perturbation and ultimately the cell lysis [33].
In this study we employed 4 crude venoms of the snakes of Viperidae and 2 crude venoms of Elapidae family for their effects on the growth of pathogenic microorganisms. Similar screening focusing on antimicrobial property has not been attempted previously among indigenous Saudi Arabian snakes.
MATERIALS AND METHODOLOGY
Materials
Snake venoms of two snake groups that belong to two families (Viperidae and Elapidae) were obtained from the Research Center, Prince Sultan Military Medical City (PSMMC), Riyadh, Saudi Arabia. The initial stocks of venoms were prepared in normal saline at a concentration of 10mg/mL and purified by passing through 0.22µm membrane filter. Further dilutions were made in sterile saline and used in this study. Freeze dried microbial cultures of S. aureus (S. aureus) ATCC 25923, E. faecalis (E. faecalis) ATCC 29212, E. coli (E. coli) ATCC 25922, P. aeruginosa (P. aeruginosa) ATCC 27853 and Candida albicans (C. albicans) ATCC 66027 were purchased from Microbiologics, Inc. (St. Cloud, MN. USA). The laboratory strain of methicillin resistant S. aureus (MRSA) 12498 was obtained from the Department of Microbiology in PSMMC. Brain Heart Infusion broth (BHIB), Sabouraud Dextrose broth (SDB), Brain Heart Infusion agar (BHIA), Sabouraud Dextrose agar (SDA) were purchased from local company agents in Riyadh, Saudi Arabia. The antifungal (Fungizone) was originally bought from Gibco Life Technology, U.K. and the antibiotic (Vancomycin) was from Sigma, USA and were used accordingly.
Venoms
The initial stocks of venoms were prepared in normal saline at a concentration of 10mg/mL and sterilized by 0.22µm membrane filter and stored in -20º C until use.
Growth Media
All of the bacterial cultures with the exception of C. albicans were grown in BHIB and the stocks prepared in 50% glycerol were stored at -40º C. C. albicans culture was grown in SDB and stored at -40º C as glycerol stock culture. The enumeration of C. albicans colonies and the growth inhibition were determined on SDA-plates prepared in the laboratory.
Microbial Inoculum
Microbial inoculum that would be used for antibacterial screening assays was prepared using the standard method of log-phase growth and were standardized to 0.5 McFarland unit and log dilutions were made in phosphate buffer saline (PBS) for each microorganism.
Agar Diffusion Procedure
Antimicrobial susceptibility tests were performed by the agar-diffusion method, which was based on Ouchterlony technique and slightly modified according to Barry and Brown [34]. Sterilized BHIA was melted in a microwave oven and then placed in a water bath set at 45ºC. Once the temperature was stable, bottles containing agar were removed and 0.1mL of overnight grown bacterial cultures were added. Two to three Petri plates were poured for each microbe and once the agar was solidified, plates were transferred to the refrigeration temperature for 2-3 hours. A dedicated agar punch was used to cut holes in agar and 0.05 mL of various dilutions of venom (0.25, 0.5, 1, 2.5, 5, 10 mg/mL) was added to each well. Normal saline, used as a diluent was employed as a negative control. After leaving plates on the bench for an hour, they were transferred to the incubator. The zones of microbial growth inhibition were recorded after 48h of incubation.
Determination of Colony Forming Units (CFU) and Minimum Inhibitory Concentration (MIC)
24h microbial colony cultures were standardized to 0.5 McFarland unit and log dilutions were made in phosphate buffer saline (PBS) for each microorganism. Several dilutions of the crude venom (0.0625, 0.125, 0.25, 0.5, 1, 2.5, 5, 10 mg/mL) in triplicate were mixed with the microbial inoculum and incubated at 37ºC incubator. The PBS was used as negative control. At 24h of incubation, aliquots of samples were removed and log dilutions were prepared in sterile PBS. Petri plates containing BHIA were inoculated with 100µL of inoculum in triplicate and this inoculum was spread using a sterile glass rod bent at 90ºC and a plate rotator (Sensoturn). The Petri plates were incubated for 48h and the CFU were counted either with naked eye or using a colony counter.
RESULTS
Table 1 shows mean zones of microbial growth inhibition (in mm), which were obtained by reading the agar diffusion plates. Out of eight concentrations ((0.0625, 0.125, 0.25, 0.5, 1, 2.5, 5, 10 mg/mL) that gave the minimum inhibitory concentrations (MIC), six most effective venom concentrations
Mean zones of microbial growth inhibition (in mm), which were caused by the six most effective venom concentrations of six snakes.
| Family Viperidae | ||||||
| E. pyramidum venom concentration - zone of Growth Inhibition (in mm) | ||||||
| 0.25 mg/mL | 0.5 mg/mL | 1 mg/mL | 2.5mg/mL | 5.0mg/mL | 10mg/mL | |
| S. aureus | 7.25 | 9.25 | 9.75 | 12.25 | 13.5 | 15 |
| S. aureus (MRSA) | 11 | 12 | 12.25 | 13 | 14 | 15.25 |
| E. faecalis | 0 | 0 | 0 | 6.25 | 8 | 9 |
| E. coli | 6 | 8.25 | 8 | 10.5 | 12 | 13.25 |
| P. aeruginosa | 0 | 0 | 0 | 6 | 7.5 | 8.5 |
| E. coloratus venom concentration - zone of Growth Inhibition (in mm) | ||||||
| S. aureus | 7 | 10 | 11 | 12 | 12 | 13 |
| S. aureus (MRSA) | 12 | 14 | 15.5 | 16.25 | 17.25 | 18.25 |
| E. faecalis | 0 | 6.75 | 8 | 10.25 | 12.5 | 13 |
| E. coli | 0 | 7 | 9 | 10.25 | 11 | 12.5 |
| P. aeruginosa | 0 | 0 | 0 | 7 | 8 | 8.75 |
| C. gasperettii venom concentration - zone of Growth Inhibition (in mm) | ||||||
| S. aureus | 6.75 | 7.25 | 8.75 | 10 | 11 | 12 |
| S. aureus (MRSA) | 14.25 | 10 | 11.5 | 13.25 | 14.5 | 15 |
| E. faecalis | 0 | 0 | 0 | 0 | 8 | 10 |
| E. coli | 0 | 0 | 0 | 6 | 6.5 | 7 |
| P. aeruginosa | 0 | 0 | 0 | 0 | 0 | 6.25 |
| Bitis arietans venom concentration - zone of Growth Inhibition (in mm) | ||||||
| S. aureus | 0 | 0 | 6.5 | 7 | 7 | 7.5 |
| S. aureus (MRSA) | 0 | 0 | 7 | 8.5 | 9.25 | 10 |
| E. faecalis | 0 | 0 | 0 | 0 | 0 | 0 |
| E. coli | 0 | 0 | 0 | 0 | 0 | 0 |
| P. aeruginosa | 0 | 0 | 0 | 0 | 0 | 0 |
| Family Elapidae | ||||||
| N. arabica venom concentration - zone of Growth Inhibition (in mm) | ||||||
| S. aureus | 0 | 6 | 7.25 | 9.25 | 10 | 11 |
| S. aureus (MRSA) | 6.5 | 9.25 | 9.5 | 11.25 | 12.5 | 14.4 |
| E. faecalis | 0 | 0 | 6 | 7 | 8 | 10.5 |
| E. coli | 0 | 6 | 6 | 7 | 9.5 | 11.5 |
| P. aeruginosa | 0 | 0 | 0 | 6.5 | 7 | 7 |
| W. aegyptia venom concentration - zone of Growth Inhibition (in mm) | ||||||
| S. aureus | 6.75 | 9.5 | 10 | 11.25 | 12 | 13.75 |
| S. aureus (MRSA) | 13 | 14 | 15.75 | 16.5 | 18.5 | 20.5 |
| E. faecalis | 0 | 7 | 8 | 10.5 | 12.5 | 13.25 |
| E. coli | 0 | 7.25 | 9 | 10 | 11 | 12.25 |
| P. aeruginosa | 0 | 6 | 6.75 | 7 | 7 | 8.25 |
were found to be the most relevant ranges of the six snake venoms, applicable for statistical analyses. The paired T-test was employed to verify the significance of venom concentration effect of all snake species with respect to control (saline). For the first viper snake E. pyramidum, the venom concentration range (0.25mg - 10mg) gave 4.00 mm - 10.00 mm mean growth inhibition zones (GIZ) for all five microbial species (Table 2).
Paired T-test analysis of viper snake venoms concentration mean microbial zone of growth inhibition against control (Normal Saline).
| Viper snake venom concentration | Mean | Std. Error Mean | t | Significance (2-tailed) |
|---|---|---|---|---|
| E. pyramidum Veonm 0.25mg | 4.00 | 1.915 | 2.089 | 0.091 |
| E. pyramidum Veonm 0.5mg | 4.83 | 2.227 | 2.170 | 0.082 |
| E. pyramidum Veonm 1mg | 5.00 | 2.295 | 2.179 | 0.081 |
| E. pyramidum Veonm 2.5mg | 7.83 | 1.973 | 3.969 | 0.011 |
| E. pyramidum Veonm 5.0mg | 9.33 | 2.171 | 4.300 | 0.008 |
| E. pyramidum Veonm 10mg | 10.00 | 2.338 | 4.277 | 0.008 |
| E. coloratus Venom 0.25mg | 3.17 | 2.104 | 1.505 | 0.193 |
| E. coloratus Venom 0.5mg | 6.33 | 2.261 | 2.801 | 0.038 |
| E. coloratus Venom 1mg | 7.33 | 2.578 | 2.845 | 0.036 |
| E. coloratus Venom 2.5mg | 9.17 | 2.197 | 4.172 | 0.009 |
| E. coloratus Venom 5.0mg | 10.00 | 2.324 | 4.303 | 0.008 |
| E. coloratus Venom 10mg | 10.83 | 2.469 | 4.388 | 0.007 |
| Cerastes gasperettii Venom 0.25mg | 3.50 | 2.391 | 1.464 | 0.203 |
| Cerastes gasperettii Venom 0.5mg | 2.83 | 1.833 | 1.545 | 0.183 |
| Cerastes gasperettii Venom 1mg | 3.50 | 2.247 | 1.557 | 0.180 |
| Cerastes gasperettii Venom 2.5mg | 4.83 | 2.344 | 2.062 | 0.094 |
| Cerastes gasperettii Venom 5.0mg | 6.50 | 2.335 | 2.784 | 0.039 |
| Cerastes gasperettii Venom 10mg | 8.33 | 2.140 | 3.895 | 0.011 |
| Bitis arietans Venom 1mg | 2.17 | 1.376 | 1.574 | 0.176 |
| Bitis arietans Venom 2.5mg | 2.50 | 1.586 | 1.576 | 0.176 |
| Bitis arietans Venom 5.0mg | 2.67 | 1.706 | 1.563 | 0.179 |
| Bitis arietans Venom 10mg | 3.00 | 1.915 | 1.567 | 0.178 |
Only concentration 2.5mg, 5.0mg and 10mg gave significant (P≤ 0.011, P≤ 0.008 and P≤ 0.008, respectively) GIZ with respect to controls (Table 2).
For the second viper snake E. coloratus, the venom concentration range (0.25mg - 10mg) gave 3.17 mm - 10.83 mm mean GIZ for all five microbial species. The concentration 0.5mg, 1mg, 2.5mg, 5.0mg and 10mg gave significant (P≤ 0.038, P≤ 0.036, P≤ 0.009, P≤ 0.008 and P≤ 0.007, respectively) GIZ with respect to controls (Table 2).
For the third viper snake C. gasperettii, the venom concentration range (0.25mg - 10mg) gave 3.50 mm - 8.33 mm mean GIZ for all five microbial species. The concentration 5.0mg and 10mg gave significant (P≤ 0.039 and P≤ 0.011, respectively) GIZ with respect to controls (Table 2).
For the fourth viper snake Bitis arietans, the only concentration range of its venom (1mg - 10mg) gave 2.17 mm - 3.00 mm mean GIZ for all five microbial species. All these concentrations (1mg 2.5mg, 5.0mg and 10mg) gave insignificant (P≤ 0.176, P≤ 0.176, P≤ 0.179 and P≤ 0.178, respectively) GIZ with respect to controls (Table 2).
With respect to the effect of elapid snake venom concentrations, the first one N. arabica venom concentration range (0.25mg - 10mg) gave 1.00 mm - 9.00 mm mean GIZ for all five microbial species. Out of six, only the concentrations 1mg, 2.5mg, 5.0mg and 10mg gave significant (P≤ 0.032, P≤ 0.007, P≤ 0.006 and P≤ 0.007, respectively) GIZ with respect to controls (Table 3).
Paired T-test analysis of elapid snake venoms concentration mean microbial zone of growth inhibition against control (Normal Saline).
| Elapid (Cobra) snake venom concentration | Mean | Std. Error Mean | t | Significance (2-tailed) |
|---|---|---|---|---|
| Naja arabica Venom 0.25mg | 1.00 | 1.000 | 1.000 | 0.363 |
| Naja arabica Venom 0.5mg | 3.50 | 1.628 | 2.150 | 0.084 |
| Naja arabica Venom 1mg | 4.83 | 1.641 | 2.945 | 0.032 |
| Naja arabica Venom 2.5mg | 6.67 | 1.520 | 4.385 | 0.007 |
| Naja arabica Venom 5.0mg | 7.83 | 1.721 | 4.552 | 0.006 |
| Naja arabica Venom 10mg | 9.00 | 2.033 | 4.427 | 0.007 |
| W. aegyptia Venom 0.25mg | 3.33 | 2.246 | 1.484 | 0.198 |
| W. aegyptia Venom 0.5mg | 7.33 | 1.892 | 3.877 | 0.012 |
| W. aegyptia Venom 1mg | 8.33 | 2.108 | 3.953 | 0.011 |
| W. aegyptia Venom 2.5mg | 9.00 | 2.160 | 4.166 | 0.009 |
| W. aegyptia Venom 5.0mg | 10.00 | 2.463 | 4.060 | 0.010 |
| W. aegyptia Venom 10mg | 11.17 | 2.738 | 4.079 | 0.010 |
The second elapid snake W. aegyptia venom concentration range (0.25mg - 10mg) gave 3.33 mm - 11.17 mm mean GIZ for all five microbial species. All six venom concentrations, except the first (0.25mg) gave significant (P≤ 0.012, P≤ 0.011, P≤ 0.009, P≤ 0.010 and P≤ 0.010, respectively) GIZ with respect to controls (Table 3).
It was observed that GIZ had a directly proportional relationship with venom concentration for all experimental venom groups.
The Tukey's Multiple Comparison Test was employed to determine the susceptibility of each microorganism compared to control (Normal saline) based on the GIZ (Table 4). S. aureus (MRSA) bacterium was the one with the largest (12.38 mm) Mean Difference and the highest (20.45) q level and a highly (P < 0.001) significant probability value. Next susceptible bacterium was S. aureus with 8.958 mm Mean Difference, q level of 14.80 and a highly (P < 0.001) significant probability value. Next were E. coli and E. faecalis bacterial species, withthe same highly (P < 0.001) significant probability values. The least susceptible bacterium was P. aeruginosa with 2.986 mm Mean Difference, q level of 4.933 and a significant (P < 0.05) probability value.
Tukey's Multiple Comparison Test of microbial growth inhibition level caused by various snake venom concentrations and a control (Normal saline).
| Tukey's Multiple Comparison Test | Mean Difference | q | P value | 95% CI of diff |
| S. aureusvs Control | 8.958 | 14.80 | P < 0.001 | 6.393 to 11.52 |
| S. aureus (MRSA) vs Control | 12.38 | 20.45 | P < 0.001 | 9.814 to 14.94 |
| E. faecalisvs Control | 4.847 | 8.008 | P < 0.001 | 2.282 to 7.413 |
| E. colivs Control | 6.021 | 9.946 | P < 0.001 | 3.455 to 8.586 |
| P. aeruginosavs Control | 2.986 | 4.933 | P < 0.05 | 0.4208 to 5.551 |
Fig. (1) shows the comparative differences between effects of the elapid and viperid snake venoms, one antibiotic (Vancomycin), one antimycotic (Fungizone) and a negative control (normal saline) on growth of five pathogenic bacterial species and a fungus shown GIZ (in mm). The standard antibiotic (Vancomycin) was the most effective antibacterial agent in inhibiting the growth of four bacterial species (S. aureus (MRSA), E. faecalis, S. aureus and E. coli), in that order. All venoms were next to it in activity, though they were more efficient in inhibiting the growth of P. aeruginosa except the venom of the viper C. gasperettii. Both Vancomycin and venoms were unable to inhibit the fungal Candida albicans growth whilst its specific antifungal Fungizone (Amphotericin B) highly efficient with no antibacterial activity.
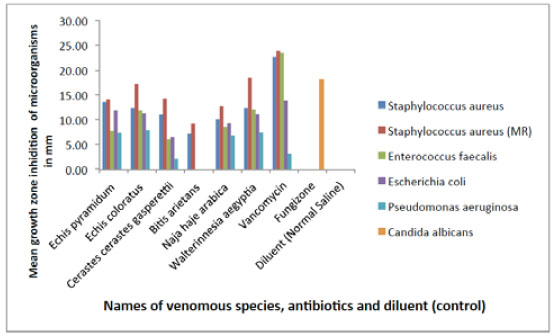
Comparative differences between effects of snake venoms, antibiotics, antimycotics and normal saline (control). Inhibition of microbial growing of six pathogenic bacterial species and a fungus shown as mean growth inhibition zone (in mm).
DISCUSSION
Venoms, especially those of snakes are a mixture of proteins and peptides including the nucleotides, free lipids and carbohydrates, which are bound to proteins [35]. They have consistently shown high levels of heterogeneity and intra and interspecies variation and this could be due to local adaptation for feeding on different prey [36]. The venoms obtained from the Viperidae family had long been recognized for their complexity of molecular composition [36]. Several studies had described the antimicrobial effect of snake venoms, which enlightened the emergence of bio-active peptides as therapeutic alternatives to combat the antibiotic resistant microorganisms [37].
Six venom concentrations were chosen out of eight that showed the least concentrations that represented the MIC but, this choice was applied according to the analysis of the GIZ. Paired T-test analysis of the selected concentration ranges of the six snake venoms has clearly shown significant GIZ, with varying degrees. With respect to venoms of the vipers, E. coloratus was the most effective against the five microorganisms, reflecting the largest mean of GIZ and a significance of five rising venom concentrations. Venoms of E. pyramidum and C. gasperettii have got medium effects, presenting only three and two significant venom concentrations, respectively. The puff adder Bitis arietans venomhas got the least effect and none of the six venom concentrations showed any significant GIZ. Previous studies have reported comparable RESULTS [5, 38].
On the other hand, venoms of the cobras showed to be very effective against the five microorganisms, with comparable significance to viper venoms and though the cobra W. aegyptia showed five significant rising venom concentrations, similar to that of E. coloratus (the first most effective viper venom) but, it exceeded it (the cobra exceeded the viper) in GIZ value. The second was the cobra N. arabica and though it had got a slightly less GIZ value than the second viper (E. pyramidum, with three significant venom concentrations), but, it got more significant venom concentrations (four) which, showed that this cobra venom was more effective than the second viper. One more venom concentration in the serial range presented a more dilute concentration that implies a more effective venom type, according to the MIC criteria and the directly proportional relationship between venom concentration and GIZ. This put the second cobra as a the third in effectiveness of the whole list of venoms. The group of cobra venoms appeared to be relatively more efficient as antimicrobial agents than viper venoms. Some previous studies [39] reported contradictory conclusions to our RESULTS though, some cobra venoms (Ophiophagus hannah) were also more active than those of the vipers. It was suggested that snake venom antimicrobial activity was due to enzymes such as PLA2 [9, 40, 41], which is also available in the venoms of cobras.
With respect to microorganism susceptibility, The Gram positive cocci S. aureus (MRSA) bacterium appeared to be the most sensitive to venoms, as having the largest mean difference and a highly significant probability value. Close to it was the second Gram positive cocci S. aureus in sensitivity to venoms, with the same highly significant probability value. Venom sensitivity of these Gram positive bacteria and other species has been reported before [42]. The Gram negative E. coli and the positive E. faecalis bacterial species, have also gotthe same highly significant probability but, their mean difference GIZ values were less, thus showing some relative resistance to venom action. The least susceptible bacterium was also the Gram negative P. aeruginosa which, had got a minimum of mean difference with a relatively significant value. Resistance of the Gram negative bacteria had been attributed to the outer membrane of the bacteria formed of lipopolysaccharides (LPS) which affected the uptake of antimicrobial peptides [43].
Levels of elapid and viperid snake venoms presented a fair pattern of effectiveness in comparative differences with the two standard treatment agents, the antibiotic (Vancomycin), and the antimycotic (Fungizone), employing GIZ parameters of selected three concentrations. Vancomycin as a specifically effective antibacterial agent, was the best in inhibiting the growth of S. aureus (MRSA), E. faecalis, S. aureus and E. coli, in that order, whilst all the venoms closely came next, with varying levels. Venoms showed to be more efficient than Vancomycin in inhibiting the growth of the more resistant P. aeruginosa,excepting the venom of C. gasperettii. Several works concerning comparisons of venoms and venom fraction with antibiotics had been reported [14, 40]. With respect to the fungus Candida albicans, RESULTS were negative, hence they were removed from the analysis tables.
CONCLUSION
It was concluded here that snake venoms have comparable activity, if not more efficient than antibiotics, whilst cobra venoms appear to be relatively more efficient as antimicrobial agents than viper venoms. Until furthermore studies, the selected six snake venoms, within the employed ranges of concentrations, cannot be a suitable solution in treatment of fungi like Candida albicans.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
The study was supported by Prince Sultan Military Medical City, Riyadh, Saudi Arabia. The authors wish to thank to Mr. Syed Ali for his help in this study.


