All published articles of this journal are available on ScienceDirect.
Distinct Interaction of Two Atypical Enteropathogenic Escherichia coli Strains with Enterocytes In Vitro
Abstract
Typical and atypical Enteropathogenic Escherichia coli (EPEC) promote attaching-effacing lesions in intestinal cells but only typical EPEC carry the EPEC adherence factor plasmid. Atypical EPEC (aEPEC) are emerging agents of acute and persistent diarrhea worldwide. We aimed at comparing the ability of two aEPEC strains, 1711-4 (serotype O51:H40) and 3991-1 (serotype O non-typeable:non-motile) to invade, persist inside Caco-2 and T84 cells, and to induce IL-8 production. Typical EPEC strain E2348/69 was used for comparisons. The strains associated more significantly with T84 than with Caco-2 cells, with 3991-1 being the most adherent (P < 0.001). In contrast, aEPEC 1711-4 was significantly more invasive than the other strains in both cell lines, and was found within vacuoles near the basolateral cell surfaces. Strains persisted within both cell lines for at least 48 hours, but the persistence index was higher for 3991-1 in Caco-2 cells. IL-8 production was significantly higher from Caco-2 cells infected with 1711-4 for at least 48 hours (P < 0.001), and from T84 cells after 24 and 48 h than with the other strains (P = 0.001). We demonstrated that aEPEC are heterogeneous in various aspects of their interaction with enterocytes in vitro.
INTRODUCTION
Enteropathogenic Escherichia coli (EPEC) are currently classified in two subgroups, based on the presence of the EPEC adherence factor plasmid (pEAF), in typical EPEC (tEPEC), and those without in atypical EPEC (aEPEC) [1, 2]. This plasmid contains the bfp operon encoding the bundle forming pilus (BFP), a fimbrial structure involved in the production of the localized adherence phenotype on HeLa and HEp-2 cells. This phenotype is not produced by aEPEC strains due to their inability to produce BFP [3].
aEPEC are emerging pathogens associated with acute and persistent diarrhea affecting children and adults even in developing countries [4-9]. aEPEC promote attaching-effacing (A/E) lesions in intestinal cells indistinguishable from those of tEPEC [2, 6, 10]. The essential virulence determinant of both EPEC subgroups is the chromosomal pathogenicity island LEE (Locus of Enterocyte Effacement). This locus contains the genes that code for effector proteins implicated in the formation of A/E lesions in eukaryotic cells, characterized by intimate adherence, local microvillus effacement and accumulation of polymerized actin beneath adherent bacteria forming structures which resemble pedestals [1].
EPEC strains are not considered typical invasive pathogens, despite the fact that some tEPEC strains were found within eukaryotic cells in vitro and ex vivo [11-13]. In addition, recent publications have provided evidence that some aEPEC strains may show varying degrees of enterocyte invasion in vitro [14-16], with most aEPEC strains tested being more invasive than tEPEC prototype strain E2348/69 [15, 17].
In a survey on the aetiology of diarrhea undertaken in three different cities in the Southeast region of Brazil, the most prevalent aEPEC serotype among typable strains was O51:H40 (10% of all isolates), while non-motile strains comprised 25% of all isolates [5]. Nguyen and colleagues [7] have provided epidemiological evidence that patients infected with aEPEC are more prone to have prolonged diarrhea than those infected with tEPEC, but the virulence factors associated with this finding are not well established.
In a recent publication, we have demonstrated that, besides promoting A/E lesions, aEPEC strain 1711-4 (serotype O51:H40) invades and induces IL-8 production in differentiated intestinal Caco-2 and T84 cells through a flagella-dependent pathway [16]. This finding is consistent with the fact that Toll-like receptor 5 (TLR5) recognizes bacterial flagellin and activates host inflammatory responses, thereby inducing enterocytes to produce IL-8 [18, 19]. In addition, it has been shown that aEPEC strain 3991-1 (O non-typable) induces mucous hyper-secretion in vitro and in rabbit ileal loops [20]. However it is currently not known whether these strains can persist in the intracellular compartment. Besides representing a mechanism of escape from the host immune response, permanence in the intracellular milieu after invasion could be a way to promote persistent disease or to prolong the patients’ carrier state. Furthermore, as aEPEC strains are heterogeneous in various features [5], it is not known whether aEPEC 3991-1 also invades and induces IL-8 production in vitro. We aimed at comparing the ability of two aEPEC strains (3991-1; 1711-4) to invade and persist inside Caco-2 and T84 cells, as well as to induce IL-8 production. Prototype tEPEC strain E2348/69 (serotype O127:H6) was also used for comparisons.
MATERIALS AND METHODOLOGY
Bacterial Strains
aEPEC strains 1711-4 (serotype O51:H40) and 3991-1 non-typable, non-motile (serotype NT:NM) [21] and tEPEC prototype strain E2348/69 (serotype O127:H6) were cultivated overnight at 37ºC in Luria Bertani (LB) broth.
EUKARYOTIC CELL CULTURE
Caco-2 and T84 cells were cultivated respectively in Dulbecco’s minimal Eagle medium (DMEM) (Gibco-BRL) supplemented with 20% fetal bovine serum (FBS) (Gibco-BRL), 1% nonessential amino acids and DMEM/NUT.MIX F12 Glutamax-1 medium (Gibco-BRL) supplemented with 10% FBS, both with 1% penicillin-streptomycin. Cultures were incubated at 37ºC in 5% CO2 - 95% air atmosphere and then seeded (105 cells/well) into 6-well plates and incubated for 10 days.
MONOLAYER INFECTION
Forty microliters of bacterial suspension from LB broth (107 CFU/ml) were added to each monolayer prior to centrifugation for 5 min at 730 x g. After incubation for 3 h at 37ºC in 5% CO2 – 95% air atmosphere, monolayers were washed with sterile phosphate buffered saline (PBS). To evaluate adhesion, 1% Triton X-100 was added to a group of monolayers before incubation at room temperature for 30 min. To evaluate invasion ability, a second group of monolayers was further incubated for 2 h in the presence of 100 μg/ml of gentamicin (Sigma). A subset of these monolayers was washed and then lysed as described above. The remaining monolayers were incubated in DMEM plus FBS and 10 µg/ml of gentamicin for 48 h, and then lysed. All lysates were serially diluted and plated on LB agar plates, which were incubated at 37ºC for 24 h with subsequent counting of the number of colony forming units (CFU). The percentages of cell-associated bacteria, which survived after treatment with gentamicin were determined [22]. Cell-association indexes (AX) were calculated using the following equation: (AB/INOC) x 100, where AB is the number of cell-associated bacteria, which includes adhered and intracellular bacteria and INOC is the number of bacteria in initial inoculums. AX represents the percentage of inoculated bacteria that adhered to and/or invaded eukaryotic cells until three hours after infection.
Invasion indexes (INVX) were calculated using the following equation: (ICB/AB) x 100, where ICB is the number of intracellular bacteria. INVX represents the percentage of cell-associated bacteria that invaded eukaryotic cells. Persistence indexes after 48 h infection (PIX48) were calculated using the following equation: PIX48 = (ICB48/ICB3) x 100, where ICB3 and ICB48 are respectively the number of intracellular bacteria at 3 and 48 h after infection. All assays were performed three times in triplicates.
TRANSMISSION ELECTRON MICROSCOPY (TEM)
Differentiated Caco-2 and T84 cells were infected as described above, except that preparations were not submitted to centrifugation. Preparations were rinsed in PBS, fixed with 2.5% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.2), post fixed in 1% osmium tetroxide, dehydrated through a series of graded ethanol solutions and propylene oxide, and embedded in Araldite®. Ultra-thin sections were stained with 2.0% uranyl acetate and 2.5% lead citrate, and examined in a transmission electron microscope (LEO 906E, Zeiss) at 80 kV.
IL-8 SECRETION
Cell cultures supernatants were collected at 3, 24 and 48 h after infection. IL-8 concentrations were measured using the Human IL-8 Duo Set Elisa Development System kit (R&D Systems).
STATISTICAL ANALYSIS
All statistical analyses were performed with Prism version 5.03 from GraphPad Software Inc., USA. ANOVA and Bonferroni’s post-hoc tests were applied to evaluate all results. A statistical significance of 95% was applied (α ≈ 0.05).
RESULTS
To compare the invasive ability of both aEPEC strains and the tEPEC prototype strain E2348/69, we first evaluated their efficiency in associating (adhering and invading) with Caco-2 and T84 intestinal cells. All three strains tested associated more efficiently with the T84 cell line than with the Caco-2 cell line (P < 0.001) (Fig. 1). The Association index (AX) of aEPEC 3991-1 in T84 cells (26.7%) was significantly greater than that observed in Caco-2 cells (1.6%) (P < 0.001). Likewise, the AXs of aEPEC 1711-4 (7.0%) and tEPEC E2348/69 (5%) in T84 cells were greater than those observed in Caco-2 cells (1.4% and 2.0%, respectively), although this difference was not statistically significant (P > 0.05).
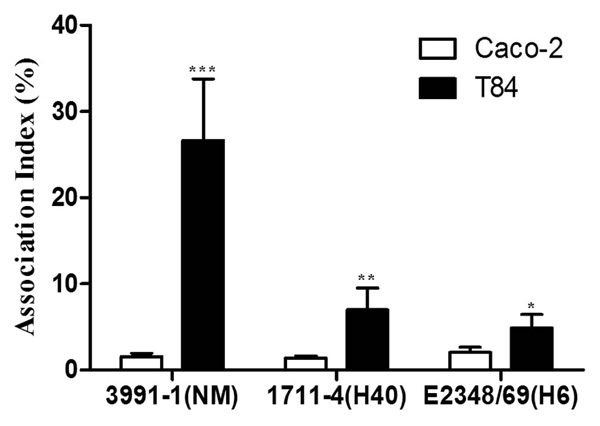
Association of aEPEC and tEPEC strains with differentiated Caco-2 or T84 intestinal cells. Experiments were performed in triplicates at least three times; error bars indicate standard deviation. *** Indicates statistically significant differences between cell lineages (P < 0.001); ** indicates statistically significant difference between aEPEC 3991-1 and aEPEC 1711-4 in T84 cells (P = 0.004); * indicates statistically significant difference between aEPEC 3991-1 and tEPEC E2348/69 in T84 cells (P = 0.004).
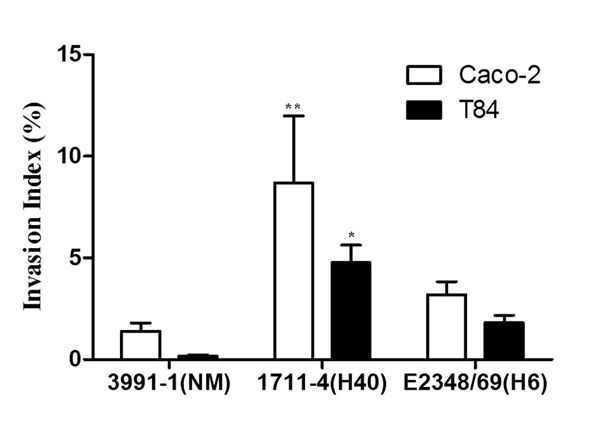
Invasion of differentiated intestinal Caco-2 and T84 cells by aEPEC and tEPEC strains. Experiments were performed in triplicates at least three times; error bars indicate standard deviation. ** indicates statistically significant difference between aEPEC 1711-4 and 3991-1 or tEPEC E2348/69 in Caco-2 cells; (P = 0.006); * indicates statistically significant differences between aEPEC 1711- 4 and 3991-1 and tEPEC E2348/69 in T84 cells (P < 0.001).

Transmission electron microscopy of differentiated Caco-2 (A, B, E and F) or T84 (C, D, G and H) cells fixed at 3 h or 6 h after infection with aEPEC 1711-4 (A, C, E and G) or aEPEC 3991-1 (B, D, F and H). Note the formation of large bacterial clusters and pedestal formation at the apical surfaces of cells at 3 h after infection. Partial internalization with presence of electron dense material below bacteria (C – black arrow head) indicating actin accumulation can be detected at 3 h after infection with aEPEC 1711-4. Black arrows indicate aEPEC 1711-4 residing within vacuoles and forming pedestals at the basolateral portion of enterocytes (E and G). NI represent uninfected Caco-2 and T84 cells respectively.
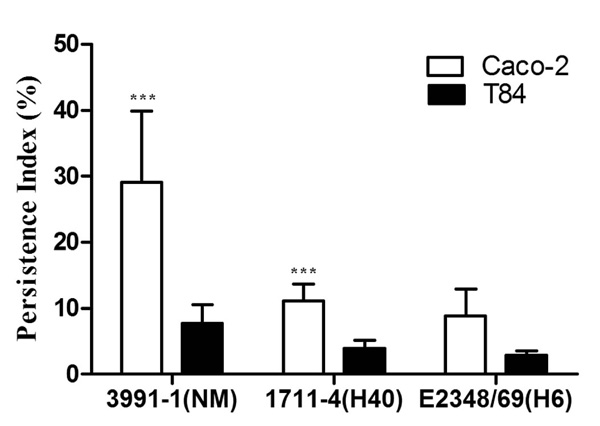
Persistence of aEPEC and tEPEC strains in Caco-2 and T84 cells 48 h after infection. Experiments were performed in triplicates at least three times. Error bars represent standard deviation. ***Indicates statistically significant difference between cell lineages (P = 0.03).

IL-8 concentrations in Caco-2 (A) and T84 (B) cell culture supernatants at 3, 24 or 48 h after infection with aEPEC or tEPEC strains or in the absence of infection. Experiments were performed in triplicates at least three times. Error bars indicate standard deviation. (A) aEPEC 1711-4 stimulated IL-8 levels at least three fold those of aEPEC 3991-1 or tEPEC E2348/69, in all periods tested. These differences were statistically significant (P ≤ 0.001). (B) - IL-8 levels induced by aEPEC 1711-4 at 3, 24 and 48 h after infection were significantly higher than those induced by the other strains (P = 0.001).
The AX obtained with aEPEC 3991-1 in T84 cells was significantly higher than those observed with aEPEC 1711-4 and tEPEC E2348/69 (P = 0.004). However, the AXs of the strains in Caco-2 cells showed no significant differences (P = 0.09) (Fig. 1).
Although in general the Invasion Indexes (INVX) obtained after gentamicin treatment (which eliminates extracellular bacteria) were higher in Caco-2 than in T84 cells, but no statistically significant differences were found between them (P = 0.50) (Fig. 2). In Caco-2 cells aEPEC 1711-4 showed the highest INVX (14.8%) compared to 1.4% and 3.2% for aEPEC 3991-1 and tEPEC E2348/69, respectively (P = 0.006). In T84 cells, the INVX of aEPEC 1711-4 (4.8%) was significantly higher than those of aEPEC 3991-1 (0.2%) and tEPEC E2348/69 (1.8%) (P ≤ 0.001) (Fig. 2).
The association of the aEPEC strains with intestinal Caco-2 and T84 cells at three and six hours after infection was analysed by TEM (Fig. 3). Like tEPEC E2348/69 (not shown), both aEPEC strains formed bacterial clusters at the apical surfaces three hours after infection with characteristic A/E lesions (Fig. 3A to D). After three hours, some aEPEC 1711-4 bacteria were seen partially internalised (Fig 3A and C). In both Caco-2 and T84 cell lines, internalised bacteria were observed six hours after infection, however, in contrast to aEPEC 3991-1, aEPEC 1711-4 bacteria were seen within vacuoles located close to the basolateral surface of the enterocytes, forming pedestal-like structures around these vacuoles (Fig 3E and G).
We next examined the presence of bacteria within enterocytes, 48 h after infection, as an indication of the potential of strains to persist in the intracellular compartment. During these experiments, enterocytes were maintained in media containing gentamicin that killed extracellular bacteria that occasionally escaped from them. Consequently, the Persistence Index (PIX48) could result from two processes: bacterial proliferation and bacterial death. For all strains tested, the PIX48 was higher in Caco-2 than in T84 cells, but only aEPEC 3991-1 showed a statistically significant difference in PIX48 between Caco-2 and T84 cells (P = 0.03) (Fig. 4). The PIX48 in Caco-2 cells were 29.1% for aEPEC 3991-1, 11.1% for aEPEC 1711-4 and 8.8% for tEPEC E2348/69, but these differences were not statistically significant (P = 0.09) (Fig. 4). Likewise, there were no statistically significant differences when PIX48 of the strains in T84 cells were compared (P = 0.17). The basal levels of IL-8 production from supernatants of the two different cell lines differed markedly and varied, from 8 to 26 pg/ml and from 114 to 156 pg/ml, in Caco-2 and T84 cells, respectively. The Caco-2 cells inoculated with aEPEC 1711-4 produced IL-8 at levels of 416 ± 115 pg/ml at 48 h and significantly greater than that of Caco-2 cells inoculated with aEPEC 3991-1 or tEPEC E2348/69 (P ≤ 0.001) over all time periods tested (Fig. 5A). In contrast there was no statistical significance between IL-8 production from Caco-2 cells inoculated with aEPEC 3991-1 and tEPEC E2348/69 (P > 0.05). For T84 cells, highest IL-8 levels were also detected from cells inoculated with aEPEC 1711-4 in all periods examined (Fig. 5B). Three hours after infection, a statistically significant difference was found only between aEPEC strains 1711-4 (181 ± 52 pg/ml) and 3991-1 (47 ± 13 pg/ml) (P = 0.05; 95% CI 9.7 – 259.6). Twenty four hours after inoculation, IL-8 level induced by aEPEC 1711-4 (484 pg/ml) was significantly higher than those induced by the other strains (P ≤ 0.001; 95% CI 191.7 – 516.2 when comparing 1711-4 and 3991-1 strains). Similar results were observed 48 h after infection, when the IL-8 levels induced by aEPEC 1711-4 (501 ± 164 pg/ml) was significantly higher than observed with the other strains (P = 0.001; 95% CI 107.6 – 512.6 when comparing aEPEC 1711-4 and 3991-1 strains).
DISCUSSION
Recent studies have demonstrated that although aEPEC are heterogeneous in nature, some strains are truly diarrheagenic [6, 23]. Furthermore, besides the characteristic ability to produce A/E lesions, some aEPEC strains may produce additional potential virulence mechanisms as compared to tEPEC strains. One such mechanism is the ability to invade epithelial cells in vitro [14-16]. Different degrees of invasiveness have been ascribed to many aEPEC strains but not all aEPEC strains are invasive [8, 17, 24].
We have recently demonstrated that aEPEC 1711-4 adheres, invades and stimulates IL-8 production in Caco-2 and T84 cells in vitro with partial involvement of the bacterial flagella in these characteristics [16]. In addition, we have shown that the non-flagellated strain aEPEC 3991-1 induced mucous hypersecretion in intestinal HT29-MTX cells, a property which did not affect its limited ability to invade these cells [20].
Enterocyte invasion could be a bacterial mechanism of escape of the extracellular immunological response. However, so far, the ability of these strains of persisting within the intracellular milieu has not been investigated. We conducted the present study to compare, under the same experimental conditions, the ability of the aEPEC strains 1711-4 and 3991-1 to adhere, invade and persist within enterocytes and to elicit IL-8 production for at least 48 hours. Two distinct enterocyte-derived lineages were used - Caco-2 and T84 cells– which spontaneously differentiate in vitro and express distinct functional and structural characteristics, mimicking the colonic intestines [25, 26]. In order to avoid interference of flagella motility in the interaction assays, cell monolayers were centrifuged immediately after inoculation. Under the same experimental conditions, aEPEC 3991-1 had a higher AX in T84 cells than aEPEC 1711-4 or tEPEC E2348/69. This finding suggests that flagella may not be the main factor required for aEPEC association (adherence) with T84 cells. Consequently, other factors expressed by aEPEC, as intimin, the Type Three Secretion System (EspA, EspB and EspD), or an as yet unknown virulence factors, could mediate this association.
In contrast to the AX data, the INVXs of all strains studied were higher in Caco-2 than in T84 cells, although these differences between tissue culture cells were not statistically significant. The aEPEC strain 1711-4 presented the highest INVX while aEPEC 3991-1 had the lowest INVX in both lineages. These findings are in accordance with our previous work showing that flagella are necessary for the efficient invasion ability of aEPEC 1711-4 [16] and with a recent publication demonstrating the importance of flagella in the invasion of eukaryotic cells by EHEC serotype O113:H21 [27]. However, since only one flagellated strain and one non-flagellated strain were tested, it is not possible to ascertain that the flagella are responsible for higher aEPEC invasion efficiency until additional flagellated and non-flagellated aEPEC strains are tested. Furthermore, aEPEC strains may differ by expressing alternative or additional effectors that might determine diverse invasion efficiencies [28].
TEM analysis indicated the presence of intracellular aEPEC in Caco-2 and T84 cells 6 h after infection. The aEPEC strain 3991-1 did not appear to readily invade either tissue culture cell line and there was no evidence of vacuole invasion (Fig. 3). However, aEPEC 1711-4 was found within vacuoles located close to the basolateral face of both T84 and Caco-2 cells. In addition, a protuberance suggestive of pedestal formation around the vacuole membrane was observed underneath adherent bacteria (Fig. 3). This finding is consistent with a previous report of pedestal formation in HeLa and Caco-2 cells with two aEPEC strains of serotypes ONT (non-typable):HNM (non-motile) [15] and O111:H9 [29], respectively. The invasive ability of the O111:H9 strain in HeLa and Swiss 3T3 cells has been shown to be due to EspT, a type three secretion system dependent effector protein encoded outside the LEE, that activates the mammalian GTPases Rac1 and Cdc42, resulting in the formation of membrane ruffles and lamelipodia that favoured invasion [29]. Interestingly, the strains of the present study lack the EspT-encoding genes [30].
High levels of IL-8 were found 24 and 48 h after infection, when all extracellular bacteria had been eliminated by the action of gentamicin. Our findings of lowest IL-8 levels induced by the non-flagellated aEPEC 3991-1 are in accordance with recent studies suggesting that flagellin is the main bacterial inducer of IL-8 production [16, 19, 31]. The finding that the non-flagellated strain 3991-1 induced IL-8 levels similar to that observed with the flagellated tEPEC E2348/69 supports the existence of a flagellin independent pathway culminating in IL-8 production stimulation. Our findings are in accordance with those published by Sharma et al. [32] who demonstrated that culture supernatants from FliC-deficient mutants of a tEPEC strain were able to trigger IL-8 production in HT-29 monolayers. We cannot exclude the possibility that the differences in IL-8 levels observed at 24 and 48 h are due to a heterogeneous capacity of the strains, which are suppressing or stimulating inducible nitric oxide synthase (iNOS) production, since invasive pathogens can activate iNOS production in the absence of cytokine treatment. Absence of iNOS expression in Caco-2 cells has been shown to result in lack of phosphorylation-associated activation of NF-κB [33]. Another possibility is that the interaction of intimin with its translocated receptor (Tir) continues to occur while bacteria are inside vacuoles, recruiting protein kinase C zeta, which is able to interact with and activate IκB kinase. This interaction leads to NF-κB release and nuclear translocation resulting in IL-8 mRNA transcription and IL-8 production. This could explain why aEPEC 1711-4, the most invasive strain studied, elicited the highest IL-8 levels.
All strains persisted in the intracellular compartment for at least 48 h. The PIXs for all strains were similar in both cell lineages, except for aEPEC 3991-1 that showed higher PIX in Caco-2 cells. Curiously, the highest PIX was observed with the aEPEC 3991-1, which had the lowest invasion index and IL-8 induction capacity. Modulation of inflammatory response resulting in lower IL-8 induction has already been described for a tEPEC strain [32]. Moreover, other virulence factors could contribute to the higher persistence of aEPEC 3991-1 inside enterocytes. Such persistence may be relevant to establish the carrier state and protect bacteria against phagocytic cells or secretory antibodies.
Although the ability to invade and persist inside enterocytes has to be confirmed in vivo, these properties could contribute to the establishment of persistent diarrhoea in some aEPEC infected patients.
CONCLUSIONS
Our data suggest that the aEPEC pathotype is heterogeneous regarding its ability to adhere, invade, persist and elicit IL-8 production by enterocytes in vitro.
ACKNOWLEDGEMENTS
This work was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (grants 2008/53812-4 and 2009/50399-1) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).


