All published articles of this journal are available on ScienceDirect.
Epithelial Upregulation of Toll-like Receptor 2 (TLR2) Fast-tracks Tissue Repair in ICR Mice Treated with Postbiotic Hydrogel Infused with Lactobacillus paracasei 7060
Abstract
Introduction
Impaired epithelial wound repair contributes to increased infection risk and delayed tissue recovery. Limitations associated with conventional topical therapies, including antimicrobial resistance, highlight the need for alternative strategies. Postbiotics, defined as bioactive microbial metabolites, have emerged as potential modulators of tissue repair and immune responses.
Methods
A postbiotic hydrogel formulated with metabolites derived from Lactobacillus paracasei 7060 was applied to full-thickness dorso-lumbar wounds in a murine model. Wound closure was assessed over a 21-day period. Histological analyses evaluated epithelialization and tissue organization, and expression of immune- and repair-associated genes was quantified by quantitative PCR.
Results
Treatment with the postbiotic hydrogel resulted in accelerated wound closure and earlier epithelial restoration compared with both the commercial hydrogel and untreated control groups. Histological assessment demonstrated improved epidermal continuity and reduced inflammatory cell infiltration in treated wounds. Gene expression analysis revealed a transient upregulation of Toll-Like Receptor 2 (TLR2) during the early phase of healing.
Discussion
The observed transient activation of TLR2 suggests a regulated immune response that may support early host defense and macrophage recruitment without sustained inflammation. Genomic features of L. paracasei 7060 are consistent with the production of metabolites involved in immune modulation, tissue repair, angiogenesis, and stress response pathways.
Conclusion
These findings indicate that a L. paracasei 7060–derived postbiotic hydrogel supports epithelial wound repair through coordinated immune and regenerative mechanisms.
1. INTRODUCTION
Epithelial tissues serve as the body’s first line of defense against environmental stressors, microbial invasion, and mechanical injury, while also supporting vital physiological functions such as absorption, secretion, and immune surveillance [1]. These tissues line the skin and mucosal surfaces, positioning them at the interface between the host and its environment. When epithelial integrity is disrupted, whether in the skin, gastrointestinal tract, or respiratory system, the body initiates a highly coordinated wound healing response involving inflammation, cellular proliferation, angiogenesis, and remodeling of the extracellular matrix (ECM). Precise regulation of each of these stages is essential, as failure to resolve injury appropriately can lead to chronic wounds, excessive scarring, fibrosis, or systemic infection, all of which significantly contribute to morbidity and mortality [2].
Loss of epithelial integrity markedly increases susceptibility to microbial invasion. Open wounds, burns, and damaged mucosal surfaces provide direct portals of entry for opportunistic pathogens, including antibiotic-resistant Gram-negative and Gram-positive bacteria as well as fungal species [3]. Once established, these infections intensify local inflammation, delay tissue regeneration, and can progress to life-threatening conditions such as sepsis if left unchecked. The clinical challenge is further compounded by the increasing prevalence of multidrug-resistant organisms, including Staphylococcus aureus, Escherichia coli, and Candida albicans, which frequently exhibit reduced responsiveness to conventional antimicrobial therapies. This growing resistance crisis underscores the urgent need for alternative therapeutic approaches that can simultaneously enhance host defense mechanisms and limit microbial burden at the wound site [4].
Inflammation plays a critical role in the early phase of wound healing by facilitating pathogen clearance and debris removal. However, when inflammatory responses become excessive or prolonged, they can perpetuate tissue damage rather than promote repair. Elevated concentrations of pro-inflammatory cytokines such as Tumor Necrosis Factor-alpha (TNF-α) and Interleukin-6 (IL-6), together with increased production of Reactive Oxygen Species (ROS), contribute to ECM degradation and interfere with epithelial cell proliferation and migration [5]. Chronic inflammatory disorders, including diabetic ulcers, inflammatory bowel disease, and chronic obstructive pulmonary disease, illustrate how unresolved epithelial injury leads to sustained tissue dysfunction and impaired healing. Persistent epithelial damage may also progress to fibrosis, a pathological condition characterized by excessive ECM deposition, particularly collagen, driven by the activation of myofibroblasts and regulated by signaling pathways involving Transforming Growth Factor-beta (TGF-β), integrins, and growth factor receptors [6, 7]. Although fibrosis initially serves to stabilize damaged tissue, uncontrolled fibrotic responses can result in disfiguring outcomes such as hypertrophic scars and keloids. Consequently, there is a critical need for therapeutic strategies that support regenerative healing while limiting excessive inflammation and fibrotic remodeling.
In recent years, increasing attention has been directed toward the microbiome and its bioactive derivatives as modulators of epithelial repair. Probiotics, defined as live microorganisms that confer health benefits to the host, have demonstrated promising effects in various wound healing and inflammatory models, largely due to their antimicrobial and immunomodulatory properties. Several studies have shown that Lactobacillus and Bifidobacterium species can enhance re-epithelialization, stimulate collagen deposition, and regulate cytokine production. For example, L. rhamnosus GG and B. animalis subsp. lactis BB12 was reported to accelerate granulation tissue formation while suppressing pro-inflammatory mediators such as TNF-α and IL-6, thereby promoting macrophage polarization toward an anti-inflammatory M2 phenotype [8, 9]. Topical application of L. plantarum protein fractions has been shown to enhance keratinocyte migration and modulate cytokine expression within wound beds [9], while oral supplementation with L. fermentum, L. casei, and B. bifidum reduced oxidative stress in inflammatory conditions [10]. Despite these benefits, the clinical use of live probiotics remains controversial, particularly in immunocompromised individuals or in open wound environments, where bacterial translocation and systemic infection represent potential safety concerns [11].
These limitations have driven growing interest in postbiotics, which are defined by the International Scientific Association for Probiotics and Prebiotics (ISAPP) as “preparations of inanimate microorganisms and/or their components that confer a health benefit on the host” [12]. Postbiotics encompass a wide range of microbial-derived molecules, including Short-Chain Fatty Acids (SCFAs), exopolysaccharides, bacteriocins, teichoic acids, peptidoglycans, lipoteichoic acids, enzymes, and heat-inactivated bacterial cells [13]. Accumulating evidence indicates that postbiotics can enhance epithelial barrier integrity through the upregulation of tight junction proteins [14], stimulate fibroblast proliferation, keratinocyte migration, and angiogenesis [15], and modulate immune responses by increasing anti-inflammatory cytokines such as IL-10 and TGF-β while suppressing pro-inflammatory mediators including TNF-α and IL-6 [16]. In addition, certain postbiotic components exhibit antimicrobial activity by disrupting pathogenic biofilms and interfering with quorum-sensing mechanisms [17].
Preclinical studies have demonstrated the capacity of postbiotic formulations to accelerate wound healing processes. For instance, supernatants derived from L. plantarum have been shown to enhance granulation tissue formation and promote re-epithelialization in cutaneous wound models [18]. Similarly, a lysate of L. paracasei CNCM I-1518 improved skin barrier function and reduced inflammatory symptoms in patients with atopic dermatitis [19, 20]. Compared with live probiotics, postbiotics offer several practical advantages, including improved safety profiles for high-risk populations, standardized composition and dosing, enhanced stability and shelf life without the need for refrigeration, and the absence of risks associated with microbial overgrowth or horizontal transfer of antibiotic resistance genes [21-23].
Despite their growing therapeutic potential, investigations into postbiotics for tissue regeneration remain limited, particularly those derived from plant-associated Lactic Acid Bacteria (LAB). Most existing studies focus on strains originating from the human gastrointestinal tract, leaving a substantial gap in knowledge regarding LAB isolated from plant sources. This gap is especially relevant for microbes derived from Nypa fruticans (nipa palm), an indigenous species found in Philippine mangrove ecosystems that is recognized for its rich and unique microbial diversity. In this study, we investigate the wound healing potential of a postbiotic hydrogel derived from Lactobacillus paracasei 7060, a strain isolated from nipa palm. Using phenotypic wound assessments, histological evaluation, and analysis of key inflammation- and regeneration-related genes (TLR2, TNF-α, and TGF-β), we aim to assess the capacity of this plant-derived postbiotic to favorably modulate the wound microenvironment. By integrating indigenous microbial resources with translational biomedical research, this work provides novel insights into the role of postbiotics in epithelial regeneration and immune modulation.
2. MATERIALS AND METHODS
2.1. Research Design
A Completely Randomized Design (CRD) was employed with five intervention groups and six biological replicates each. Blocking was based on tissue harvest time: half of each group was sacrificed on day 3 post-wounding, and the remaining on day 21. All mice (except the basal control) underwent wound excision under aseptic conditions. Thirty male ICR mice were randomly assigned to the following groups:
Basal Control – No wound or treatment
Experimental – Wound treated with postbiotic-formulated hydrogel
Negative Control 1 – Wound treated with hydrogel base only
Negative Control 2 – Wound without any treatment
Positive Control – Wound treated with DuoDERM hydrogel + povidone-iodine
2.2. Preparation of Postbiotic-formulated Hydrogel
Powdered nipa fronds underwent soda treatment followed by sequential NaOH washing, filtration, bleaching, and drying to yield α-cellulose. Carboxymethylcellulose (CMC) was synthesized from purified cellulose. A chilled alkaline/urea solution (7% NaOH, 12% urea, 81% H2O) was pre-cooled to –12.6°C. CMC (1 g) was added and stirred (1500 rpm, 10 min), followed by centrifugation (8000 rpm, 20 min), neutralization with 10% H2SO4, and autoclaving. L. paracasei 7060 was cultured in MRS broth (35–37°C, 48 hours). Aseptically, 50 mL CMC solution was combined with sterilized carbomer while stirring until homogeneous. Gelation was initiated with triethanolamine, followed by the addition of 50 mL probiotic-inoculated MRS broth. The final hydrogel was transferred into sterile petri dishes and stored at room temperature.
2.3. In Vitro Antibacterial Assay of Postbiotic-formulated Hydrogel
Mueller-Hinton Agar (MHA) was prepared, sterilized, and poured into petri dishes (4–5 mm thickness). Bacterial suspensions of Staphylococcus aureus and Escherichia coli (100 µL) were lawned on separate MHA plates. Four wells were punched per plate and filled with: (1) probiotic hydrogel, (2) probiotic cream, (3) L. paracasei supernatant, and (4) negative control. Plates were incubated at 37°C for 24 and 48 hours. Zones of inhibition were measured using a vernier caliper and interpreted as: strong (>20 mm), intermediate (10–20 mm), or weak (<10 mm).
2.4. In Vivo Wound Healing Assay of Postbiotic-formulated Hydrogel
Thirty healthy male ICR mice (Mus musculus), eight weeks of age and weighing 25–30 grams at the start of the study, were obtained from the Laboratory Animal Care Facility of Mariano Marcos State University (MMSU), Ilocos Norte, Philippines. Animals were housed individually under controlled environmental conditions (22–25 °C; 55–75% relative humidity; 12 hours light/dark cycle) and were provided standard laboratory chow and water ad libitum. A five-day acclimatization period was observed prior to the initiation of experimental procedures. After acclimatization, twenty-four mice were subjected to full-thickness excisional wounds created in the dorso-lumbar region along the midline (vertebral levels L1–L6) using a sterile 1.0 cm biopsy punch. All procedures were performed under topical lidocaine anesthesia. The remaining six mice were maintained as unwounded basal controls.
Treatments commenced 24 hours after wound induction and were administered once daily using sterile applicators, with formulations applied to cover the wound bed and extend approximately 5 mm beyond the wound margins. Animals were assigned to the following groups: (1) experimental group treated with L. paracasei 7060–formulated nipa hydrogel; (2) negative control group receiving hydrogel base only; (3) positive control group treated with DuoDERM hydrogel supplemented with povidone-iodine; (4) untreated negative control group; and (5) basal group (unwounded).
Wound size and gross morphological features were monitored at designated time points. Tissue samples were collected on days 3 and 21 (n = 3 per group per time point) for gene expression analysis. All animal procedures were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Mariano Marcos State University and were conducted in compliance with institutional and national guidelines for the ethical use of laboratory animals.
2.5. Gene Expression Analysis
Gene-specific primers for selected wound healing markers were designed using the Integrated DNA Technologies PrimerQuest tool, based on genomic sequences from the NCBI database. All primers were validated for specificity and amplicon size prior to use in qPCR. Total RNA was extracted from wound tissues using TRIzol Reagent (Invitrogen) and quantified with a ScanDrop2 spectrophotometer (Analytik Jena). Two micrograms of RNA per sample were treated with RQ1 RNase-Free DNase I (Vivantis) to eliminate genomic DNA. qRT-PCR was performed using the SuperScript™ III Platinum™ One-Step qRT-PCR Kit (Invitrogen) on a QuantStudio™ 5 Real-Time PCR System (Applied Biosystems). Thermal cycling was conducted at 95°C for 30 seconds, followed by 39 cycles of 95°C for 5 seconds, 60°C for 30 seconds, and 72°C for 30 seconds. Melt curve analysis confirmed amplification specificity. Relative gene expression was normalized to GAPDH using the ΔCt method. Samples were collected on days 3 and 21 post-wounding, corresponding to the inflammatory and remodeling phases, respectively (Table 1).
2.6. Whole Genome Sequencing
Genomic DNA was extracted from L. paracasei 7060 using the Zymo Quick-DNA™ Fungal/Bacterial Miniprep Kit (Cat. No. D6005), with mechanical disruption performed on the MP Biomedicals FastPrep 96™ system to enhance cell lysis efficiency. DNA quality and yield were assessed using the LightBench® platform, which confirmed the presence of high-molecular-weight DNA at concentrations suitable for long-read sequencing.
High-molecular-weight DNA was subsequently sheared and size-selected following the PacBio SMRTbell library preparation guidelines. End-repair and adapter ligation steps were carried out to generate SMRTbell libraries. Sequencing was performed on the PacBio Sequel IIe system, producing long-read data with read lengths exceeding 10 kb.
Raw subreads were assembled using the Hierarchical Genome Assembly Process (HGAP), resulting in a highly contiguous draft genome assembly. The assembled contigs were then submitted to the Rapid Annotation using Subsystem Technology (RAST) server for automated genome annotation, enabling the assignment of putative functions and subsystem classifications to predicted coding sequences.
2.7. Promoter Prediction and Transcription Factor Binding Site Analysis
Putative promoter elements upstream of selected genes were identified using in silico analysis. Upstream regions (approximately 250 bp) were extracted from the assembled whole genome sequence of L. paracasei 7060. Promoter motifs, including –10 and –35 elements, were predicted using the BPROM tool (Softberry, Inc.), which provided positional information and predicted Transcription Start Sites (TSS). To further identify potential transcription factor binding sites, sequences were analyzed using the Virtual Footprint tool from the PRODORIC database, which screens for conserved regulatory motifs across known bacterial transcription factor binding profiles.
| Gene and ID | Species | Primer Sequence(5' - 3') | Product Size(bp) |
|---|---|---|---|
|
TNF-a 21926 |
Mus musculus L. | F- TTGTCTACTCCCAGGTTCTCT | 107 |
| R- GAGGTTGACTTTCTCCTGGTATG3 | |||
|
TGF-β 21803 |
Mus musculus L. | F- CCTGCAAGACCATCGACAT | 90 |
| R- GACTGGCGAGCCTTAGTTT | |||
|
TLR2 24088 |
Mus musculus L. | F- CACTATCCGGAGGTTGCATATC | 113 |
| R- GGAAGACCTTGCTGTTCTCTAC |
2.8. Histological and Morphometric Assessments
Wound tissues were evaluated histologically using a semi-quantitative grading system adapted from Gupta and Kumar [24]. The assessment focused on key parameters of wound healing, including granulation tissue formation, degree of inflammatory infiltrate, collagen fiber orientation, collagen pattern, and the deposition of early and mature collagen ( Table 2, reproduced from Gupta A, Kumar P. Assessment of the histological state of the healing wound, under the terms of the Creative Commons Attribution 4.0 International License [24]). Histological scoring was performed in a blinded manner to minimize observer bias.
| Number | Histological Parameters |
|---|---|
| 1 | Amount of granulation tissue (profound-1, moderate-2, scanty-3, absent-4) |
| 2 | Inflammatory infiltrate (plenty-1, moderate-2, a few-3) |
| 3 | Collagen fiber orientation (vertical-1, mixed-2, horizontal-3) |
| 4 | Pattern of collagen (reticular-1, mixed-2, fascicle-3) |
| 5 | Amount of early collagen (profound-1, moderate-2, minimal-3, absent-4) |
| 6 | Amount of mature collagen (profound-1, moderate-2, minimal-3) |
Macroscopic wound appearance was graded based on established qualitative criteria. Scores ranged from A (very red, swollen, moist) to F (hair regrowth and complete wound closure).
Digital images of wounds were captured every 24 hours post-treatment. Wound area was measured using ImageJ® software (v1.54p), calibrated with a ruler. Wound contraction rate (%) was computed as (Eq. 1):
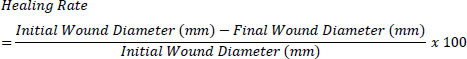
Red, green, and blue (RGB) values of wound areas were extracted from standardized images using ImageJ®, with normalization against a reference image to control for lighting variability. Chromatic red (r) was calculated as (Eq. 2), and luminance (L) was calculated as (Eq. 3):
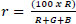
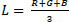
2.9. Data Analysis
Statistical differences among treatment groups were assessed using one-way ANOVA. When significant F-values were observed, Tukey’s Honest Significant Difference (HSD) test was applied for post hoc comparisons. Analyses were performed using SAS software (v9.4; SAS Institute Inc.).
2.10. Ethical Considerations
All procedures involving animals were approved by the Institutional Animal Care and Use Committee (IACUC) of Mariano Marcos State University and conducted in accordance with the ethical guidelines of the National Committee for Research Ethics in Science and Technology (NENT, 2018). Efforts were made to minimize animal suffering and ensure humane care throughout the study.
3. RESULTS
3.1. Morphoanatomical Changes in the Wound under Postbiotic Hydrogel
On day 0, all wounds were of similar size (~10 mm) and exhibited bright red color, characteristic of acute injury. By day 3, the L. paracasei 7060 hydrogel-treated wounds (iv) and DuoDerm-treated wounds (iii) showed early signs of fibrin clotting and color shift to yellow-brown, indicating reduced inflammation. By days 6–9, these same wounds showed significant contraction, scab darkening, and drying, which are morphological hallmarks of wound healing and epithelialization. Full closure was observed in group (iv) by day 12, followed by fur regrowth and minimal scarring between days 15–21. In contrast, groups treated with base hydrogel (ii) and DuoDerm + povidone-iodine (iii) healed more slowly, with persistent scabbing and incomplete closure by day 15. Untreated wounds (i) showed the poorest recovery: open wounds with little fur regrowth even on day 21 (Fig. 1a). Wound contraction was fastest in the L. paracasei 7060 hydrogel and DuoDerm groups, reaching 60–70% reduction by day 9 and complete closure by day 15. Other groups retained more than 50% of wound area by day 9 and showed incomplete healing at day 21 (Fig. 1b).
Color changes were most evident in the L. paracasei hydrogel group (iv). Over the course of healing, these wounds gradually transitioned from a dark reddish-brown appearance to lighter pink hues. In comparison, wounds in the untreated group (i) largely retained darker coloration throughout the observation period, consistent with sustained inflammation or slower remodeling (Fig. 2a). The Chromatic Red Index (CRI) decreased in all treatment groups; however, the most pronounced reduction was observed in group ii between days 6 and 15 (Fig. 2b, top). Luminance values (Fig. 2b, bottom) showed a marked increase in this group after day 15, reflecting increased wound brightness and suggesting ongoing re-epithelization. In contrast, the DuoDerm (iii) and base hydrogel (ii) treatments exhibited only moderate reductions in CRI and comparatively smaller changes in luminance. Tukey’s post hoc analysis confirmed significant differences in both CRI and luminance among treatment groups at several time points, with the strongest effects detected after day 6 (p ≤ 0.01).
3.2. Histological Evaluation of Wound Healing
Histological examination of wound sections harvested on day 21 revealed clear differences in tissue organization and inflammatory status among the treatment groups (Fig. 3a). Healing scores were determined using the semi-quantitative histological grading criteria outlined in Table 2. Wounds treated with the base hydrogel showed pronounced inflammatory cell infiltration and poorly organized granulation tissue. Collagen fibers in this group were predominantly immature and vertically oriented, reflecting limited matrix remodeling. Accordingly, this group received a healing score of 4, consistent with suboptimal tissue regeneration.
In comparison, wounds treated with Duoderm combined with povidone-iodine displayed moderate improvements in tissue architecture. These sections exhibited partial re-epithelization, reduced inflammatory infiltrates, and early signs of extracellular matrix organization, corresponding to a healing score of 8. Interestingly, wounds from the L. paracasei-treated group and the untreated group demonstrated largely comparable histological features. Both groups showed a well-reestablished epidermal layer, evidence of hair follicle regeneration, and minimal residual inflammation, indicative of advanced wound repair. As a result, both groups received high healing scores of 12, reflecting favorable tissue recovery by day 21. Detailed semi-quantitative lesion scores are provided in Supplementary A.
Despite the observed histological improvements associated with probiotic treatment, agar diffusion assays indicated that none of the hydrogel formulations, including the L. paracasei 7060 hydrogel, exhibited detectable antimicrobial activity against Staphylococcus aureus, Escherichia coli, or Candida albicans. In contrast, penicillin-treated positive controls produced distinct zones of inhibition against S. aureus and C. albicans (>20mm), whereas E.coli remained resistant to all treatments, including penicillin (Fig. 3b).
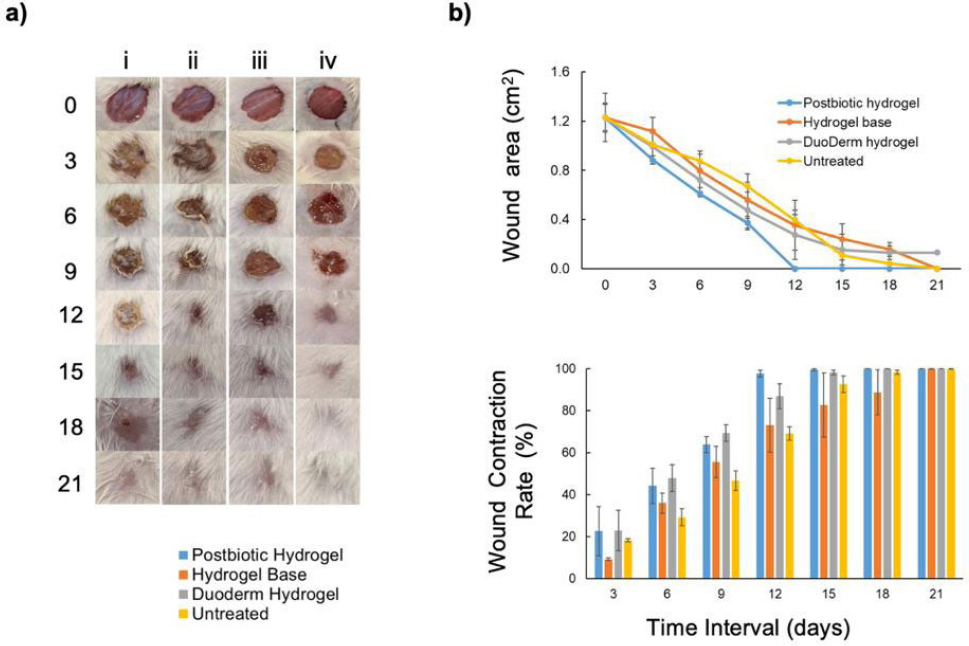
Epithelial tissue repair is accelerated when the wound is treated with a probiotic gel containing L. paracasei 7060. a) Representative photographs of epithelial tissue repair in different treatments. Each column represents a treatment which includes i) untreated, ii) hydrogel base, iii) Duoderm hydrogel with povidone-iodine, and iv) postbiotic hydrogel. b) Comparative wound size and wound contraction of the different treatments in the 21-day observation period. Statistical differences were assessed using ANOVA and Tukey’s multiple comparison test at α = 0.05. Error bars indicate the standard error calculated from biological triplicate. The condition was found to be a significant source of variation at p ≤ 0.01.
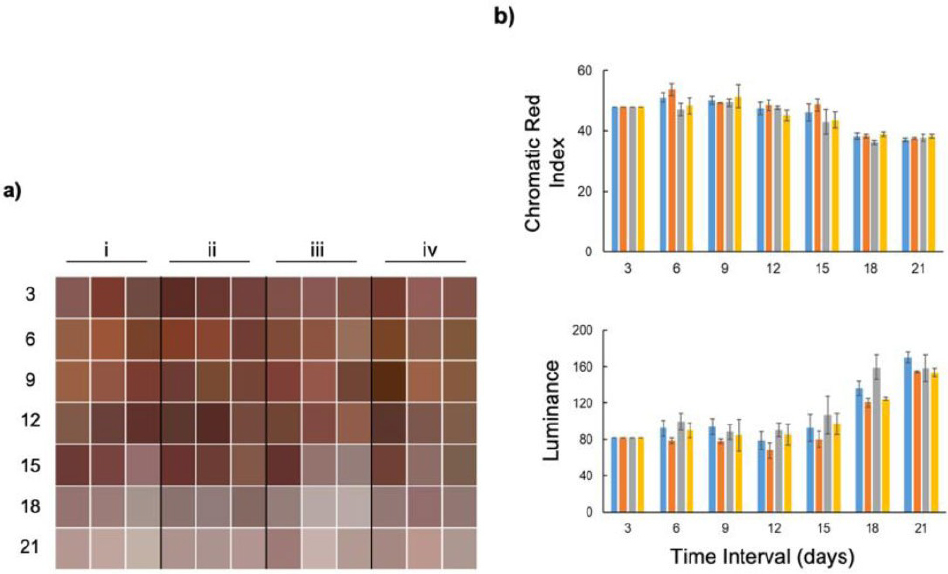
Epithelial tissue repair is accelerated when the wound is treated with a probiotic gel containing L. paracasei 7060. a) Visual representation of color change in wound from day 3 to day 21 (rows) across four treatment groups (i–iv, columns). Each square represents the mean color appearance of replicates at a given time point. Each column represents a treatment which includes i) untreated, ii) hydrogel base, iii) DuoDerm hydrogel, and iv) postbiotic hydrogel. b) Quantitative assessment of color parameters: Top panel, Chromatic Red Index values showing a general decline in redness across all treatments with increasing storage time. Bottom panel, Luminance values indicating a progressive lightening of samples over time, particularly after day 15. Statistical differences were assessed using ANOVA and Tukey’s multiple comparison test at α = 0.05. Error bars indicate the standard error calculated from biological triplicate. The condition was found to be a significant source of variation at p ≤ 0.01.
3.3. Differential Expression of Tissue-repair Associated Genes
The relative expression levels of three key genes involved in inflammation and tissue repair, TLR2, TNF-α, and TGF-β, were assessed in ICR mice on days 3 and 21 post-wounding across four treatment groups: Postbiotic Hydrogel, Hydrogel Base, DuoDerm Hydrogel, and Untreated Control (Fig. 4a).
At day 3, TNF-α expression was significantly elevated across all experimental groups relative to baseline levels. The hydrogel base group exhibited the highest TNF-α expression, surpassing even that observed in the untreated control. In contrast, the postbiotic hydrogel–treated group showed substantially lower TNF-α and TGF-β expression compared with the hydrogel base group, with TNF-α levels falling below those of the untreated control. TLR2 expression was upregulated in all treatment groups at day 3, with the most pronounced increase detected in the postbiotic hydrogel group.
By day 21, expression levels of TNF-α and TLR2 had decreased markedly in most groups, consistent with progression beyond the acute inflammatory phase. The most pronounced reduction in TLR2 expression was observed in the postbiotic hydrogel group, whereas wounds treated with DuoDERM® hydrogel maintained comparatively higher TLR2 levels. In contrast, TGF-β expression remained at moderate levels in the postbiotic hydrogel group, while more substantial downregulation was observed in the other treatment groups.
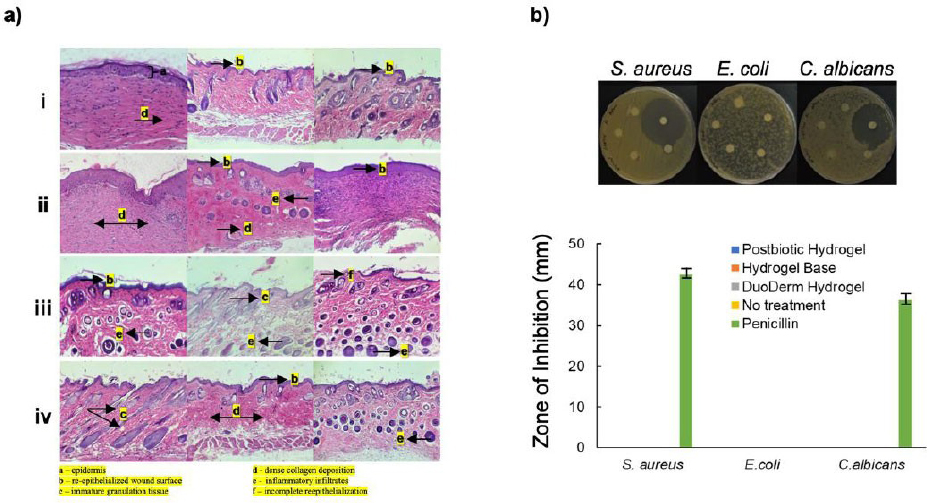
Histological characteristics of epithelial tissues in the dorso-lumbar (midline over L1-L6) 21 days post-treatment and antimicrobial effects of postbiotic hydrogel treatment. a) Representative photomicrographs of H&E-stained histological sections showing epithelial tissue repair across treatment groups: i) postbiotic hydrogel, ii) hydrogel base, iii) DuoDerm hydrogel with povidone-iodine, and iv) untreated control. b) antimicrobial activity of treatments against Staphylococcus aureus, Escherichia coli, and Candida albicans. Top panel: Representative images of disk diffusion assay; bottom panel: quantitative assessment of the inhibition zones. Statistical differences were assessed using ANOVA and Tukey’s multiple comparison test at α = 0.05. Error bars indicate the standard error calculated from biological triplicate. The condition was found to be a significant source of variation at p ≤ 0.01.
At three days post-wounding, L. paracasei 7060 demonstrated a modulatory effect on the inflammatory response, facilitating the transition toward the early proliferative phase of wound healing. As illustrated in Fig. (4b), L. paracasei 7060 was shown to interact with TLR2, leading to pronounced TLR2 activation, a recognized indicator of innate immune engagement. This interaction was associated with downstream activation of the NF-κB signaling pathway, which subsequently supported the transcription of immune response-related genes. Activation of this pathway did not translate into excessive inflammatory signaling. Instead, TNF-α expression appeared to be tightly regulated, suggesting a controlled pro-inflammatory response that is conducive to effective tissue repair rather than sustained tissue damage. At the same time, an increase in TGF-β expression was observed, consistent with the initiation of the transition from the inflammatory phase toward the early proliferative stage of wound healing. These molecular signatures are indicative of a controlled immune modulation by L. paracasei 7060, supporting its role in enhancing wound healing dynamics (Fig. 4b).
3.4. Whole Genome Sequence (WGS) Analysis of L. paracasei 7060
Whole genome analysis of L. paracasei 7060 revealed a diverse set of genes linked to the production of bioactive metabolites and cellular components involved in antimicrobial activity, immune modulation, and tissue repair (Fig. 5b). In particular, the genome contained biosynthetic gene clusters encoding bacteriocins, lactic acid, Short-Chain Fatty Acids (SCFAs), NADH, and Lipoteichoic Acid (LTA) (Fig. 5a and 5b). The identification of bacteriocin-related genes supports the strain’s capacity to produce antimicrobial peptides that may contribute to the inhibition of pathogenic bacteria. Genes associated with lactate and acetate biosynthesis were also detected, indicating a potential role in promoting angiogenesis and reinforcing epithelial barrier function, respectively. In addition, the presence of NADH-producing genes suggests a contribution to cellular redox balance and mitigation of oxidative stress within the wound microenvironment. Genes involved in lipoteichoic acid biosynthesis were likewise identified and are known to participate in host immune modulation through TLR2-mediated signaling.
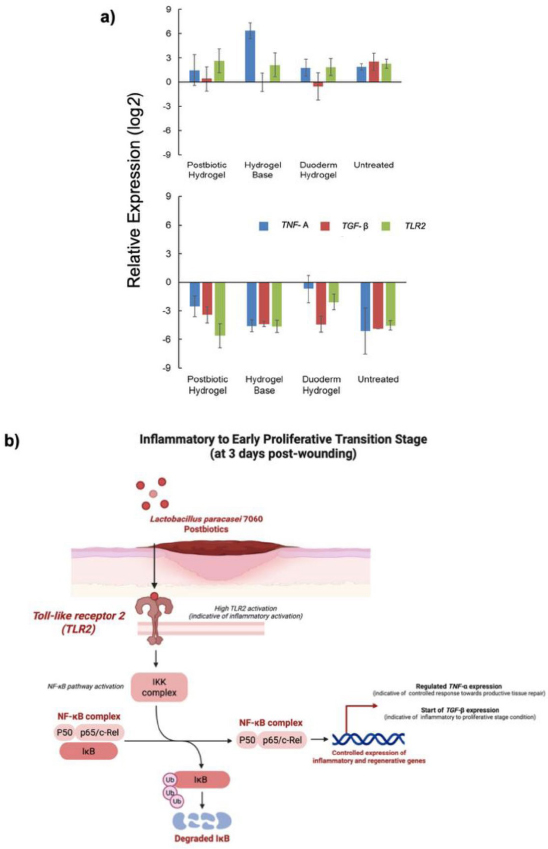
Gene expression and involvement in tissue repair. a) Relative gene expression levels (log2 fold change) of pro- and anti-inflammatory markers: Tumor Necrosis Factor alpha (TNF-α), Transforming Growth Factor beta (TGF-β), and Toll-Like Receptor 2 (TLR2) in treated wounds. The top panel shows upregulation; the bottom panel shows downregulation of gene expression across treatment groups. b) Schematic representation of L. paracasei 7060-mediated modulation of the inflammatory to early proliferative transition phase at 3 days post-wounding. Statistical differences were assessed using ANOVA and Tukey’s multiple comparison test at α = 0.05. Error bars indicate the standard error calculated from biological triplicate. The condition was found to be a significant source of variation at p ≤ 0.01.
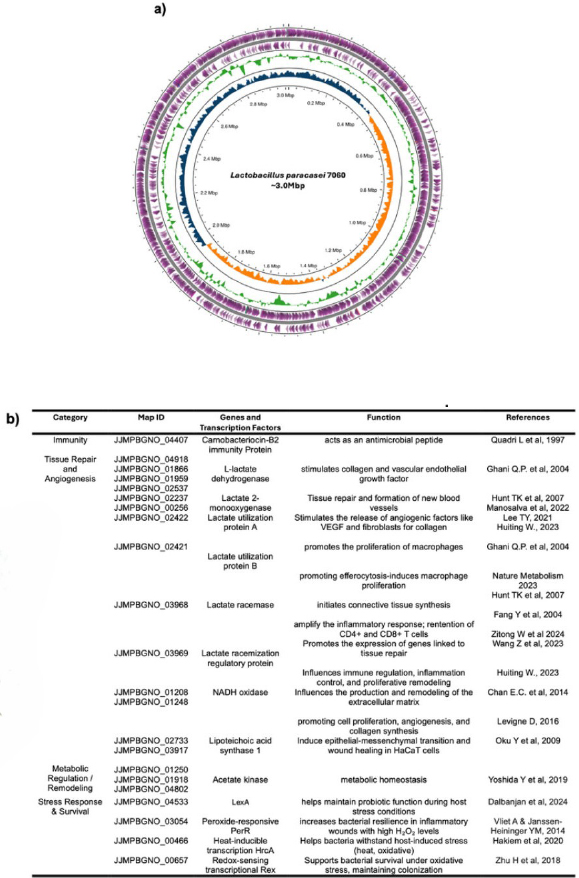
(a) Circular genome map of L. paracasei 7060. The outermost rings represent predicted coding sequences (CDSs) on the forward (outermost, purple) and reverse (second outermost, purple) strands. The next two inner rings (green) display the locations of rRNA and tRNA genes. The blue and orange rings show the GC content and GC skew. (b) Functional classification of genes and transcription factors in L. paracasei 7060 associated with the biosynthesis of metabolites potentially involved in tissue repair.
Beyond metabolic functions, the genome also harbored genes associated with stress adaptation and host interaction. Among these, trxA, encoding thioredoxin, is implicated in redox regulation and protection against Reactive Oxygen Species (ROS). The detection of msoR and gshA, which are involved in metal sensing and glutathione biosynthesis, respectively, further points to roles in detoxification processes and epithelial protection. Genes encoding the spaCBA pilus cluster were present, suggesting a potential mechanism for adhesion to host epithelial surfaces. Multiple copies of ef-Tu were also observed; this protein is recognized for its moonlighting functions, including interactions with host pattern recognition receptors and modulation of immune responses. The genome map (Fig. 5a) illustrates the circular architecture of the L. paracasei 7060 chromosome, highlighting coding sequences (CDSs), rRNA and tRNA loci, as well as regions of GC content and GC skew.
4. DISCUSSION
The integration of phenotypic, histological, gene expression, and genomic data in this study provides converging evidence supporting the pro-regenerative potential of a postbiotic hydrogel formulated with Lactobacillus paracasei 7060. The findings indicate that treatment with this hydrogel markedly accelerated tissue repair, as demonstrated by complete wound closure by day 7 and full re-epithelialization by day 21. These outcomes suggest that microbial-derived metabolites present in the postbiotic formulation actively modulate host immune responses and repair mechanisms rather than merely providing passive wound coverage. Similar regenerative effects have been reported for probiotics and their postbiotic derivatives, which have been shown to promote epithelial regeneration, enhance collagen deposition, and regulate inflammatory responses during wound healing [25-27].
In the present study, wounds treated with the L. paracasei 7060 postbiotic hydrogel exhibited clear improvements in surface characteristics, including color, luminance, homogeneity, smoothness, and consistency. These parameters are widely recognized as indicators of accelerated and high-quality tissue repair [28]. By day 21, complete re-epithelialization was accompanied by histological evidence of increased collagen deposition, likely involving collagen types I and III, as well as enhanced angiogenesis [29]. Both collagen remodeling and neovascularization are central to wound maturation and are essential for restoring tissue integrity, tensile strength, and long-term functionality [30, 31]. Phenotypic differences observed at earlier stages were not reflected histologically by day 2 [32], suggesting that the most dynamic phase of tissue regeneration may have occurred earlier in the healing timeline, potentially around day 12[33.34]. This interpretation is consistent with previous reports indicating that, by day 21, wound resolution is generally underway even in untreated wounds, although the quality of healing, including scar formation and tissue organization, remains strongly influenced by early inflammatory and reparative events [35, 36].
At the molecular level, the postbiotic hydrogel induced coordinated changes in the expression of genes involved in inflammation and tissue remodeling. Early downregulation of the pro-inflammatory cytokine TNF-α and the pro-fibrotic mediator TGF-β suggests attenuation of excessive inflammatory signaling during the initial repair phase. TNF-α is a key driver of acute inflammation, and its suppression is associated with reduced leukocyte infiltration, diminished secondary tissue damage, and improved healing outcomes [37, 38]. Likewise, although TGF-β plays a critical role in extracellular matrix deposition and wound remodeling, sustained or excessive expression of this cytokine can promote pathological fibrosis and impair normal tissue regeneration [39]. The reduced expression of both TNF-α and TGF-β early in the repair process, therefore, points toward a healing trajectory that favors regeneration over fibrosis, ultimately contributing to improved tissue architecture and function [40].
In contrast, Toll-Like Receptor 2 (TLR2) expression was significantly upregulated during the early stages of healing, followed by a gradual decline at later time points. Activation of TLR2 by microbial ligands, including lipoteichoic acids derived from Gram-positive bacteria, is known to stimulate innate immune responses and support tissue repair when appropriately regulated [41-43]. The observed pattern of early induction followed by controlled downregulation may have created a transient window of immune activation sufficient to promote pathogen clearance, macrophage recruitment, and early repair signaling, while preventing prolonged inflammation. Such temporal regulation of TLR signaling is increasingly recognized as a hallmark of optimal wound healing responses [44]. The coordinated dynamics observed between TLR2, TNF-α, and TGF-β suggest that the postbiotic hydrogel fosters an immunological environment conducive to rapid inflammation resolution and effective tissue remodeling, potentially minimizing long-term scarring. This profile aligns well with current wound healing paradigms emphasizing early inflammatory control, timely macrophage polarization, and structured extracellular matrix reorganization [45].
Genomic characterization of L. paracasei 7060 further supports the biological plausibility of these observed effects. The genome harbors genes encoding enzymes involved in the production of lactic acid and Short-Chain Fatty Acids (SCFAs), including acetate, metabolites with well-established roles in inflammation modulation and tissue regeneration. Lactate has been shown to promote angiogenesis by stimulating endothelial cell proliferation and Vascular Endothelial Growth Factor (VEGF) expression [46], while acetate contributes to epithelial barrier reinforcement and immune homeostasis through signaling pathways involving G-protein-coupled receptors such as GPR43 [47]. In addition, the presence of genes linked to NADH biosynthesis suggests a capacity for antioxidant support, which is particularly relevant in the highly oxidative environment of acute and chronic wounds. NADH functions as a critical cofactor for glutathione reductase and other redox enzymes, helping to counteract Reactive Oxygen Species (ROS)-mediated damage and maintain cellular viability during tissue repair [48, 49].
Genes associated with lipoteichoic acid (LTA) biosynthesis were also identified, providing a potential molecular basis for the observed modulation of TLR2 signaling [50]. Controlled interactions between LTA and TLR2 are known to activate beneficial immune pathways without triggering excessive inflammation, thereby supporting macrophage-mediated tissue repair processes [51]. Furthermore, the presence of antioxidant-related genes such as trxA, msoR, and gshA, encoding components of the glutathione system, underscores the strain’s potential to maintain redox balance and cellular homeostasis under stress conditions, which are factors that are critical not only for microbial resilience but also for epithelial healing [52, 53].
The identification of the spaCBA gene cluster, which encodes pilus-associated proteins involved in adhesion, suggests that close microbial–host interactions may facilitate localized delivery of bioactive metabolites and enhance targeted immune modulation at epithelial surfaces [54]. Additionally, the presence of multiple copies of ef-Tu introduces another layer of immunomodulatory potential, as this moonlighting protein has been reported to interact with host pattern recognition receptors and influence cytokine expression profiles [55, 56]. Taken together, these genomic features provide a mechanistic framework explaining how postbiotics derived from L. paracasei 7060 may orchestrate a multifaceted regenerative response.
The findings of this study demonstrate that L. paracasei 7060-derived postbiotics promote wound healing through coordinated suppression of excessive early inflammation, enhancement of antioxidant defenses, stimulation of angiogenesis, and support of epithelial regeneration. The integration of phenotypic observations with molecular and genomic data strengthens the case for this strain as a promising candidate for future wound care applications, including hydrogel-based delivery systems, tissue-engineering scaffolds, or topical formulations. Future investigations should aim to validate these effects in large animal models and human skin equivalents, as well as to delineate the individual contributions of specific microbial metabolites to tissue repair. Longitudinal analyses of wound-associated microbiome dynamics and immune cell phenotypes would further advance understanding of the complex host–microbe interactions that shape wound healing outcomes.
CONCLUSION
This study provides evidence for the wound healing potential of a postbiotic hydrogel derived from L. paracasei 7060. Treatment with the formulation accelerated wound closure, enhanced collagen deposition, and promoted complete re-epithelialization, reflecting an optimized balance between inflammation and regeneration. The coordinated molecular and phenotypic outcomes highlight L. paracasei 7060 as a promising source of bioactive postbiotics for wound healing applications. Further studies using advanced preclinical models will be important to confirm these effects and to clarify the specific roles of individual microbial metabolites in driving tissue regeneration.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contribution to the paper as follows: A.G.B.P., J.Z.P.F., M.J.T.T.A., A.D.: Methodology; A.G.B.P.: Data collection; A.G.B.P., J.Z.P.F., J.P.A.A.: Analysis and interpretation of results; A.G.B.P., J.Z.P.F., P.J.I.G.: Draft manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| ANOVA | = Analysis of Variance |
| bp | = Base Pair |
| cDNA | = Complementary DNA |
| CMC | = Carboxymethylcellulose |
| CRD | = Completely Randomized Design |
| CRI | = Chromatic Red Index |
| DNA | = Deoxyribonucleic Acid |
| ECM | = Extracellular Matrix |
| ef-Tu | = Elongation Factor Tu |
| GAPDH | = Glyceraldehyde-3-Phosphate Dehydrogenase |
| GC | = Guanine–Cytosine |
| gsha | = Glutathione Synthetase Gene |
| H&E | = Hematoxylin and Eosin |
| HSD | = Honest Significant Difference |
| IACUC | = Institutional Animal Care and Use Committee |
| IDT | = Integrated DNA Technologies |
| IL | = Interleukin |
| ISAPP | = International Scientific Association for Probiotics and Prebiotics |
| LAB | = Lactic Acid Bacteria |
| LTA | = Lipoteichoic Acid |
| MHA | = Mueller-Hinton Agar |
| MRS | = de Man, Rogosa, and Sharpe (culture medium) |
| msor | = Metal-Sensing Oxidoreductase Gene |
| NCBI | = National Center for Biotechnology Information |
| NF-κb | = Nuclear Factor Kappa B |
| NENT | = National Committee for Research Ethics in Science and Technology |
| PBS | = Phosphate-Buffered Saline |
| qPCR | = Quantitative Polymerase Chain Reaction |
| RAST | = Rapid Annotation using Subsystem Technology |
| RGB | = Red–Green–Blue (color analysis) |
| RNA | = Ribonucleic Acid |
| ROS | = Reactive Oxygen Species |
| RPM | = Revolutions Per Minute |
| SCFA | = Short-Chain Fatty Acid |
| SMRTBELL | = Single Molecule Real-Time Bell (PacBio library format) |
| TGF-β | = Transforming Growth Factor-Beta |
| TLR2 | = Toll-Like Receptor 2 |
| TNF-α | = Tumor Necrosis Factor-Alpha |
| TSS | = Transcription Start Site |
| trxa | = Thioredoxin |
| VEGF | = Vascular Endothelial Growth Factor |
| WGS | = Whole Genome Sequencing |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Institutional Animal Care and Use Committee (IACUC) of Mariano Marcos State University under protocol number 2024-001. In addition, animal research clearance was secured from the Bureau of Animal Industry of the Philippines under reference number AR-2024-0505.
HUMAN AND ANIMAL RIGHTS
This study adheres to internationally accepted standards for animal research, following the 3Rs principle. The ARRIVE guidelines were employed for reporting experiments involving live animals, promoting ethical research practices.
All procedures involving animals were conducted in accordance with the ethical guidelines of the National Committee for Research Ethics in Science and Technology (NENT, 2018).
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this published article.
FUNDING
This study was supported by the Commission on Higher Education (CHED), Philippines, with project number LAKAS 2022-005.
ACKNOWLEDGEMENTS
The authors are thankful to the CHED Philippines for supporting the project. The authors are also thankful to Dr. Shirley C. Agrupis, Dr. Dionisio S. Bucao, and Dr. Rhian Jaymar D. Ramil for their administrative support.


