All published articles of this journal are available on ScienceDirect.
First Record of Dissemination of BLBLI-Resistant Enterobacter cloacae from Public Hospitals in Baghdad, Iraq
Abstract
Background:
Enterobacter cloacae are most frequently isolated from human clinical specimens.
Objective:
This cross-sectional study aimed to investigate the dissemination of E. cloacae clinical isolates resistant to β-lactam-β-lactamase inhibitor (BLBLI) combinations from different clinical specimens of hospitalized patients.
Methods:
E. cloacae isolates were recovered from different clinical samples of hospitalized patients in three main hospitals in Baghdad city. E. cloacae isolates were identified based on their morphology and biochemical tests, and the identification was confirmed using Vitek-2 system. The antibiotic susceptibility testing of E. cloacae isolates to a variety of antibiotics was achieved using disc diffusion test (DDT) and Vitek-2 system.
Results:
Results found that among 335 culture-positive samples, 30 isolates (8.9%) belonged to E. cloacae. A high rate of isolation was observed in urine isolates (46.6%), followed by wounds (burns) isolates (26.6%). Out of 30 E. cloacae strains isolated during this study, 18 (60%) showed reduced susceptibility to BLBLI combinations. TEM genes (TEM-1 and TEM-2) were successfully amplified from 7/18 isolates (38.8%) and high rate of BLBLI genes was detected (CTX-M, bla-SHV, SHV-2, and OXA-1). However, no BLBLI genes of bla-AmpC, bla-OXA-2, and bla-OXA-10 were found in E. cloacae isolates when tested using specific primers for bla-AmpC and bla-OXA genes.
Conclusion:
From this study, we can conclude that the production of inhibitor-resistant β-lactamases by E. cloacae isolates could be increasingly common in nosocomial pathogens other than E. coli or K. pneumoniae in public hospitals in Baghdad, Iraq.
1. INTRODUCTION
Enterobacter cloacae are pathogens that are commonly isolated from human clinical specimens. They cause an extensive range of nosocomial infections involving skin and soft tissue, the lower respiratory tract, urinary tract, and the CNS infections. The antibiotic-resistance properties of these bacteria had been described by several authors worldwide [1].
Resistance to BLBLI combinations in Gram-negative enteric rods may be related to different mechanisms, such as TEM-1 penicillinase hyperproduction, chromosomal and plasmid-mediated AmpC production, OXA-type β-lactamase production, deficiencies in outer membrane permeability, inhibitor-resistant TEM (IRT)-ß-lactamase production, CTX-M, type-β-lactamase production, and more recently, carbapenemase production [2].
β-lactam-β-lactamase inhibitor (BLBLI) is characterized as an adaptive resistance mechanism definitely established by the organism to beat off the activity of β-lactamase inhibitors [2]. Inhibitor-resistant TEM (IRT) β-lactamases, which emerged in the 1990s, are most commonly observed in urine strains. IRT enzymes include an assembly of plasmid-mediated types of TEM-1 and TEM-2 with reduced affinities for penicillins and changed interactions with irreversible inhibitors, like clavulanic acid, sulbactam, and tazobactam [2].
BLBLI β-lactamases-producing isolates are frequently resistant to ampicillin-sulbactam and amoxicillin-clavulanate combinations. Inhibitor-resistant beta-lactamase enzymes have previously been described to be present in a diverse range of pathogenic bacteria, like E. coli, Enterobacter cloacae, Proteus mirabilis, Klebsiella spp., Pseudomonas aeruginosa and Citrobacter freundii [2-4].
In Iraq, there is little to no information available on the prevalence of BLBLI β-lactamases in E. cloacae clinical isolates; in addition, there is no information regarding their antibiotic resistance patterns. Thus, the objective of this study was to inspect the occurrence of BLBLI β-lactamases in E. cloacae isolates recovered from different clinical specimens of hospitalized patients in three main hospitals of Baghdad city, Iraq.
2. METHODS
2.1. Study Design
This cross-sectional study was designed to examine the incidence of BLBLI β-lactamases in E. cloacae isolates recovered from different clinical specimens of hospitalized patients. In this study, E. cloacae isolates were recovered from 386 different clinical samples of patients hospitalized in three public hospitals of Baghdad city, Iraq, in addition to some private clinics during the period of December 2019 to April 2020. Bacteria were isolated from different clinical specimens, including blood, urine, burn, ear, skin, stool, eye, nose, and bronchial fluid. E. cloacae strains were isolated and identified based on their morphological characteristics and biochemical tests [5]. Identification was confirmed using Vitek-2 system (BioMerieux, France).
2.2. Eligibility Criteria
Selection criteria included hospitalized patients of both sexes and all ages (neonates: aged less than 28 days; infants: aged 28 days to 1 year; children: more than 1 year to 17 years of age; adults: aged 18 years or more). Exclusion criteria included clinical samples obtained from previously hospitalized individuals who had received antibiotic therapy for more than seven days.
2.3. Antibiotic Susceptibility Testing
The antibiotic susceptibility of E. cloacae bacteria to a variety of antibiotics was tested using Vitek-2 system (BioMérieux, France) and disc diffusion test (DDT). The results were interpreted based on CLSI documents [6]. The following BLBLI combinations were purchased from Oxoid, UK, as standard antibiotic disks with an identified potency for lab use: Amoxicillin/Clavulanic acid (30 μg), Ceftazidime/Clavulanic acid (30/10 μg), and Ceftriaxone-Tazobactam (30/10 μg). E. coli ATCC 25922 was used as the reference strain for antimicrobial susceptibility testing.
2.4. Detection of β-Lactamase by Chromogenic (Nitrocefin) Disc Method
For detection of the ability of 30 E. cloacae isolates to produce β-lactamase, Chromogenic (Nitrocefin) Disc Method was used. β-lactamase production was noticed by the appearance of a pink color within 15 minutes.
2.5. Detection of AmpC and Extended Spectrum β-Lactamases
Disk diffusion method was used for testing E. cloacae isolates for cefoxitin susceptibility [6]. The resistant isolates (≤18 mm inhibition zone diameter) were considered as primarily AmpC producers. E. cloacae isolates were also analyzed for their ability to produce extended spectrum β-Lactamase (ESBL) by initial screen test. The isolate would be treated as ESBL producer, if the inhibition area of ceftazidime would be ≤ 22 mm [6]. ESBL producer strains positive on initial screen test were further confirmed using confirmatory ESBL production as follows: ceftazidime alone and in combination with clavulanate were established. Inhibition zone of ≥5 mm increase in diameter for ceftazidime in combination with clavulanate versus its zone alone confirmed an ESBL producing strain. The antibiotic resistance patterns (MDR, XDR, and PDR) of all E. cloacae isolates were detected according to the method proposed by Magiorakos et al. [7].
2.6. DNA Extraction and PCR Assay
E. cloacae strain was inoculated in 5 mL of Broth Heart Infusion medium and grown overnight at 37º C. DNA was extracted from bacterial cell using plasmid Mini Prep Kit DNA extraction (Bioneer, USA). The PCR reactions were performed in a thermal cycler (Clever, U.K). For quantification of the DNA (ng µL1), 1 µL of Genomic DNA was quantified with a NanoDropTM spectrometer (NanoDrop Technologies LLC, USA), according to the manufacturer’s guidelines. DNA concentration was assessed from absorbance at 260 nm.
2.7. PCR Protocols
The DNA purified from E. cloacae strains were exposed to a set of primers by PCR. A set of primers of β-lactamases genes [8-13] were used (Bioneer, USA) in an attempt to differentiate BLBLI-resistant isolates (Table 1). The PCR conditions are illustrated in Table 1. The PCR products were identified by agarose gel electrophoresis. Ethidium bromide (EtBr) at a concentration of 0.5 μg/ml was used for visualization of the amplicons, and the gel images were acquired using a regular gel-documentation system (Cleaver Scientific, U.K).
Table 1.
|
Primer name |
Oligo Sequence (5′→3′) |
Product Size (Pb) |
Conditions | Refs. |
| bla-AmpC | AmpC-F ATC AAA ACT GGC AGC CG | 550 | 94°C 3 min 1x 94°C 45 sec 60°C 45 sec 35x 72°C 1 min 72°C 5 min 1x |
[8] |
| AmpC-R GAG CCC GTT TTA TGC ACC CA | ||||
| TEM-1 | TEMU1 ATG AGT ATT CAA CAT TTC CG | 867 | 95°C 5 min 1x 94°C 1 min 58°C 1 min 35x 72°C 1 min 72°C 10 min 1x |
[9] |
| TEML1 CTG ACA GTT ACC AAT GCT TA | ||||
| TEM-2 | TEMU2 ACT GCG GCC AAC TTA CTT CTG | 374 | 95°c 5 min 1x 94°c 30 sec 62°c 30 sec 30x 72°c 30 sec 72°c 10 min 1x |
[10] |
| TEML2 CGG GAG GGC TTA CCA TCT G | ||||
| SHV-1 | SHV-F GGT TAT GCG TTA TAT TCG CC | 867 | 95°c 5 min 1x 94°c 30 sec 62°c 30 sec 30x 72°c 30 sec 72°c 10 min 1x |
[11] |
| SHV-R TTA GCG TTG CCA GTG CTC | ||||
| SHV-2 | SHVU2 CCG CAG CCG CTT GAG CAA A | 374 | 96°c 5 min 1x 96°c 1 min 60°c 1 min 35x 72°c 1 min 72°c 10 min 1x |
[10] |
| SHVL2 GCT GGC CGG GGT AGT GGT GTC | ||||
| OXA-1 | OXA-1F ACA CAA TAC ATA TCA ACT TCG C | 867 | 96°c 5 min 1x 96°c 1 min 61°c 1 min 35x 72°c 2 min 72°c 10 min 1x |
[12] |
| OXA-1R AGT GTG TTT AGA ATG GTG ATC | ||||
| OXA-2 | OXA-2F TTC AAG CCA AAG GCA CGA TAG | 702 | 96°c 5 min 1x 96°c 1 min 65°c 1 min 35x 72°c 2 min 72°c 10 min 1x |
[12] |
| OXA-2R TCC GAG TTG ACT GCC GGG TTG | ||||
| OXA-10 | OXA-10F CGT GCT TTG TAA AAG TAG CAG | 651 | 96°c 5 min 1x 96°c 1 min 61°c 1 min 35x 72°c 2 min 72°c 10 min 1x |
[12] |
| OXA-10R CAT GAT TTT GGT GGG AAT GG | ||||
| CTX-M- | CTX-M-F CGC TTT GCG ATG TGC AG | 550 | 94°c 4.5 min 1x 94°c 50 sec 58°c 50 sec 35x 72°c 50 sec 72°c 7 min 1x |
[13] |
| CTX-M-R ACC GCG ATA TCG TTG GT |
2.8. Statistical Analysis
All BLBLI β-lactamases-producing E. cloacae isolates were analyzed using the Statistical Package for the Social Sciences version 26 (SPSS Inc., Chicago, IL, USA). Numerical data were evaluated using the chi-squared and Fisher’s exact test, and compared with that of cefoxitin resistance among BLBLI β-lactamases-producing isolates; cefoxitin resistance (≤18 mm inhibition zone) was considered an indicator of AmpC producers. P-value < 0.01 was considered significant.
3. RESULTS
3.1. Isolation of E. cloacae Isolates:
In the present study, among a total of 386 clinical samples, 335 (86.7%) showed positive cultures, whereas no growth was observed in the remaining 51 samples (13.3%). Among 335 culture-positive samples, 30 isolates (8.9%) belonged to E. cloacae, of which, high rate of isolation was observed for urine strains 14/30 (46.6%), followed by wounds (burns) isolates 8/30 (26.6%), while the remaining isolates were obtained from blood, ear swab, and bronchial fluid samples. E. cloacae were isolated and identified based on biochemical tests, and the identification was confirmed using the Vitek-2 system. Results of the present study revealed that a high isolation rate (46.6%) of E. cloacae isolates was from urine samples recovered from the hospitalized patients with UTIs in three public hospitals of Baghdad.
3.2. Production of β-Lactamase and Antibiotic Susceptibility to BLBLI Combinations
Results revealed that 28/30 isolates (93.3%) produced β-lactamase by changing the color of nitrocefin disk from yellow to reddish-orange. Regarding the resistance of E. cloacae isolates to BLBLI combinations, results revealed that only 18 (60%) of 30 E. cloacae were found to be resistant to one or more of BLBLI combination antibiotics.
Out of 18 E. cloacae isolates resistant to BLBLI, 17 (94.4%) isolates were resistant to ampicillin-sulbactam, 11 (61%) resistant to amoxicillin-clavulanate, and only 9 (50%) were resistant to pipracillin-tazobactam.
3.3. Production of ESBL and Ampc β-Lactamase Enzymes
Results of this study revealed that out of the 18 E. cloacae isolates that were resistant to BLBLI, 15 (83.3%) displayed zone augmentation with clavulanate, confirming ESBL production. Vitek-2 system was also employed for recognition of ESBLs in 18 E. cloacae isolates. Vitek-2 system was also used to identify AmpC β-lactamases in 18 E. cloacae isolates. All isolates were susceptible to cefoxitin disks and the inhibitor area was ≥ 18 mm based on CLSI guidelines.
3.4. Antibiotics Susceptibility of E. cloacae Isolates
All 30 E. cloacae isolates showed multidrug resistance. The frequency of 18 E. cloacae isolates that were resistant to BLBL inhibitor combination was determined. The results revealed that all E. cloacae isolates were fully resistant to ampicillin and amoxicillin (100%), but they were fully susceptible to cefoxitin (100%).
Results revealed that the majority (46.6%) of E. cloacae isolates were recovered from patients with UTIs. All these isolates were found to be MDR, β-lactamases producers, and highly resistant to amoxicillin and ampicillin. The results found that most E. cloacae strains were resistant to at least 6 agents, which imply that a new option of antibiotic is required to eliminate E. cloacae related to UTI infections. It was found that out of 30 E. cloacae isolates, 80% of them showed reduced susceptibility to cephalexin, cephatholin, and the monobactam (aztreonam). The rate of resistance to 3rd generation cephalosporins (cefotaxime, ceftriaxone, and ceftazidime) was 76.6% while only 26.6% of isolates were resistant to cefepime (4th generation cephalosporin). Isolates were found to be fully susceptible to carbapenems and nitrofurantoin, and low rate of resistance to gentamicin was discovered (46.6%).
The greatest active quinolone towards all E. cloacae strains was levofloxacin (20%), followed by ciprofloxacin (30%) and nalidixic acid (53.3%). The resistance rates of E. cloacae strains to tobramycin, cotrimoxazole, and tetracycline were 43.3%, 73.3%, and 70.0%, respectively.
Regarding the distribution based on the type of antibiotic resistance patterns, high rate of MDR 30/30 (100%) was noticed, followed by XDR 6/30 (20%); however, no PDR type was observed among the E. cloacae isolates.
3.5. Molecular Detection of E. cloacae BLBLI Genes by PCR
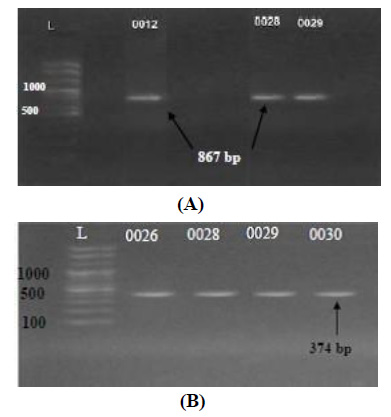
A pair of primers of two TEM enzymes was used for detecting the existence of TEM genes in E. cloacae strains. TEM genes (TEM-1 and TEM-2) had been positively amplified from 7/18 isolates (38.8%) of E. cloacae with molecular length of 867 bp and 374 bp, respectively, indicating their antibiotic resistance to Inhibitor-resistant TEM (IRT) beta-lactamases. The amplicons were noticed in the gel and matched with the DNA ladder (Figs. 1A and 1B).
Regarding SHV enzymes, a pair of primers of two SHV enzymes was used for detecting the presence of SHV genes in E. cloacae isolates. SHV genes (SHV-1 and SHV-2) had been positively amplified from 6/18 isolates (33.3%) of E. cloacae (4 isolates had bla-SHV and 2 isolates had bla-SHV-2) with a molecular length of 867 bp and 477 bp, respectively. The amplicons were noticed in the gel and matched with the DNA ladder.
A pair of primers of bla-AmpC enzyme gene was employed for identifying the occurrence of bla-AmpC gene in E. cloacae isolates. The result exhibited that bla-AmpC gene was not present in any of the isolates. However, the phenotypic assays showed all 18 E. cloacae strains to be susceptible to cefoxitin and the zone of inhibition was ≥ 18 mm based on CLSI guidelines, which confirmed our molecular results regarding this resistance gene.
Regarding OXA type of BLBLI genes, a pair of primers of three OXA enzymes (OXA-1, OXA-2, and OXA-10) was employed for detecting the existence of OXA genes in E. cloacae strains. Only OXA-1 gene had been positively amplified from 6/18 isolates (33.3%) of E. cloacae with molecular length 813pb. The result showed E. cloacae isolates to not be able to yield bla-OXA-2 and bla-OXA-10) genes. The results regarding OXA-2, and OXA-10 resistance genes revealed that these genes did not originate in any of E. cloacae strains when established by using OXA gene primers.
In the present study, a pair of primers of bla-CTX-M enzyme gene was used for detecting the occurrence of bla-CTX-M gene in E. cloacae strains. CTX-M gene had been amplified from 5/18 isolates (27.7%) of E. cloacae with molecular length 550pb.
4. DISCUSSION
In the present study, 86.7% of the clinical samples showed positive cultures, whereas 13.3% of them showed no bacterial1growth. The negative culture may be due to fungal, viral, or fastidious bacterial infection that might be missing during sample transportation. Among 335 culture positive samples, only 30 isolates (8.9%) belonged to E. cloacae.
Regarding the identification of the isolates, E. cloacae were recognized based on biochemical tests and the identification was confirmed using Vitek-2 system. Nimer et al. [14] investigated the performance of the Vitek-2 system for direct rapid identification of Gram-negative rods and 74 Gram-positive cocci from inpatients, and they suggested that VITEK-2 cards inoculated directly from positive blood culture bottles are appropriate for rapid diagnosis and sensitivity testing of the pathogenic bacteria.
Saffar et al. [15] investigated the incidence and pattern of antimicrobial sensitivity of uropathogens in UTIs from three hospitals in Iran, and they found E. coli to be the prominent cause of UTI in hospitalized patients followed by Pseudomonas spp. (10.4%) and Enterobacter spp. (5.7%).
Harris et al. [16] investigated cases of bacteriaemia caused by Enterobacter spp. in hospitals in Australia. They found that only 3.4% of patients were at an overall risk of relapsed or persisting bacteriaemia, and only two patients with an emergent resistance to 3GCs.
Chromogenic (Nitrocefin) disc method was used for detection of β-lactamase in E. cloacae isolates. Nitrocefin is more sensitive to recognize all β-lactamases. The reason may be that nitrocefin is more sensitive to recognize all β-lactamases formed by Gram-negative bacilli [17]. Also, this technique is suitable for the recognition of most β-lactamase types and sensitive for identifying low level of β-lactamases yielded by gram negative rods.
Results of this study regarding the antibiotic susceptibility of E. cloacae isolates to BLBLI combinations revealed 60% of the isolates to be resistant to one or more of BLBLI combinations antibiotics. Harris et al. recognized cases of bacteriaemia caused by Enterobacter spp. in patients in Australia and they found that the condition of patients receiving complete therapy with a broad-spectrum BLBLI did not relapse; they concluded that more studies are required to determine whether BLBLIs denote an efficient option to avoid carbapenem for bacteriaemia caused by Enterobacter spp [16].
Miro et al. [18] found that 7% of E. coli isolates from a university hospital in Spain exhibited resistance to Augmentin. Kaye and his colleagues [10] reported that 69 clinical isolates resistant to ampicillin-sulbactam were also resistant to Augmentin. Among the isolates, 12 (4.7%) were nosocomial and 57 (2.8%) were community acquired.
Leflon-Guibout et al. [19] studied the epidemiological features of E. coli isolates from French hospitals that exhibited reduced susceptibility to amoxicillin-clavulanate (MIC >16 mg/ml) over a 3- year period. The incidence of resistant isolates (15%) was higher in surgical wards than in medical wards.
Results of the study found 15 of E. cloacae isolates (out of 18 inhibitor-resistant isolates) to be ESBL producers. Malloy and Campos [20] reported that the recognition of ESBL-producing strains can be hard since the existence of ESBLs in these bacterial cells does not continually yield a resistance phenotype.
Manzur et al. [21] described the epidemiology and microbiological features of an outbreak caused by ESBL-producing E. cloacae in a cardiothoracic intensive care unit (CT-ICU), and they found that 7 patients with ESBL-producing E. cloacae were successfully recognized. Since the majority of ESBL enzymes had been found to be consistently sensitive to β-lactamase inhibitors, BLBLI combinations were promoted as possible alternative therapy [22].
Results of this study also found all 8 E. cloacae isolates recovered to be susceptible to cefoxitin disks as per CLSI guidelines [6], confirming their ability to produce AmpC β-lactamases. Derbyshire et al. [23] reported that, in spite of the fact that certain AmpC-producing Gram-negative rods are sensitive to cefoxitin, a low level of enzyme gives negative or slightly positive results. Several authors worldwide reported more than 60% of bacterial isolates to be cefoxitin resistant when they tested AmpC producers by nitrocefin disk method.
AmpC β-lactamases are the greatest significant β-lactamases in Gram-negative rods. Failure to recognize AmpC β-lactamase producers in hospital and other health care centers may lead to unsuitable antimicrobial therapy, and consequently, an increase in the mortality rate [24]. This result is compatible with the results of several authors worldwide [25], who have reported E. coli to be the main uropathogen in patients with bacteriuria, displaying high rate of resistance for both amoxicillin and ampicillin.
Manzur et al. [21] investigated an outbreak initiated by E. cloacae strains in a CT ICU, and they revealed that ESBL-producing strains exhibited reduced susceptibility to quinolones and aminoglycosides.
Regarding the molecular detection of E. cloacae BLBLI genes by PCR, results found that 38.8% of E. cloacae isolates were inhibitor-resistant TEM (IRT) beta-lactamases while only 6/18 isolates (33.3%) were inhibitor-resistant SHV (SHV-1 and SHV-2) beta-lactamases.
Kaye et al. [10] analyzed the β-lactamase inhibitors resistant E. coli strains isolated in the Northeastern United States and found that TEM type alone was predominant among the bacterial strains, and there was one strain that harbored both TEM and SHV types of BLBLI genes.
Miro et al. [18] found that 0.8% of clinical strains isolated from a Spanish hospital were TEM-1 hyperproducers and they exhibited resistance to Augmentin. Martín et al. [26] found that 0.5% of E. coli strains producing IRT enzymes were isolated mainly from urine (77.8%), and a high degree of TEM β-lactamase inhibitors diversity was recognized (TEM-30, -32, -33, -34, -36, -37, -40, and -54).
Miro´ et al. [18] found that 0.15% of clinical strains isolated from a Spanish hospital were SHV-1 hyperproducers and they exhibited reduced susceptibility to amoxicillin-clavulanate. Several authors reported resistant E. coli strains isolated from human and animal sources to express bla-SHV type β-lactamase inhibitors [27].
Kaye and his colleagues [10] analyzed the amoxicillin-clavulanate resistance in E. coli isolates from a tertiary care hospital in United states and revealed that one isolate had both SHV type and TEM type enzyme.
The result showed that bla-AmpC gene was not present in any E. cloacae isolates recovered in this study. Regarding OXA type of BLBLI genes, only OXA-1 gene had been positively amplified from 6/18 isolates of E. cloacae, while OXA-2 and OXA-10 resistance genes were not present in any of E. cloacae strains.
Davin-Regli and Pagès [1] reported that the expression of novel β-lactamase enzymes, like AmpC, is not common among E. aerogenes and E. cloacae strains. Coqueand his colleagues [28] reported that 2 E. coli strains were nominated as negative for ESBL production in phenotypic tests but they expressed the TEM gene, perhaps owing to the expression of new β-lactamases, like AmpC. Thus, the implementation of molecular assays together with phenotypic methods is vital for final recognition of these variants of β-lactamases.
Abid and his colleagues revealed a majority of AmpC genes (40%) among clinical isolates of E. cloacae from Iraqi hospitals [29]. Soltan Dallal et al. [27] found that reduced susceptibility to β-lactamase inhibitors was encoded mostly by AmpC, OXA, TEM, and SHV β-lactamases. Kaye et al. [10] reported that the resistance to amoxicillin-clavulanate in E. coli clinical isolates in United States was encoded by TEM, SHV, OXA, and AmpC β-lactamases.
The inability of E. cloacae isolates to yield bla-OXA-2 and bla-OXA-10 genes was compatible with AL-Tememy and Al-Charrakh [4], who showed OXA-2 and OXA-10 resistance genes to not be detected in any of E. coli strains from patients with UTIs in Iraq.
Abid et al. [29] revealed a majority of bla-TEM genes (60%) among E. cloacae strains from Iraqi hospitals, followed by the bla-OXA gene, which was detected only in 20% of these strains. Several authors reported amoxicillin-clavulanate resistant clinical strains of K. pneumoniae, K. oxytoca and E. coli to express bla-OXA type β-lactamase inhibitors [18].
Results of this study also revealed CTX-M gene to be positively amplified from 6/18 isolates of E. cloacae isolates. A study conducted by Nishida et al. in Japan revealed the emergence of E. cloacae complex co-producing IMP-1-type carbapenemase and CTX-M ESBL [30]. Baba Ahmed et al. [31] revealed a widespread presence of CTX-M-15 extended-spectrum β-lactamases among enteric bacterial isolates in ICU in an Algerian Hospital. Several authors worldwide have described the coexistence of bla-CTX-M and bla- TEM genes among ESBL producers bacterial strains [32]. Nimri and Azaizeh [32] reported that more than 80.7% of ESBL-producing isolates had at least one ESBL gene (TEM or CTX-M).
Finally, although our results have been obtained from a single center, they involve high internal validity and the sample size included with random sampling was large enough; thus, we think that our results can be safely generalized worldwide.
CONCLUSION
From this study, we can conclude that the production of inhibitor-resistant β-lactamases by E. cloacae isolates could be increasingly common in nosocomial pathogens other than E. coli or K. pneumoniae in public hospitals in Baghdad, Iraq.
AUTHORS’ CONTRIBUTIONS
Jawad R. Alzaidi contributed to conceptualization, data curation, original draft preparation, and supervision. Ahmed S. Mohammed validated the findings and contributed to writing, reviewing and editing of the manuscript.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Written and verbal consent was obtained from each patient before obtainment of their samples. The study protocol was approved by the committee on publication ethics at Southern Technical University under the reference No. BMS/0276/018.
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All procedures performed involving human participants were in accordance with the ethical standards of institutional and/or research committee, and with the 1975 Declaration of Helsinki, as revised in 2013.
CONSENT FOR PUBLICATION
Written and verbal consent was obtained from each patient before obtainment of their samples.
STANDARDS OF REPORTING
STROBE guidelines and methodologies were followed in this study.
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


