All published articles of this journal are available on ScienceDirect.
Single Cell Protein for Foods and Feeds: A Review of Trends
Abstract
Introduction:
Predictions on the world’s population in the next few decades suggest that the global demand for animal-derived proteins may not be met if current conventional agriculture approaches are used. One promising solution to this complex crisis lies in the use of single-cell proteins (SCP). SCP refers to the edible biomass of unicellular microorganisms and can be developed as animal feeds or human foods. This paper provides a detailed overview on research towards the production and utilisation of SCPs and trends within the field.
Study Design:
A bibliometric based study was conducted on 425 SCP research articles collected from the Web of Science database, analysing the most cited papers using VOSviewer software, and contributing authors, affiliations and country of origin. Research publications on SCP started in 1961 and has grown steadily over the years.
Discussion:
Emerging research topics within SCP production focused on the use of improved fungal strains, the composition and characteristics of SCPs based on the type of substrates used, industrial production processes and the use of waste for SCP production, which serves the dual purpose of mitigating the cost associated with waste disposal and production of a valuable product.
1. INTRODUCTION
It is estimated that by 2050, the world’s population will exceed 9 billion people [1]. Based on current consumption trends, the world will need to produce 1,250 million tonnes of meat and dairy products yearly to meet the global demand for animal-derived proteins with this increased population size [2]. However, given the current rate of food production, this increased demand cannot be met with conventional agriculture approaches currently in use. Thus, there is a need for alternate approaches to achieve sustainable development goals focused on food security. A promising approach is the use of single-cell proteins.
Single-cell proteins (SCP) generally refer to the edible biomass of unicellular microorganisms. This can be the whole biomass or protein extracts from single or mixed cultures of various microbial groups, including algae, bacteria, yeasts/fungi and others [3]. Single-cell proteins can be utilised as animal feeds or human foods and consumed whole or applied as supplements. There is growing interest in the utilisation of SCP to meet the global demand for nutritious food as they have various advantages over conventional plant and animal proteins [4]. These benefits include; their ease of production, which requires a very short period of time, compared to the time interval of weeks, months or years required for crop farming and animal husbandry. In addition, SCPs are cost-effective as they can be grown in bioreactors. Therefore, they do not require extensive land use or high-water demand that is synonymous with conventional agriculture [5]. Furthermore, the industrial production of SCP in bioreactors ensures uniformity of products and high yields because the product does not compete with pests and weeds. Importantly, SCPs are not prone to diseases associated with conventional agricultural efforts which are responsible for a significant cost in crop farming and animal production with loss in products. In addition, SCP production does not result in biodiversity loss, environmental degradation or contribute to greenhouse gases and climate change [6].
Agricultural waste is continually generated in the food sector and generally are expensive to dispose of [7]. Single-cell protein can be produced using these wastes. This conversion of wastes to foods not only reduces wastage and pollution but ensures that increased demand for food from the ever-growing world population will be met using SCP which has minimal carbon fingerprint [7]. Furthermore, with the global increase in agricultural wastes, it is imperative to devise innovative approaches to dispose of these waste products to safeguard environmental and public while saving cost. Thus, the ability to convert inorganic substrates such as methane, petroleum waste and CO2 to biomass offers an incredible sustainability potential that is not currently available using conventional agriculture.
Although the current production and consumption of SCP only account for a small percentage of protein intake for humans, the growing demand for protein is likely to make SCPs increasingly important [8, 9]. For animal feed, there is a greater diversity of microbial species which can be used as SCP, and they can be prepared from varied substrates, including wastes. This is unlike SCP for human consumption, which must be prepared from food-grade substrates. This is because single-cell proteins for animal feed are not held to the same regulatory standards as those destined for human consumption, which requires more expensive substrates, product controls, and monitoring. Because of this factor, it may be more advantageous and cost-effective to produce single-cell proteins from diverse substrates such as inexpensive waste materials from forestry and agricultural sources, food and beverage processing [10] as well as other non-food grade sources and use these as animal feeds. This will reduce demand for foliage and feed formulations from edible foods for animals.
Over the years, research on SCP has grown, with contributions across the globe. This study aims to review the body of research on SCP using a bibliometric and bibliographic approach, highlighting commercialisation efforts in SCP production and industry developments. Bibliometric indicators such as the most cited authors, countries, and institutions are discussed. A critical analysis of the production processes and microorganisms used for SCP production, feedstock, biotechnological improvements and potential uses was undertaken. The information provided in this review will guide researchers on recent developments in SCP production and help in harmonising industry and laboratory research towards the use of SCP for food and feed. It will also support researchers within the subject area in fostering collaborations with more experienced research teams/groups in different countries and institutions.
2. STUDY DESIGN
2.1. Database
The Web of Science database was searched for articles related to single-cell proteins. Web of Science was chosen because of its extensive coverage, ease of use, citation records, and inbuilt tools that facilitate easy data analysis with interdisciplinary coverage. Furthermore, it indexes reputable peer-reviewed cited journals, with a complete index of authors, their affiliations and bibliographic reference [11]. A focused title search was conducted using the keywords (“single cell protein” OR “single-cell protein” OR “microbial proteins” OR “SCP”) for only research articles in the English language. The title search retrieves only publications with the keywords in their titles, allowing the retrieval of only articles directly related to SCP. Furthermore, the Boolean operators (“ ”) were used to obtain matching keywords only. Research articles were chosen because they represent an original contribution to the field and indicate progress and trends in single-cell protein research.
2.2. Inclusion Criteria
The inclusion criteria are research articles related to single-cell proteins and published in the English Language. Related research not in the English Language or other types of publications was excluded.
2.3. Article Categorisation
All retrieved articles were exported to Endnote Online, and their abstracts were evaluated individually by two independent researchers to categorise them based on the class of microorganisms employed in the production of single-cell proteins. Research articles were grouped into; bacteria, fungi, algae and others. Articles not related to single-cell proteins were manually deleted. All vetted articles were preliminarily categorised into different research groups using the Web of Science analytical tools. Further analysis was undertaken to determine participating institutions, publishing journals and countries. Finally, trends in research output over time were analysed using Microsoft Excel, and the articles were arranged based on the total number of citations. VosViewer Software [12] was used to visualise the co-occurrence of keywords used in the retrieved articles. These indicate relevant keywords that can be used for retrieving articles related to single-cell protein research.
3. RESEARCH TREND IN SINGLE CELL PROTEINS
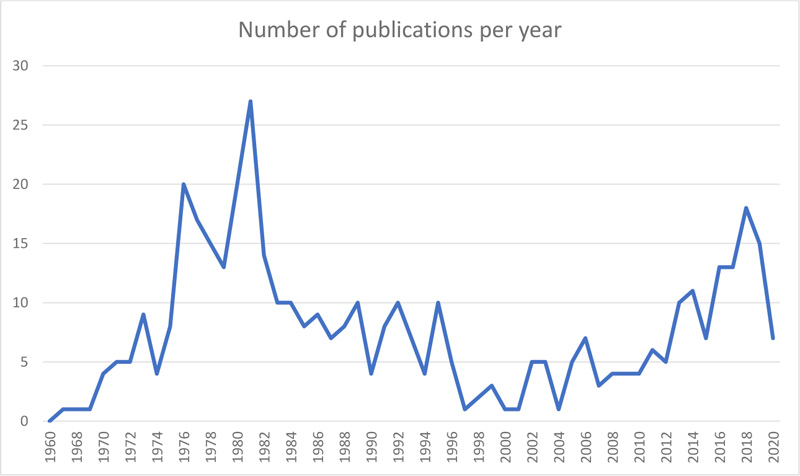
A total of 425 published articles were retrieved from the Web of Science Database. The first recorded publication in single-cell protein was by Mateles et al. [13], which focused on studying the growth of the thermophilic bacterium Bacillus stearothermophilus on hydrocarbons and the use of this as a new source of SCP. There was a gradual increase in publications over the next two decades, with a peak in 1981 of 27 papers (Fig. 1). This growth was followed by a fluctuation in the number of publications over the next four decades, with a second peak in 2018 when 17 papers were published. Overall, an average number of 7 publications per year was made over the 60 years period. Although a limitation of this study is searching and using only articles in which the selected keywords appeared in the title, this is necessary to screen and obtain only articles related and relevant to SCP research. From this, the data obtained suggests that despite the length of time SCP has been a focus of research, there has been no significant growth in the number of publications over the last six decades. More scientific research is required to improve understanding of growth requirements and process conditions for SCP production and to position SCP as a viable protein source to help in meeting the global demand for protein.
3.1. Categorisation of Research
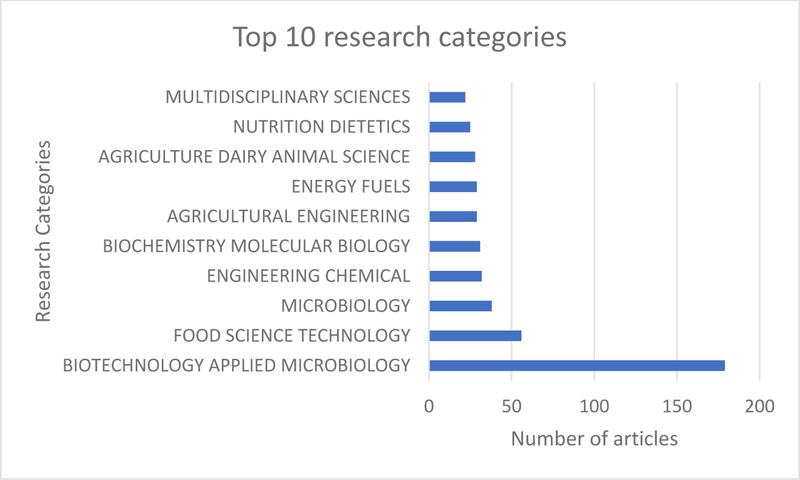
Research on the production and utilisation of SCP for food and feeds is growing. Retrieved articles highlighted the major themes within SCP research. These include the different microbial species utilised for SCP production, substrates used and process optimisation. The retrieved articles are categorised mainly into the biotechnology and applied microbiology, food science and technology and chemical engineering subject areas, as shown in Fig. (2). The highest number of published articles were in Biotechnology and Applied Microbiology. This category represents the principal research focus directed to discovering suitable microorganisms for use in SCP production, optimisation of fermentation, and growth conditions necessary for the production of high biomass. The second-highest category is Food Science and Technology, with articles published under this category focusing on the nutritional composition and safety of produced SCPs. Furthermore, research under this category investigated the formulation of suitable food products from SCP into edible food and feed for human and animal consumption by the supplementation of existing food products with portions of single-cell proteins.
3.2. Producing Organisms
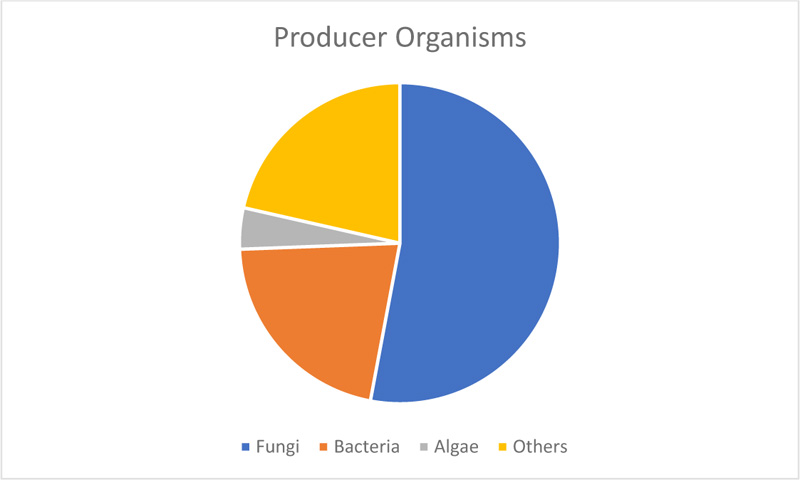
Analysis of retrieved research articles on SCP showed that of the total published articles, fungi were utilised in 242 articles, algae (19), and bacteria (98) amongst others, for the production of single-cell proteins, as shown in Fig. (3). Of the Fungi genera, the predominant organisms were Candida lipolytica, Candida tropicalis and Kluyveromyces marxianus, which were employed as mixed cultures to produce single-cell proteins from various food wastes. Bacteria species employed are mainly Bacillus species and methanotrophic bacteria [14], while marine algae widely employed include Chlorella sp. and Euglena gracilis [15, 16].
3.3. Publication by Countries
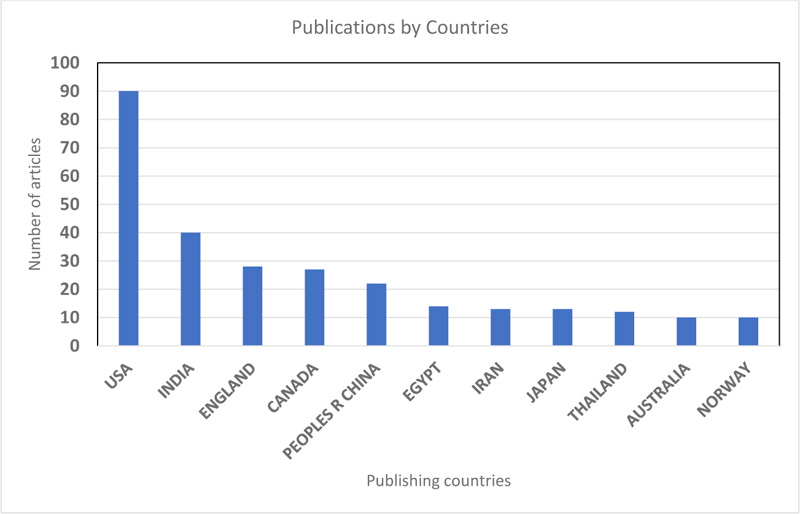
A total of 62 countries have participated in SCP based research (Fig. 4). An article is assigned to a country based on the affiliation of the first author. The country with the highest number of publications was the United States of America with 90 publications, followed by India (40) and England (28). Within the last ten years (2010-2020), The United States of America had 20 publications, followed by The Peoples Republic of China (18), Iran and Thailand (8 each). Publications from the United States of America focused mainly on the modelling and validation of the production processes of single-cell proteins for food uses and the genetics of single-cell protein in yeast populations [17-19]. China’s research focus is mainly on the volarization of food wastes for single-cell protein production, making use of indigenous micro-flora [20-22] and the quantification of single-cell protein production [23]. Interestingly, England and Norway were the top publishing countries amongst other European nations. Although in Europe, there is high consumption of proteins at about 70% above recommended levels (Westhoek et al., 2011), there is low research output on alternative protein sources such as SCP, which is more sustainable and economically viable in Europe (Voutilainen et al., 2021). Africa, Asia-Pacific and South American regions have low participation in SCP research.
| S.No | Institution | Publications | Percent |
|---|---|---|---|
| 1 | Dalhousie University | 10 | 2.35% |
| 2 | Iowa State University | 10 | 2.35% |
| 3 | Massachusetts Institute of Technology MIT | 9 | 2.12% |
| 4 | Council of Scientific Industrial Research CSIR india | 8 | 1.88% |
| 5 | Cornell University | 7 | 1.65% |
| 6 | National Research Centre NRC | 7 | 1.65% |
| 7 | Ocean University of China | 7 | 1.65% |
| 8 | University of California System | 7 | 1.65% |
| 9 | Norwegian University of Life Sciences | 6 | 1.41% |
| 10 | Punjab Agricultural University | 6 | 1.41% |
| 11 | Technical University of Denmark | 6 | 1.41% |
| 12 | University of Quebec | 5 | 1.18% |
3.4. Publication by Institutions
Similar to country participation in single-cell protein publications, many institutions (413) were associated with publications in this field of research. Institutions with five publications and above are shown in Table 1. The three most productive research institutions are based in the United States. In addition, of the top 12 institutions, five are in the United States. This finding is consistent with the data on country participation in which the United States of America had the highest publication output.
3.5. Publications by Journals
A total of 217 journals were involved in SCP related research (Table 2). Biotechnology and Bioengineering Journal had the highest number of articles (30). It is of note that these articles were published between the years 1970 and 1985. This was followed by Bioresource Technology with 19 articles. Finally, the highest number of publications from the last ten years was published by Bio-resource Technology, Scientific Reports and Springer Briefs in Molecular Sciences with five publications each.
Table 2.
| S.No | Journal | Publications | Percent |
|---|---|---|---|
| 1 | Biotechnology and Bioengineering | 30 | 7.06% |
| 2 | Bioresource Technology | 19 | 4.47% |
| 3 | Applied Biochemistry and Biotechnology | 10 | 2.35% |
| 4 | Journal of Food Science | 10 | 2.35% |
| 5 | Biotechnology Letters | 9 | 2.12% |
| 6 | Nutrition Reports International | 8 | 1.88% |
| 7 | Applied and Environmental Microbiology | 7 | 1.65% |
| 8 | Applied Microbiology and Biotechnology | 7 | 1.65% |
| 9 | Journal of Animal Science | 7 | 1.65% |
| 10 | Process Biochemistry | 7 | 1.65% |
| 11 | Biomass Bioenergy | 6 | 1.41% |
| 12 | British Journal of Nutrition | 6 | 1.41% |
| 13 | Journal of Fermentation Technology | 6 | 1.41% |
| 14 | World Journal of Microbiology Biotechnology | 6 | 1.41% |
| 15 | European Journal of Applied Microbiology and Biotechnology | 5 | 1.18% |
| 16 | Food Technology | 5 | 1.18% |
| 17 | Journal of the Science of Food and Agriculture | 5 | 1.18% |
| 18 | Scientific Reports | 5 | 1.18% |
| 19 | Single Cell Protein Production from Lignocellulosic Biomass | 5 | 1.18% |
| 20 | Springer Briefs in Molecular Science | 5 | 1.18% |
3.6. Top Cited Publications
A search of the most prominent publications showed that 16 papers had citations of 50 and above. These are highlighted in Table 3. The most impactful paper in SCP research investigated the improved conversion of methanol to SCP by Methylophilus methylotrophus [24]. The next most cited article discussed SCP production using Euglena gracilis and carbon dioxide fixation in an innovative photo-bioreactor [15]. In this study, a kinetic model was used to determine the relationship between specific growth rate and light intensity to optimise the growth of Euglena gracilis, this yielded SCP with high protein content and digestibility for animal feed. About half of the highly cited papers are from the earlier years (1970 to 1985) of SCP research, while the remaining half is from the last decade. Although older papers are more likely to be cited more, as they have been available for longer, the trend in citation indicates a resurgence of interest in SCP research.
3.7. Keyword Analysis
Keyword analysis shows that a total of 1207 keywords were used to describe research in this field. Expectedly, the keywords; single, cell, and protein were used in all publications. This is followed by production (189) times, yeast (41), waste (35) and whey (27). The spread of keywords used indicates the focus of research within the field. The co-occurrence of keywords that have occurred a minimum of 5 times is visualised using VosViewer in Fig. (5).
| S/N | Author | Title | Citations | Journal | Year | Ref |
|---|---|---|---|---|---|---|
| 1 | Windass JD, Worsey MJ, Pioli EM, Pioli D, Barth PT, et al. | Improved conversion of methanol to single-cell protein by Methylophilus methylotrophus | 157 | Nature | 1980 | [24] |
| 2 | Chae SR, Hwang EJ, Shin HS | Single cell protein production of Euglena gracilis and carbon dioxide fixation in an innovative photo-bioreactor | 117 | Bioresource Technology | 2006 | [15] |
| 3 | Gefen O, Gabay C, Mumcuoglu M, Engel G, Balaban NQ | Single-cell protein induction dynamics reveals a period of vulnerability to antibiotics in persister bacteria | 96 | Proceedings of the National Academy Of Sciences of the United States of America | 2008 | [25] |
| 4 | Cysewski G.R. and Wilke C.R | Utilisation of cellulosic materials through enzymatic hydrolysis. I. Fermentation of hydrolysate to ethanol and single-cell protein | 85 | Biotechnology and Bioengineering | 1976 | [26] |
| 5 | Papanikolaou S, Chevalot I, Galiotou-Panayotou M, Komaitis M, Marc I, et al. | Industrial derivative of tallow: a promising renewable substrate for microbial lipid, single-cell protein and lipase production by Yarrowia lipolytica | 73 | Electronic Journal of Biotechnology | 2007 | [27] |
| 6 | Howells ER | Single-cell protein and related technology | 68 | Chemistry & Industry | 1982 | [28] |
| 7 | Shahi P, Kim SC, Haliburton JR, Gartner ZJ, Abate AR | Abseq: Ultrahigh-throughput single cell protein profiling with droplet microfluidic barcoding | 67 | Scientific Reports | 2017 | [29] |
| 8 | Albert FW, Treusch S, Shockley AH, Bloom JS, Kruglyak L | Genetics of single-cell protein abundance variation in large yeast populations | 64 | Nature | 2014 | [19] |
| 9 | Ferrer J, Paez G, Marmol Z, Ramones E, Garcia H, et al. | Acid hydrolysis of shrimp-shell wastes and the production of single cell protein from the hydrolysate | 61 | Bioresource Technology | 1996 | [30] |
| 10 | Shipman RH, Kao IC, Fan LT | Single-cell protein production by photosynthetic bacteria cultivation in agricultural by-products | 56 | Biotechnology and Bioengineering | 1975 | [31] |
| 11 | Humphrey AE, Moreira A, Armiger W, Zabriskie D | Production of single cell protein from cellulose wastes | 55 | Biotechnology and Bioengineering | 1977 | [32] |
| 12 | Moon NJ, Hammond EG, Glatz BA | Conversion of cheese whey and whey permeate to oil and single-cell protein | 55 | Journal of Dairy Science | 1978 | [33] |
| 13 | Avnimelech Y, Mokady S, Schroeder GL | Circulated ponds as efficient bioreactors for single cell protein production | 52 | Israeli Journal of Aquaculture-Bamidgeh | 1989 | [34] |
| 14 | Aggelopoulos T, Katsieris K, Bekatorou A, Pandey A, Banat IM, et al. | Solid state fermentation of food waste mixtures for single cell protein, aroma volatiles and fat production | 52 | Food Chemistry | 2014 | [35] |
| 15 | Revah-moiseev S, Carroad PA | Conversion of the enzymatic hydrolysate of shellfish waste chitin to single-cell protein | 50 | Biotechnology and Bioengineering | 1981 | [36] |
| 16 | Bothe H, Jensen KM, Mergel A, Larsen J, Jorgensen C, et al. | Heterotrophic bacteria growing in association with Methylococcus capsulatus (Bath) in a single cell protein production process | 50 | Applied Microbiology and Biotechnology | 2002 | [37] |
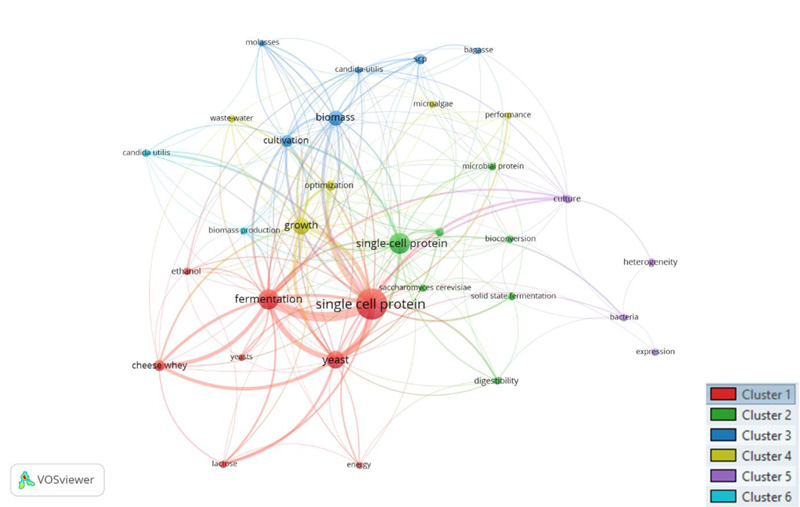
Of all the keywords used, 32 occurred a minimum of 5 times. The highest co-occurrence of keywords was between single-cell protein and fermentation used together in 20 documents. This is followed by yeast and single-cell protein (15) and yeast with fermentation (11), then finally fermentation and cheese whey (8). These keywords can guide researchers to retrieve relevant papers within this field of study.
4. MICROBIAL GENERA AND RAW MATERIALS UTILISED FOR SINGLE-CELL PROTEIN
Microbial genera employed in single-cell protein production include; bacteria, yeasts/moulds, or algae. Although the most investigated genera are fungi, algae are the richest source of protein as they typically contain over 40% of rough protein (dry weight) [38]. The production of SCP by these microbial genera can be achieved using several substrates, including; wood, straw, cannery, spent liquor, whey, hydrocarbons, among others. The traditional raw materials utilised for SCP production are mostly mono and disaccharides, including glucose, other hexose and pentose sugars. Other possible substrates for SCP include; bagasse, citrus squanders, molasses, compost, whey, starch, and sewage [39, 40].
4.1. Fungi as Single-Cell Protein
Fungi are the principal microorganism of interest in single-cell protein research. They are the most studied organism thus far because of several advantages which encourage their ease of growth and harvest, large size (5 – 10μm) and low water activity that enables easier recovery during harvest [41]. Furthermore, the use of fungi as SCP is desirable because of their nutritional composition. They have a high protein and essential amino acid content and contain a high proportion of vitamins and lipids [39, 42-44]. Similarly, they have the ability to grow at low pH, making their production cost-effective and rapid [39, 41]. Advances have been made in selecting suitable fungal strains for SCP production, selecting suitable substrates and optimising substrates and bioprocess parameters for improved yield. For the selection of producer organisms for single-cell production, the major criteria considered are; total biomass yield and protein content, nucleic acid content, utilisation of diverse substrates and digestibility by the host [2, 45, 46]. Various fungi (yeasts, moulds and mushrooms) have been demonstrated to meet some of these criteria and Table 4. presents some representatives of interesting strains.
| Fungi | Advantages | References |
|---|---|---|
| Candida intermedia FLO23 | High protein content (48.4% of dry weight); Efficient utilisation of lignocellulosic materials and xylose | [47] |
| Candida utilis | High protein content; amino acids particularly lysine, vitamin B complex and ergosterol; Ability to bind metal ions; Shorter generation time | [48-50] |
| Cryptococcus aureus G7a | Synthesis of inulinase in addition to useful protein content | [51] |
| Dipodascus spp. | Useful protein content (30-36%); Good amino acid composition | [52] |
| Geotrichum candidum | High protein content (40%); good digestibility potential; low-cost method of recovery | [53] |
| Kluyveromyces marxianus | High protein content (59%); Ability to utilise diverse substrates | [35] |
| Pleurotus florida | High protein content (63%) | [54] |
| Saccharomyces cerevisiae | High protein content (24% - 50%) | [35, 53] |
| Wickerhamomyces anomalus | High protein content (56%); Synthesis of a large number of essential amino acids; Synthesis of killer toxin against contaminating yeasts | [55] |
| Yarrowia lipolytica | High protein content (48% - 54%); Ability to utilise hydrophobic substrates; Attractive expression system for homologous and heterologous protein production | [56-60] |
| Yarrowia lipolytica (Genetically engineered strains) | High protein content (53.7%, 151.2g/L of SCP); Synthesis of inulinase | [61, 62] |
4.1.1. Production of Single Cell Proteins from Fungi
Beyond the criteria listed earlier for single-cell protein production, new research has been directed towards the selection of improved fungal strains with enhanced abilities to be utilised in single-cell protein production. For example, Candida utilis, a natural source of some essential bioactive compounds [63], have been recently used for SCP production, incorporating biomass and extracts of C. utilis in fermented dairy beverages [64]. Recently, it was shown in a study that S. cerevisiae could be used to enrich cereal bars to improve human nutrition [65]. These studies may eventually pave the way for the formulation of innovative bioproducts for human food, departing from the traditional use of SCP for animal feeds. Moreover, in the bid to find such fungal strains suitable for other valuable applications in addition to SCP, researchers are beginning to focus on marine yeasts and plant endophytic fungi [46, 66]. The use of mixed fungal strains for SCP production and the development of genetically engineered fungal strains for improved substrate utilization has been highlighted by various authors [20, 61, 67] and promises to be a veritable avenue for the discovery of more suitable fungal strains for SCP production.
Apart from the producer organism, the composition and characteristics of SCP depend on the type of substrates used for the production [2]. Substrates used for SCP production can be diverse, and this is one of the advantages of SCP as its production can convert waste materials into foods of improved nutritional value and reduces pollution in the environment posed by these wastes [68]. Generally, studies on fungal SCP had always involved substrates of agricultural origin such as molasses [69], sugarcane bagasse, rice husk, sugar beet pulp, wheat straw residue, orange peel, cassava waste, coconut waste, grape waste and mango waste [70-72]. Other agricultural substrates that have gained attention are those that are available in large quantities, renewable and cheap such as corn cobs, stalks, sugarcane press mud, Miscanthus straw, corn stover [47, 67], poultry wastes [39, 73], pineapple waste [74], capsicum powder [75], Jerusalem artichoke tubers [61] and carrot peels [76]. Similarly, liquid substrates such as glycerol, vegetable oil and industrial derivative of tallow have been used to support the growth of fungi for SCP with good biomass yield [27, 77-79]. The use of these substrates, which are generally not utilized as food and are relatively easy to source, makes SCP production desirable as it does not put pressure on the production of crops for human food or animal feed.
The efficacy of these substrates in the production of SCP has been recently demonstrated [42]. In this study, the protein content of Saccharomyces cerevisiae was boosted up to 39.8% using substrates from fruit wastes and vegetable products. This highlights that SCP production can not only serve in meeting our food needs but can help in the reduction of conventional food wastes, which is estimated by the United Nations to cost about $940 billion per year and reportedly represents about one-third of all produced foods, contributing to 8% of global greenhouse gas emissions [80]. However, as noted by some authors, some agricultural substrates such as corn stover, beet and sugarcane molasses require pre-treatment processes, including hydrolysis and detoxification, which may add to the cost of production [67, 81, 82]. Some studies have evaluated alternatives to ameliorate these challenges and make pretreatment simpler and more efficient [47].
A challenge facing SCP production is the availability and sustainability of feedstock, especially from agricultural sources and wastes. Thus, better alternatives, such as using two-stage bioprocessing systems, need to be conceived to achieve sustainability. This system requires inorganic sources of feedstocks such as ammonia and carbon dioxide as substrates in a bioreactor gas-fermentation system. Two studies we identified have investigated the use of Saccharomyces cerevisiae for this new concept [83, 84], producing encouraging results. More studies will be required to develop viable alternatives for the increased and sustainable production of SCP from fungi.
4.2. Algae as a Single-Cell Protein
Algae is a well-known source of SCP, as reported by several authors [2, 15, 39, 85, 86]. This is because algal biomass contains proteins in high concentrations, with an amino acid profile composition that compares well with proteins found in conventional sources like soya, eggs, milk, fish, beef, and peanuts [38]. Furthermore, the growth of algal biomass for SCP production is a cheap and sustainable alternative to the conventional agricultural protein sources, as they are neither limited by land nor require the measure of resources peculiar with agricultural practices [39].
4.2.1. Algal Genera Used in SCP Production
There have been recorded reports of the direct consumption of algae such as Spirulina sp. in some parts of Africa, Mexico and India in the early 20th century [87]. Ever since, there has been a surge in the utilisation of algae for SCP production and a consequent increase in the different genera being utilised [39, 88]. However, most of these genera are used as feeds and supplements more than they are used as food [39, 88]. Table 5 presents some algae genera used in SCP production.
| Genera | Use | Reference |
|---|---|---|
| Chlorella sp. | Microalga species used in human diet Food | [89] |
| Arthrospira sp (Spirulina) | Used as human food | [90] |
| Dunaliella sp. | Food and supplement | [91] |
| Spirogyra, Oedogonium | Food additive | [39] |
| Ulva sp. | Regular diet and salad and soup making | [39] |
| Caulerpa rosemosa | Food | [39] |
| Aphamizomenon sp. | Dietary supplement | [92] |
| Nostoc sp. | Food and Suplement | [92] |
| Scenedesmus sp. | Biofuel and food | [93] |
| Ascophylluem, Fucus, Laminaria | Feed for cattle, pig and poultry | [39] |
| Porphyra sp. | Food | [39] |
| Sargassum sp. | Food | [39] |
| Alaria sp. | Food | [39] |
Microalgae have been shown to have high protein content with unique amino acid composition and nutritional acceptability [38]. The amino acid profile of algae compares well against that of egg and soybean and it can serve as an alternative, considering the high cost of the former [94, 95]. Its protein content has high digestibility once the cell wall is disrupted during clarification using physical methods such as drying under high temperature, boiling or freeze-drying [96, 97]. Furthermore, algae contain high-quality fatty acids such as the Omega-3 fatty acid, essential minerals and vitamins of high nutritional value [98]. Studies have shown that algae produce some chemical compounds that can influence the flavour or aroma of the biomass. These compounds include unsaturated aldehydes, dimethyl sulphide and organohalogens, improving the organoleptic quality of microalgal enriched food materials [38]. Similarly, algal-derived long-chained aldehydes have been reported to have a positive influence on flavour [99].
The genera of algae that have been widely reported as food include Spirulina sp. and Chlorella sp [89, 90]. The macroalgae commonly consumed are the red algae Porphyra spp. (nori) Aparagopsis taxiformis, Gracilaria sp., Chondrus crispus, Palmaria palmata, The Kelps Laminaria sp., Undaria and Macrocystis sp., and the green algae Caulerpa racemosa, Codium and Ulva [100-102]. These algae are either harvested from the wild or are grown in bioreactors [103]. Similarly, some macroalgae from the family of Gelidiaceae and Rhodophyceae are used as food supplements, thickeners and stabilisers [103].
Other microalgae include the blue-green algae, Arthrospira platensis, Arthrospira maxima Dunaliella salina, Chlorella pyrenoidosa, and Euglena gracillis used as food, feeds, food additives and supplements [2]. Currently, algae are used more as feed additives and supplements. However, their use as single-cell proteins has gradually increased over the years [88]. They are preferred to the other sources of single-cell proteins because they have high protein content, essential fatty acids (omega 3), vitamins and low nucleic acid content [41, 88].
4.2.2. Production of Single Cell Proteins from Algae
There are different considerations to be made when processing algae for single-cell protein. These include the end product, for example, whether the protein is to be used as food for humans, food additives, or animal feed, the cost of the raw materials to be used, and the characteristics of the starter culture [104]. The use of cheap substrates such as waste effluents for the mass production of algae in open ponds reduces the cost of production of SCP using algal cells [105]. Generally, the algal cells can be grown in open ponds, using wastewater from agriculture or sewage [106]. It can also be grown in a photobioreactor where the environmental variables such as light intensity, temperature and pH are controlled [107]. The processing methods generally include the culturing of the algae either in a natural open pond or in an artificial photobioreactor [108] for a period of 5- to -8 days and the harvesting and clarification of algal biomass to obtain the final product [109].
The natural open pond (oxidation pond) used for the growth of algae is supplied with the needed substrates for growth [105], mainly organic substances gotten from human or agricultural wastes [110]. The oxidation pond is a complex environment that contains heterotrophic bacteria that break down organic matter within the pond, releasing carbon dioxide, which the algae use as a substrate for their growth. This is termed a cooperative system, as the photoautotrophic algae use the methane and carbon dioxide produced by the bacteria as substrate and, in the process, release oxygen for aerobic bacteria metabolism [2]. Factors that affect algal growth in oxidation ponds are temperature, effluent composition (pH, CO2, phosphorus), light intensity, agitation, pond depth and residence time.
Oxidation ponds come in different sizes and can be partitioned. They help in sustaining the bacteria and algae suspension and facilitate the exchange of metabolites [106, 110]. In most cases, the oxidation pond is an integrated system for water/waste treatment and SCP production [106]. This system has been reported to be cost-effective, especially when the substrates are gotten from the waste effluents [2]. However, some disadvantages of oxidation ponds are the presence of contaminants, low-quality proteins and human perception [2]. Another method of mass-producing algae for SCP production is through the use of a photobioreactor which is a closed system capable of being operated both outdoor and indoor using LED lanterns or sunlight [15, 107], allowing for both batch and continuous production of algae using either organic or chemical substrates.
Produced algae cells are usually clarified to transform the harvested algae into the final product (food, food additives or animal feed). The clarification process includes harvesting, mechanical disruption of cells to eliminate the cell wall and the nucleic materials and drying of the cell, which can be done using different methods, including sun drying and drum drying with further processing to isolate the proteins [111]. Harvesting algae from an oxidation pond is a complex process because the pond contains many organic particles and bacteria contaminants and more so because the specific gravity of algae is closer to that of water. The algal slurry can be harvested using a sieve of fine mesh size, but the slimy nature of the organism may block the sieve, rendering the sieving process inefficient [85]. Various authors have reported the use of centrifugation, flocculation and froth floatation as a more efficient method of harvesting algal biomass after growth [15, 105]. Centrifugation is used to concentrate the algal biomass to enhance biomass recovery using flocculants like lime, iron (iii), chloride (FeCl3), potash Alum and other polyelectrolytes [110]. Alum has been reported to be the best flocculant, as it yields high-quality algal biomass with minimal residue, while lime and FeCl3 leave unwanted residue in the sludge, making it unsuitable for use as animal feed [110]. Alum flocculant also has the advantage of precipitating soluble phosphates, a growth-limiting factor for algae, and permits seasonal collection of clarified effluent with minor eutrophication problems. Algal flocculated biomass is further subjected to froth-floatation using different chemicals such as hydrochloric acid and sodium chloride [85]. Finally, the algal biomass harvested is dried either by sun drying, freeze-drying, air drying or drum drying [95]. Of the drying methods, drum dried algal biomass has the highest digestibility coefficient compared to the other drying methods [96, 112]. Furthermore, the drying process helps to rupture the cell wall of the algae making the protein content much more available for digestion.
4.2.3. Algae Selection for SCP Production
A variety of factors need to be considered when selecting an organism for SCP production on an industrial scale. These factors are Important to assure optimal productivity and include; a high growth rate and low generation time, a high rate of substrate conversion and no requirement for expensive growth factors, resistance to contamination and adverse effects of the growth environment. Furthermore, the possibility of genetic enhancement, high nutritional content, low content of nucleic acid, high digestibility, non-toxicity and a suitable sensorial/functional property [113] are important considerations. Of the microalgae utilised for single-cell protein production, the class: Cyanophyceae, Chlorophyceae, Bacillariophyceae and Chrysophyceae are most commonly employed [114]. The algal genera commonly used for the production of SCP include Chlorella, Spirulina (Arthrospira) and Dunaliella [89, 115]. Of these, Chlorella is most dominant for SCP production [89]. This is because it contains about 45% protein, 20% carbohydrates, 20% fat and 10% minerals, along with fibres and vitamins [104]. An example is Chlorella stigmatophora, a marine microalga, that has high protein content, fast growth rate, improved sensorial quality and uses a mixotrophic mode of nutrition thus, making it suitable for the production of single-cell protein [109]. Other Chlorella species that have been utilised in single-cell protein production include Chlorella sorokiniana and Chlorella pyrenoidosa. Another microalgae utilised is Arthrospira (Spirulina) which has been commercially produced and utilised for human consumption over several centuries [90]. Spirulina sp. is an alkaliphile and grows best at the pH range of 9.5 to 9.8 [90]. This particular property gives the organism a competitive advantage when grown in the wild against other competitors, thus, making it suitable for an integrated oxidation pond production system [115]. Spirulina sp. has been granted GRAS for use as food additives in the United States of America [38], and the microalgae have a high protein and essential fat content. Furthermore, its consumption has other medicinal values, such as weight loss and antioxidant activities [116]. Dunliella sp. is another algae grown in an oxidation pond because of its ability to adapt to different pH and salt concentrations [117]. Other algal genera that have been used in the production of Single Cell-Protein are; Porphyrum sp., Scenedesmus sp., Alaria sp., Ascophylluen sp., Fucus sp., Laminaria sp., Caulerpa rosemosa, Durvilleaantartica, Ulva sp., Laminaria sp., Ecklonia sp., Eisenia sp., Nostoc sp., Pelvwtia sp., Porphyratenera, Monostroma sp., Undaria sp., Sargassum sp., Rhodomenia sp., Rhodymenia sp., Gelidium sp., Grateloupia sp., Synechococcus sp., Sargassum sp., Spirogyra, and Oedogonium [104].
4.2.4. Uses and Current Applications of Algae in Food and Feeds
Algae provide tremendous benefits when consumed either directly or as food additives or feed [118]. Besides having high protein content, microalgae have also been effective in controlling obesity, mopping up free radicals in the body, enhancing tumour necrosis, and serving as a thickener to certain food products [119]. Table 6 shows some products and ingredients derived from the commercial growth/ production of microalgae.
| Commercial Products | Characteristics | Company | Website/References |
|---|---|---|---|
| Microalgal flour | Chlorella is used to replace dairy fats, vegetable oils and egg yolks in products such as ice cream, cookies, cakes, dressings, chocolates milk and pasta sauce | AlgaVia | https://algavia.com/how-we-innovate/ |
| Bread and Cookies | Chlorella is inserted into the dough loaves to reducethe oil content and replace eggs. It is also used to create a healthier product and maintain the texture and structure of the bread | Solazyme | http://solazymeindustrials.com/ |
| Spaghetti | Chlorella vulgaris and Spirulina are added to spaghetti tocompletely remove butter, oil and egg yolks and maintain the texture, mouthfeel and overall flavour of the product. | Weihai Qingzheng Foods | [120] |
| Juice powder | Juice powder is enriched with Athrospiraflos-aquae,improving the nutritional content, producing a healthier product, which can be used in diets. | E3Live Original | https://www.e3live.com/p-2-e3live-original.aspx |
| Dairy drinks | Euglena is used in the fermentation of yoghurt as its excellent stability in low pH beverages means there is no grittiness from the protein. When fermented, it presents sensory characteristics that are acceptable | Kemin | https://www.nutraingredients.com/ Article/2020/12/08/ Kemin-s-algae-based-ingredient-gets-EU-Novel-Food-approval |
| Extraction of astaxanthin | Haematococcus pluvialis improves the texture, emulsification, water binding and flavour delivery in food. Astaxanthin is principally consumed by the salmon feed industry | Vitamin Express | https://www.vitaminexpress.org/uk/ astaxanthin-royal-astaxanthin-capsules |
| Capsules | Chlorella, Spirulina, Haematococcus Pulvialis and Dunaliella in these capsules are potent dietary supplements to boost daily nutrient intake | Ocean Drop | http://oceandrop.com.br |
| Docosahexaenoic acid (DHA) | Produced by Schizochytruim, DHA has severalapplications as food and dietary supplements in infant formulas, beverages and products for pregnant women. | Daesang | https://daesangeurope.com/ |
| Beta-Carotene | Dunaliella is rich in Beta-carotene and is used asa natural food colourant and an additive for animal feed | Natural Beta Technologies | [121] |
| Bacteria SCP | Organism Used | Substrate Used | Industry | Use | Ref. |
|---|---|---|---|---|---|
| Pruteen | Methylophilus methylotrophus | Methanol, methane |
Imperial Chemical Industry; UniBio A/S |
Pig feed | [123] |
| UniProtein | Methanotrophic bacteria | Methane | UniBio A/S | Animal Feed | [124] |
| FeedKind | Methanotrophic bacteria | Methane | Calysta Inc.; Cargill | Animal feed | [2] |
| KnipBio Meal | Methylobacterium extorquens | Methanol, methane | KnipBio | Fish feed | [17] |
| Novacq | Bacteria and Microalgae | CSIRO Canberra, Australia | Animal feed | [125] |
4.3. Bacteria as Single Cell Protein
Single cell protein derived from bacteria is desirable because the cells contain about 50-80% of dry protein by mass, are easy to grow using cheap substrates as sources of carbon and energy [2, 41, 104]. (Table 7) shows some examples of bacteria used for SCP production. Generally, the genus Methylotrophus sp. is mainly used by different companies to produce SCP [2, 107]. Bacteria have some advantages over algae and fungi for use in SCP production, not only because of their protein composition and use of cheap substrates but also because of their faster growth rate, which is higher compared to those of algae and fungi. The specific growth rate of bacteria varies from 0.28μ to 0.65μ per hour, while that of algae and fungi varies between 0.60μ to 0.70μ and 0.16μ to 0.39μ per hour [122]. These qualities make bacteria a choice organism for SCP production [2, 104]. However, the use of bacteria SCP production presents some challenges:
1. Bacterial cells have high nucleic acid content, which can be toxic to both man and animals. The removal of nucleic acid materials from the cells introduces additional processing costs, thereby impeding commercial production.
2. Bacterial cell size and low density makes harvesting difficult and expensive from the fermentation medium.
3. Human bias has also made the acceptability of bacteria SCP difficult [41].
4. The presence of toxins produced by some gram-negative bacteria can lead to food poisoning.
Bacteria cells are fermented in bioreactors to produce SCP. Recent developments in technology have focused on improving the processing of SCP from bacteria cells to maximise production [2]. The use of bioreactors for SCP production also reduces the risk of contamination and increases growth efficiency.
Other bacteria used for the production of SCP include; Aeromonas hydrophylla, Acinetobacter calcaoceticus, Alcaligenes eutrophus, Bacillus sp., Cellulomonas sp., Methylomonas sp., Mycobacterium sp., Nocardia sp., Pseudomonas sp., Rhodopseudomonas sp., Brevibacterium sp., Methanomonas methanica, and Methylophilus sp [104].
4.3.1. Production of Single-cell Proteins from Bacteria
The production of SCP from bacteria is carried out in a bioreactor or fermenter followed by other downstream processing, including; concentration of cells, clarification and packaging [5, 39, 104, 126]. Production of SCP from bacteria is usually achieved using a semi-solid fermentation process [5] using agro-wastes and purified substrates such as methanol, methane and a pure culture of the bacteria [127]. Fed-batch or continuous fermentation can also be employed in bacteria SCP production. However, the continuous fermentation technique is more sustainable for commercial production [41]. This is because, during the fed-batch technique, the oxygen demand of the process would exceed the carrying capacity of the reactor, making it very expensive and less sustainable. Similarly, the batch fermentation technique is not recommended because of the changes in the conditions of the reaction medium over time [128].
In addition to the concentration of the cell biomass through centrifugation and filtration using semi-permeable membranes, there are other downstream processing required in the production of SCP from bacteria. These include the removal of amino acids in the cells through chemical methods using acidified alcohol, salt, acid, alkalis or enzymatic treatments such as the activation of endogenous RNAase by brief heat treatment up to 60-700C for 20 mins. The use of these chemical and enzymatic methods has proven effective in reducing the nucleic acid content with very little loss in the protein content [129]. However, these processes have been implicated in the production of some harmful chemicals. For example, the hydrolysis of nucleic acid at elevated temperatures causes the formation of Lysnoalanine- an unusual amino acid involved in the cross-linking of alkaline proteins. Lysinoalanine has been implicated in reducing digestion and causing kidney changes in rats [41]. Finally, the cells are dried and clarified and then packaged [5].
The use of bacteria for SCP production is costly because of the various downstream processes carried out. However, recent developments in recombinant DNA technologies have focused on improving and selecting organisms with desirable traits for SCP production for improved yield and economic profitability [130]. For example, VTT Ltd. is investigating the reactor design and options for combining farm methane generation with the production of microbial oil and feed protein [2]. This investigation aims at identifying key factors that would encourage commercial production of SCP for food and feed at reduced cost through the volarisation of cheap industrial substrates while improving yield and nutritive value in terms of protein and vitamins [39]. In another instance, genetic modification of Pseudomonas methylotrophus by the Imperial Chemical Industries (ICI) was used to improve protein yield, growth rate and subsequent use in the commercial production of SCP [131]. This was achieved through the isolation of the glutamate dehydrogenase system of Escherichia coli and transferring this to the glutamine-keto-acid-transaminase (GOGAT) of the mutant P. methylotrophus, which forms glutamate and NAD(P) from alpha-ketoglutarate, NAD(H) and glutamine, thus leading to improved nitrogen assimilation [131].
4.4. Advantages and Challenges Associated with Single Cell Protein Production
Proteins derived from microorganisms offer certain advantages compared to animal or plant-derived proteins, and these can be considered in their nutritional quality, ease of propagation and production, resource requirements and the ancillary metabolites it produces.
The qualities and quantities of the various nutrients derived from organisms used for SCP production vary from one organism to another and rely on extrinsic factors such as the nature and quality of the substrate and presence of contaminant [105]. Of the microorganisms used for SCP, bacteria produce the highest quantity of protein by dry mass, but due to their high nucleic acid content and presence of toxins, fungi and algae are preferable sources of SCP [112]. Generally, SCP microbes are primarily needed for their protein content and quality. However, what determines the acceptability of a particular species as food or feed are growth rate, the substrate used, contamination and presence of toxins [39]. Algae are one of the most studied SCP microorganisms, with authors comparing the essential amino acid constituents of algae with some conventional protein sources such as egg and soybean. This comparison shows that microalgae are good surrogates for conventional protein sources [95].
The amino acid composition of microbial proteins and their high protein value makes them potent alternative sources of proteins for human and animal consumption. Furthermore, the cheap propagation method of single-cell proteins gives it a selective advantage over conventional protein sources. This includes high growth rate and low generation time, cheap multiple substrates, less demand for space and the possibility of all year production [2]. Generally, algae have the lowest growth rate, followed by yeast and filamentous fungi, with bacteria having the fastest growth rate of about 20 minutes to two hours [39]. However, the growth rate may vary depending on the growth conditions such as the availability of nutrients in certain forms, which is usually determined by the fermentation technique adopted and environmental factors such as pH, presence of contaminants or absence of competition [104]. This is unlike animal or plant-based proteins which usually take months to years for growth and maturation [2].
Organisms utilised for single-cell protein production have different modes of nutrition, including heterotrophic, chemoautotrophic, photoautotrophic and methylotrophic [132], enabling them to utilise a wide range of substrates and survive in different growth conditions, unlike heterotrophic organisms, which require organic matter as a source of carbon and the photoautotrophs which can only fix carbon using carbon dioxide in the presence of sunlight. The ability of SCP organisms to engage in the dual phototrophic and heterotrophic modes of nutrition is referred to as mixotrophy. This is common in microalgae and confers a unique advantage over other organisms with a single mode of nutrition [132]. Furthermore, this mixotrophic nature of algae makes it possible for integrated propagation of algal single-cell protein with simultaneous treatment of waste [106]. This has further reduced the cost of production of protein, making it commercially feasible [105]. Additionally, single-cell proteins require less space and can be grown in a controlled environment, thus making it possible for an all-year supply of protein [104].
In addition to the viable role for SCP as food and feed, SCP produced by microorganisms contains vitamins and metabolites with diverse health and other benefits. Examples include some single-cell proteins fractions that exhibit foaming and gelation properties and can be used in food to increase palatability [133].
Although SCPs present numerous advantages compared to conventional protein sources, there are some challenges associated with single-cell protein production. These include the cost of production, sensory bias, regulatory challenges, underdevelopment of food technology techniques and safety concerns [2]. Furthermore, the downstream processing of single-cell protein can be capital intensive. The series of processes involved in cell biomass concentration, extraction, and possible purification of proteins demands significant resources. For instance, proteins from bacteria sources need to be purified to remove the nucleic acid content in the cell biomass, as this could have serious adverse health implications. At the same time, algae and fungi derived single-cell proteins needs to undergo some downstream processing to enhance the biological availability of the proteins. These organisms have thick cell walls that cannot be digested by animals and thus need to be removed to increase the protein efficiency ratio (PER) [94]. In some cases, the cost of obtaining purified substrates and the upgrading/valorisation of specific substrates for use by feedstock organisms can also be relatively high, especially for chemoheterotrophic organisms thus, limiting the commercialisation of numerous single-cell protein production [132].
Generally, humans are sceptical about consuming single-cell proteins. This bias can be attributed to safety concerns, organoleptic qualities and concern for the substrate used in production. Single-cell proteins derived from algae have poor organoleptic quality, which makes it difficult for human consumption. Also, safety is a vital aspect of food production and foremost consideration for SCP for human consumption. Several bacteria-derived SCP are yet to be granted the GRAS status because of the risk of intoxication commonly associated with gram-negative bacteria.
An important consideration about single-cell protein consumption is the potential for the bioaccumulation of toxic substances and heavy metals within the cells of the organism from waste effluents from homes or industries used for their production as these can lead to cases of food poisoning [134]. Similarly, fungi derived SCP are also less desired because of toxins in some filamentous fungi [104]. On the other hand, algal cells are relatively more desired than bacteria and fungi because of the low potential of toxin accumulation in algal-derived SCP [104]. Furthermore, bacteria and fungi cells contain high nucleic acid, inimical to health when consumed in high quantities [41]. These safety challenges have elongated the process of certifying some SCP, creating some bottlenecks for them to attain the generally regarded as safe (GRAS) status. The attainment of GRAS status is a requirement for novel foods before they can be approved for consumption. Generally, in the United States and the European Union, novel foods are subjected to long trial periods before being considered safe for consumption. In Europe, Regulation (EU) 2015/2283 governs the development and consumption of novel foods, including SCP [135] and adequate care is taken when the food is from microbial sources to validate the safety status of these foods [136]. Finally, a key challenge in advancing the production and utilisation of SCP is a lag in developing novel food technology equipment that can facilitate the propagation and processing of single-cell proteins on a commercial scale without incurring a high cost of production [137].
CONCLUSION
In this review, a bibliometric study was used to analyse the body of research on single cell proteins. Our study revealed that although papers in this area have been published over the last 60 years, there has been minimal growth. Thus, investigating the role of microorganisms as a significant source of dietary proteins requires further attention from the scientific research community. Research on SCP is multinational involving several institutions, with major research contributions emanating from the United States of America. With the ever-growing world population and projections on the inability of conventional agriculture to cater to the food needs of the future, especially in Africa and Asia, more collaborative research activities are needed to increase food production using SCP through capacity building and increasing infrastructure for SCP production especially within these regions.
A more in-depth analysis of the original research showed that SCP from fungi is the focus of most research activity in this area, followed by algae and bacteria. Some benefits of SCP identified from our study include the relatively lower cost compared to other dietary protein sources; the possibility of all year-round production of proteins and increased production rate due to the rapid growth rate of microbes. However, this review identified several challenges with SCP production, including the high cost of downstream process, poor yield and safety issues. In order to address these challenges, future research should focus on (1) use of genetic engineering approaches to improve potential strains with desirable characteristics, e.g. removing toxin producing genes (2) understanding consumer perception to the use of Single cell protein, to identify misconceptions and address these (3) simplifying and reducing the downstream processing required to make SCP readily consumable. Overall, SCP has an important role to play in ensuring food security in the coming years and the dedication of scientific effort in improving this aspect of food production will be beneficial in providing nutritious food and feed while ensuring a cleaner and more sustainable environment as SCP production is generally carbon neutral without adverse environmental impact as obtainable in conventional agriculture.
LIST OF ABBREVIATION
| SCP | = Single Cell Protein |
AUTHORS' CONTRIBUTIONS
Conceptualization and Design, H.O., T.M. and C.K.A. methodology, C.K.A., C.O., A.O.; software and Analysis O.A.O., F.I. and C.K.A; writing original draft preparation, A.A.; writing review and editing, C.K.A, A.A and T.M.; visualization, C.K.A. and O.O.; project administration, H.O. All authors have read and agreed to the published version of the manuscript.
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
No funding was received to assist with the preparation of this manuscript.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


