All published articles of this journal are available on ScienceDirect.
Phagoburst Response Level of Neutrophils to Septic and Non-Septic Acinetobacter baumannii Isolates
Abstract
Background:
Acinetobacter baumannii is an opportunistic pathogen causing community-acquired and nosocomial infections. Dissemination of infection to blood causes septicemia associated with serious morbidity and mortality of patients. Neutrophils are essential for the control of A. baumannii infections by different mechanisms, including oxidative burst.
Aims:
This study was conducted as an attempt to determine the effect of septicemic and non-septicemic A. baumannii isolates on the phagoburst response of neutrophils.
Methods:
Neutrophils were isolated from an immunocompetent individual; chronic myeloid leukemia (CML) and lung solid tumor (ST) patients. The phagoburst response of these neutrophils to various strains of A. baumannii isolated from septic and non-septic patients was investigated by flow cytometer.
Results:
The presence of this pathogen lowered the phagoburst response in the different types of neutrophils compared to their response to the opsonized Escherichia coli. The phagoburst response of the neutrophils from the immunocompetent individual was significantly higher than that of neutrophils from the immunodeficient patients when stimulated by the septicemic or nonsepticemic A. baumannii isolates. The isolate type (septicemic or non-septicemic) had no significant effect on the neutrophil phagoburst response of the immunocompetent individual and a significant effect on the phagoburst response of neutrophils from the immunodeficient patients. The phagoburst response of the neutrophils from the immunodeficient patients stimulated by septicemic A. baumannii isolates was significantly lower than that when neutrophils stimulated by the nonsepticemic isolates. Also, there was a significant difference in the phagoburst response of neutrophils from the CML and ST patients when stimulated by the septicemic and non-septicemic isolates. This observation might be due to the combined effect of virulent A. baumannii isolates and the chemotherapy regime the patient was undertaking.
Conclusion:
The results suggest that both the isolate type and the source of neutrophils have a significant effect on the neutrophil phagoburst response. The potential virulence of the septicemic A. baumannii isolates and dissemination to blood may be dependent on the host’s immune status and the neutrophils phagoburst response.
1. INTRODUCTION
Acinetobacter baumannii is a Gram-negative opportunistic coccobacillus belonging to the family Moraxellaceae [1]. It is a pathogen of increasing medical importance, causing nosocomial [2] and community-acquired infections [3], which include pneumonia, urinary tract, wound and life-threatening infections such as meningitis and sepsis especially in immunocompromised patients [2, 4-6]. Following infection, epithelial cells produce antimicrobial peptides that recruit neutrophils to the site of infection [7]. The specific interaction between A. baumannii and neutrophils remains largely unknown [8]. Unlike many pathogenic bacteria, A. baumannii suppresses the adhesion ability of neutrophils and prolongs the lifespan of neutrophils by inhibiting neutrophil extracellular traps (NETs) formation and thereby escaping the neutrophil defense mechanism at an initial point (at 1 h) [8]. Interestingly, A. baumannii is transported with the invading neutrophils, which are no longer needed to fight bacteria and re-enter the vasculature through increased IL-8 production in a mechanism referred to as the “bacterial immunity taxi”. This reverse neutrophil migration can disseminate an infection to other organs and bloodstream [8]. In contrast, Lazaro-Diez et al. 2017 [9] reported that A. baumannii is phagocytosed by neutrophils and induces neutrophil extracellular traps (NETs) formation. These discrepancies may be due to differences in the experimental conditions.
Migration of the polymorphonuclear neutrophils to inflammatory sites is guided by chemotaxis in response to different bacterial products (chemo-attractants), where microbes are recognized and killed. Killing occurs through oxygen-dependent (oxidative burst) and oxygen-independent mechanisms. The latter is used primarily in anaerobic environments through the fusion of phagosomes with lysosomes containing mainly lysozymes [10]. In an oxidative burst, neutrophils generate reactive oxygen species to kill the invading microorganisms. The source of these species is active nicotinamide adenine dinucleotide phosphate (NADPH) oxidase. This enzyme catalyzes NADPH dependent reduction of O2 to form superoxide anions (O2 −) and reactive oxygen species (ROS) derived from this radical, including hydrogen peroxide (H2O2), hydroxyl radical (OH.), and hypochlorous acid (HClO] [11, 12]. NADPH dependent reduction of O2 to O2− is activated through integrin-dependent signals generated during neutrophil adhesion to the pathogen [13].
Studies about the interaction of septicemic and non-septicemic A. baumannii isolates with neutrophils and the effect of the host clinical condition on oxidative burst are scarce. Therefore, this study represents an attempt to understand the ability of septicemic and non-septicemic A. baumannii isolates to elicit oxidative burst in neutrophils of immunocompetent and immunodeficient individuals using phagoburst assay. Understanding the variation in the ability of these isolates in the generation of oxidative burst could help in developing a new therapeutic agent to treat the infections caused mainly by the septicemic isolates.
2. MATERIALS AND METHODS
2.1. Blood Samples
Human peripheral blood (4ml) was obtained using heparin test tubes (BD Biosciences, USA) from 3 adults: a healthy immunocompetent and two immunodeficient patients. The patients were a chronic myeloid leukemia (CML) patient under hydroxyurea and imatinib mesylate therapy and a solid lung tumor (ST) patient under cisplatin and fluorouracil (5FU) chemotherapy. All human subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the declaration of Helsinki, and the protocol was approved by the Ethics Committee of the University of Jordan (Project identification code 16/2012/1613).
The fresh blood samples were used directly in the phagoburst activity assay. The neutrophil count per mL of blood was determined using the automated hematology analyzer Sysmex XP100 (Sysmex, Japan) and was then adjusted with 0.9% normal saline to 3000 cells/ μl in each sample.
2.2. Bacterial Strains
A total of 10 clinical septicemic and non-septicemic A. baumannii isolates were included in this study. Three septicemic isolates were obtained from the blood of cancer patients admitted to King Hussain Cancer Center [KHCC] in Amman-Jordan [14]. Seven non-septicemic isolates, which included: 4 isolates (3 nasal and 1 urine) obtained from cancer patients admitted to KHCC [14] and 3 urine community-based isolates obtained from MedLabs Consultancy Group’s Reference Laboratory, a private-sector laboratory in Amman-Jordan. Identification of the isolates was carried to the species level using Microscan Wakaway (Beckman Coulter, USA).
The septicemic A. baumannii ATCC BAA 1710 and the non-septicemic A. baumannii ATCC BAA 747 (isolated from the ear) were used as control isolates throughout this study.
2.3. Maintenance of Isolated Strains
A. baumannii isolates were stored at -80°C in Tryptic Soy Broth (TSB) broth with 25% glycerol (15). Bacteria were thawed in a water bath at 30°C for 2 minutes, gently mixed, then plated on Muller Hinton agar and incubated at 37 o C overnight.
2.4. Phagoburst Activity
The phagoburst response of neutrophils from the healthy immunocompetent individual and patients was performed in duplicates using the FagoFlowEx kit (EXBIO, Praha, Czech Republic) and Accuri C6 Flow cytometer (BD Biosciences, USA) following the manufacturer’s instructions. An aliquot of each blood sample containing 150000 neutrophil/50μl was used. The number was normalized to 1500 cells per gate to measure the phagoburst response after separate stimulation with 7 x 107 CFU/ml [15, 16] of clinical and control A.baumannii isolates. Upon stimulation and during ingestion of bacteria, phagocytes activate NADPH oxidase to produce reactive oxidative intermediates (hypochlorite ions), which destroy bacteria inside the phagosome. The reactive oxidants formed during the oxidative burst was monitored by the addition of the fluorogenic substrate dihydrorhodamine 123 (DHR123) and oxidation to fluorescent rhodamine 123. The relative fluorescence intensity [mean fluorescence intensity (MFI)] of DHR and relative proportion (%) of neutrophils that are positive for DHR fluorescence after bacterial stimulation were detected at 488 nm (blue-green excitation light). Also, the phagocytic activity of neutrophils was measured after their stimulation with the opsonized E. coli isolates included in the kit. The lyophilized E. coli was reconstituted using 250 μl of deionized water, of which 10 μl was used as a positive stimulus according to the manufacturer’s instructions. A neutrophil sample (150000 cells/50 μl) without stimulus served as negative background control.
2.5. Statistical Analysis
SPSS Version 16.0 (SPSS Inc., Chicago, IL, United States) was used for all analyses. Some figures were produced using the ggplot2 package in R. Two-way ANOVA was used to test the effect of “neutrophils’ source” and “isolate type” on neutrophil phagoburst response. Tukey post hoc method was used to test the differences between the group combinations. p-values of <0.05 were considered significant.
3. RESULTS
3.1. Phagoburst Response of Neutrophils Stimulated by E. coli
The phagoburst response of neutrophils from the three adults was first assessed using E. coli as a stimulus. The MFI for neutrophils of the healthy immunocompetent individual, CML and ST immunodeficient patients was 99.7%, 96.7%, and 61.5%, respectively (Table 1, Fig. 1). This activity was within the kit’s normal range, for both the healthy individual and CML neutrophils but lower than the kit’s normal range, for ST neutrophils. The negative background of the negative control was <5% for the three neutrophil sources (Table 1) and was considered acceptable, as it is lower than the kit’s normal range width of 10%. Results’ MFIs of each category were corrected by excluding the respective negative control MFI.
3.2. Phagoburst Response of Neutrophils Stimulated by A. Baumannii Isolates
The ability of the clinical and control septicemic and non-septicemic A. baumannii isolates to elicit phagoburst response in neutrophils of the 3 adults was tested. The MFI of their neutrophils (Table 1) was below the kit normal range (90-100%) when incubated with any of the A. baumannii isolates. The MFI profile for the phagoburst response of neutrophils from the 3 adults challenged separately with some representative septicemic and non-septicimic A. baumannii isolates is shown in Fig. (2).

| Source of neutrophils | Negative Control |
Stimulus | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
E.coli Positive control |
Clinical A. baumannii isolates | Septicemic Control ATCC BAA 1710 | Non-septicemic Control ATCC BAA 747 | |||||||||||
| Septicemic | Non-septicemic | |||||||||||||
| Blood |
Nasal |
Urine |
||||||||||||
| No.1 | No.2 | No.3 | No.1 | No.2 | No.3 | No.1 | No.2 | No.3 | No.4 | |||||
| Healthy individual | 4.5 | 99.7 | 80.0 | 76.1 | 79.1 | 83.0 | 84.8 | 67.0 | 85.3 | 73.0 | 74.0 | 75.2 | 69.8 | 88.0 |
| CML Patient | 1.2 | 96.7 | 34.0 | 43.0 | 40.0 | 67.0 | 61.0 | 53.0 | 68.0 | 64.3 | 69.0 | 70.0 | 53.5 | 71.0 |
| ST Patient | 2.5 | 61.5 | 28.0 | 32.0 | 30.0 | 52.0 | 51.0 | 35.0 | 54.0 | 42.0 | 40.0 | 54.0 | 34.0 | 45.0 |
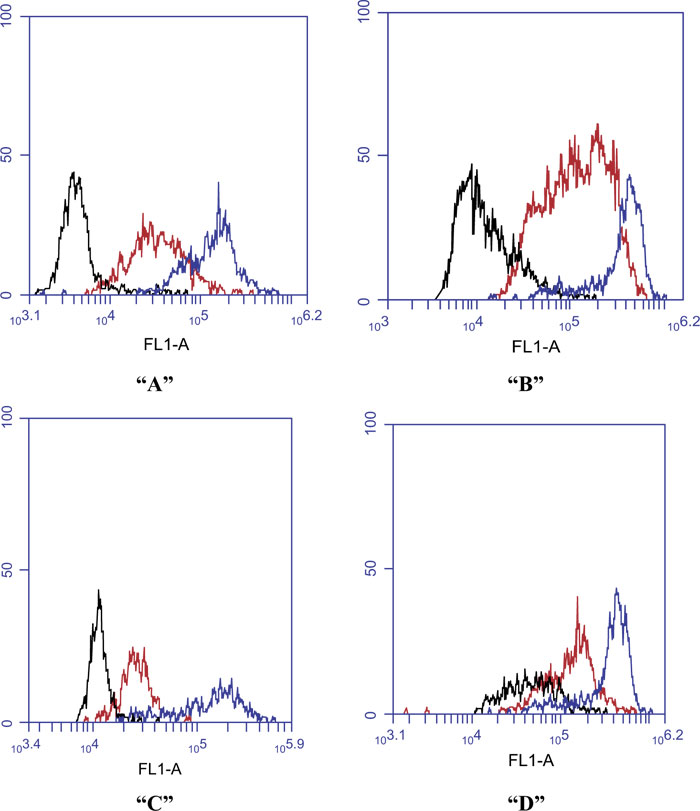
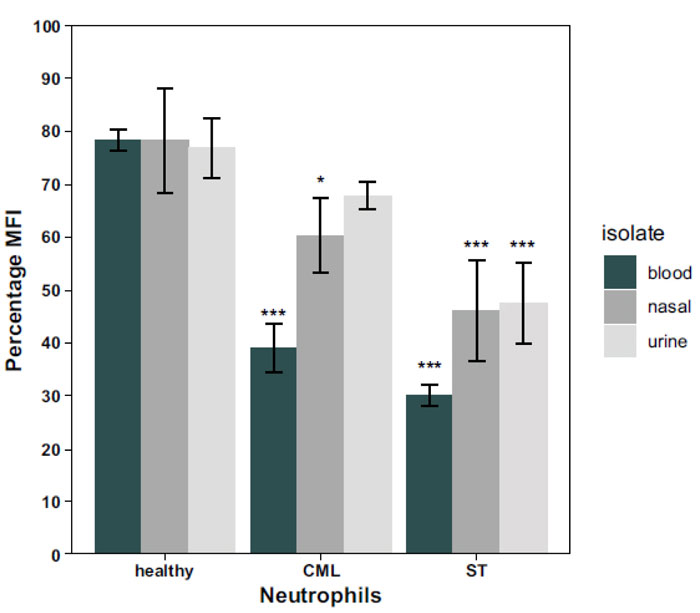
To compare the phagocytic activity of the neutrophils from the 3 adults, three different models were prepared using two-way ANOVA. In the first model, the clinical A. baumannii isolates in Table 1 were pooled and grouped into blood, nasal or urinary samples. Two-way ANOVA test revealed a significant interaction (p=0.0038) between the effect of both the isolate type and the source of neutrophils on the overall neutrophil phagoburst response. Therefore, a simple effect analysis for both factors was performed individually. Simple effect analysis for the isolate type showed that there was no significant effect (p= 0.9419) of the isolate type on the neutrophil phagoburst response of the healthy individual (Fig. 3). On the other hand, the isolate type had mainly a significant effect on the phagoburst response of CML (p=5.99x10-5) and ST (p= 0.0062) neutrophils. Fig. (3) shows that there was a significant difference in the phagoburst response of CML (p= 0.01) and ST (p = 0.09) neutrophils between blood and nasal isolates. Also, there was a significant difference in the phagoburst response of CML (p= 0.0015) and ST (p= 0.03) neutrophils between blood and urine isolates (p= 0.0015). However, there was no significant difference in the phagoburst response of CML (p= 0.81) and ST (p = 0.999) neutrophils between nasal and urine isolates (Fig. 3).
When the simple effect of the source of neutrophils was analyzed (Fig. 3), it was found that the neutrophil phagoburst response of the healthy individual was significantly higher than that of CML neutrophils for the blood (p < 0.00001) and nasal (p < 0.05) isolates but not for the urine isolates (p> 0.05). Also, the phagoburst response of these neutrophils was significantly (p< 0.0001) higher than the response of the ST neutrophils for the three types of isolates (Fig. 3). Detailed p values of (Healthy-CML) interactions were: p = 0.0000043 for blood, p = 0.043 for nasal, and no significant interaction p = 0.53 for urine, whereas the p values of (Healthy - ST) interactions were: p = 0.0000001 for blood, p = 0.000082 for nasal, and p = 0.000041 for urine. When ST and CML neutrophil phagoburst responses were compared, a significant difference was found for the urine isolates (p < 0.05) only, while no significant difference was found for both blood and nasal isolates (p> 0.05). Detailed p values of (ST-CML) interactions were: p = 0.7017084 for blood, p = 0.1694793 for nasal, and p = 0.0039141 for urine.
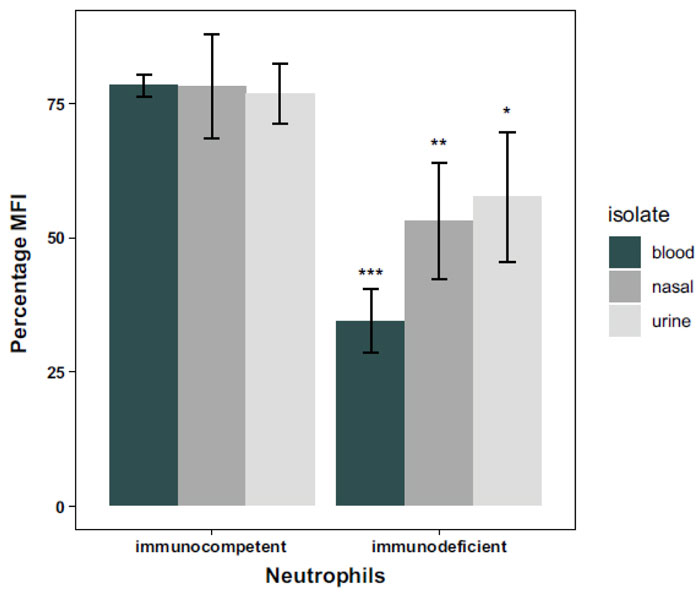
In the second model, all clinical A. baumannii isolates in Table 1 were pooled and grouped into blood, nasal or urinary samples, depending on the isolate’s type. The neutrophils of CML and ST patients were pooled and treated as one group of immunodeficient patients. Two-way ANOVA was used to test for differences between groups. There was a significant (p = 0.027) interaction between isolate and neutrophils’ source effects in this model. Thus, the simple effects of neutrophils’ source and isolate type were analyzed, followed by Tukey post hoc method for pairwise comparisons. It was found that there was no significant difference between the phagoburst response of the neutrophils from the healthy immunocompetent individual (Fig. 4) when stimulated separately by A. baumannii isolated from blood and nasal samples (p= 1), from blood and urine samples (p= 0.9999) and even from nasal and urine samples (p= 1). Whereas for the immunodeficient patients, there was no significant difference (p= 0.9438) between the phagoburst response of the neutrophils when stimulated by either the nonsepticemic nasal or urine A. baumannii isolates. However, the phagoburst response of the neutrophils of those patients stimulated by septicemic blood A. baumannii isolates was significantly lower than that when neutrophils stimulated by the nonsepticemic nasal (p= 0.0212) and urine (p= 0.0014) isolates (Fig. 4).
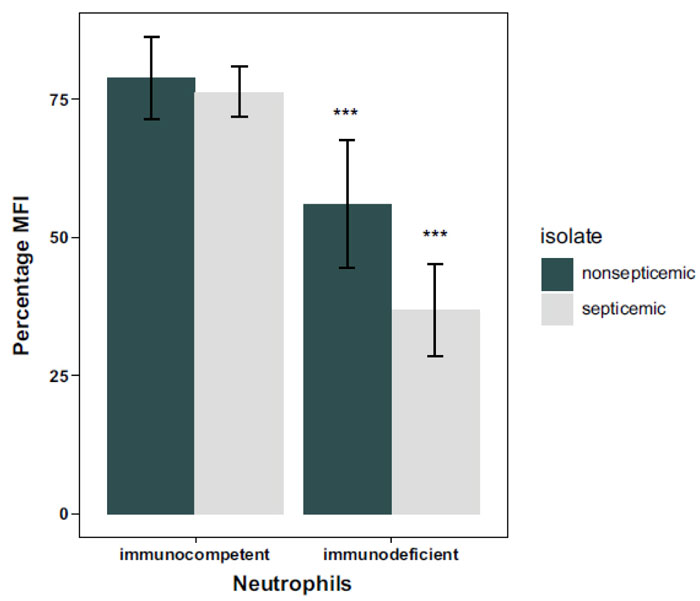
In the third model, the neutrophils of CML and ST patients were pooled and treated as one group of immunodeficient patients as in the second model. All clinical and control A. baumannii isolates in Table 1 were grouped as septicemic and nonsepticemic isolates. Two-way ANOVA revealed a significant (p = 0.028) interaction between the effect of isolate and individual type in this model. Thus, we performed simple effect tests followed by Tukey pairwise comparisons. It was found that the phagoburst response of the neutrophils from the immunocompetent individual was significantly (p<0.00003) higher than that of neutrophils from the immunodeficient patients when stimulated by the clinical and control septicemic and nonsepticemic A. baumannii isolates (Table 1, Fig. 5). There was no significant difference (p=0.67) between the phagoburst response of neutrophils from the immunocompetent patient to septicemic and nonsepticemic isolates (Fig. 5). However, the phagoburst response of neutrophils from the immunodeficient patients to septicemic isolates was significantly (p= 0.00012) lower than that to the nonsepticemic isolates (Fig. 5).
4. DISCUSSION
Respiratory burst (phagoburst) is part of polymorphonuclear cells' defense against invading bacteria [11]. The ability of neutrophils to exert the phagoburst response gives an indication of the immunological status of the individual and the pathogenic capability of the invading bacteria [17, 18]. It has been stated that the lower of the activity detected is associated with, the lower ability of the host to fight its pathogen [19]. The measurement of phagoburst response and differences in the neutrophils’ response after incubation with A. baumannii isolated from different clinical origins, may give an indication of the future course of the infection and the extent of pathogenicity.
The isolates type showed to have a significant effect on the phagoburst response of CML and ST neutrophils only. CML neutrophils were affected by the type of isolate, although initially seemed to have a normal response to the non-viable E. coli. This could be explained by CML being a condition of a fairly slow-growing myeloid leukemia, in which cells of myeloid origin affected, mainly neutrophils, mature partly but not completely and do not have the same ability to fight infection as normal neutrophils do [20]. It is complicated even further by the phase of the disease [21] and the patient’s immunosuppressive therapy [22, 23]. Hence the phagoburst response of CML neutrophils towards the isolates was significantly decreased overall, and the response to blood isolates was the lowest. Compared to the urine isolates, the significantly lower response to blood and nasal isolates could imply a greater virulence for this type of isolates. Blood isolates may be considered more virulent as they can cause septicemia. Also, nasal isolates can cause more serious infections such as septicemia when suitable conditions are provided, for instance, a weak immune status of the infected host. Septicemia is occasionally secondary to other infections like respiratory infections [24]. A. baumannii is a well-known cause of respiratory infections in hospitals, especially following the respiratory intubation of patients [2]. Therefore, nasal colonization and/or infections could be a possible route for this pathogen to reach the blood stream. Consequently, these results may suggest that the ability of A. baumannii to colonize or cause infection depends greatly on the patient condition, and low immunity could be a major factor facilitating infection. Same patterns of significance were obtained with the ST neutrophils, which in contrast to CML neutrophils, are not immature but their deteriorated immune response or function could be owed to the chemotherapy regime the patient was following [25]. The altered immune response towards the urine isolates may be owed again to the nature of the isolate and its pathogenicity, implying that urine group isolates could be in fact less virulent than the other groups while blood isolates the most virulent, an assumption that should be further studied to be confirmed.
On the other hand, the neutrophils source also had a significant effect on the phagoburst response, being the highest in healthy neutrophils and lowest in the ST group for the three different types of isolates. The response of healthy neutrophils was clearly owed to their proper immune response and immunocompetent status, while ST neutrophils had much lower phagoburst response, being the lowest with the blood isolate and thus highly significant. Their response could have been affected by the chemotherapy regime undertaken by the patient, inhibiting the overall immunity to a great extent producing immunosuppression, and affecting the neutrophils’ ability to fight infection [25]. The greater immunosuppression status of ST neutrophils compared to the other immunodeficient condition i.e. CML, was shown by the lower activity of ST neutrophils to the non-viable kit control. Otherwise, CML neutrophils’ response varied, since they were significantly different from the other two neutrophil categories for some types of the isolates. They were not significantly different from healthy neutrophils for the urine isolates type. Once more this may be due to the suggested lower virulence of the urine group of isolates and better immune response for the CML neutrophils compared to ST neutrophils. They were as well not significantly different from ST neutrophils for both blood and nasal isolates, mostly due to a similar lower immune response for the CML and ST neutrophils compared to healthy neutrophils. While there was a significant result between CML and ST neutrophils for the urine isolates, that could be deduced by the weaker isolate type nature and as well by a likely better immune status of CML neutrophils compared to ST neutrophils. An observation that was anticipated by their better initial phagoburst response to the non-viable E. coli control.
In the second model, CML and ST neutrophils were pooled as an immunodeficient group of neutrophils and compared to the immunocompetent neutrophils for the three different types of isolates. For the immunocompetent neutrophils, there was no significant effect to the type of isolate, implying that the healthy neutrophils are not affected by the assumed degree of virulence of the different types of isolates. In contrast, neutrophils of the immunodeficient patients demonstrated a significant difference in the phagoburst response for the blood isolates over both nasal and urine isolates. This supports our suggestion of different virulence effects for some isolates, being the blood isolates solely more virulent in this model triggering the lowest phagoburst response, which is probably accentuated by the immunodeficiency status of the neutrophils. However, this assumption should be further studied for other types of immundeficiencies.
The immunodeficient neutrophils response for the septicemic isolates was highly significant with a much lower phagoburst reaction. This could be due to the defective immune status, whether caused by chemotherapy agents or the condition of the illness itself as well as the nature of interaction of the particular isolate with the neutrophils and its ability to cause infection if given the opportunity. In fact, many of the septicemic infections may be secondary to other conditions such as respiratory infections, urine infections and many others. Afterward, the pathogen could find its way to the bloodstream [24], causing invasive infections like septicemia through altering the interaction that occurs between the isolate and the defending neutrophils of the host [8, 26].
The distinctive and singular ways in which A. baumannii can interact with host cells, neutrophils particularly, may determine the development of infection or the persistence of colonization in a particular patient, as well as the progression to septicemia that may be originated from other less critical A. baumannii infections [27, 28]. Consequently, although yet to be proven, A. baumannii could take advantage of the host’s weaker neutrophils, similar to those in the CML and ST categories, for its transportation by means of the previously mentioned mechanism “bacterial immunity taxi” [8] to disseminate from its original site of infection into other organs and/or the bloodstream causing septicemia.
Different studies have addressed the oxidative responses of neutrophils and their role in CML [ 29,30] and different solid tumors [31,32], but to the best of our knowledge, none have compared their phagoburst responses one to the other and with underlying bacterial infection with a pathogen like A. baumannii. The different phagoburst responses to isolate types and the significantly lower response of the immunodeficient neutrophils to the septicemic isolates shown in the present study, may suggest the use of burst assay as an aid in predicting the capability of the infecting A. baumannii to cause invasive septicemic infections in immunosuppressed patients. More extended studies should be performed to validate this suggestion, including a higher number of isolates besides involving several other immunosuppressive conditions and including a wider number of neutrophils samples for each source since the present study was conducted on a single sample for each source of neutrophils to eliminate possible factors of variability among the sources.
CONCLUSION
The results suggest that A. baumannii’s potential ability to infect and the nature of its infection depends greatly on the immunological patient condition and the pathogen interaction with the host’s neutrophils. This unique interaction between A. baumannii and neutrophils should be further elucidated at the molecular level. Also, the genetic nature of the isolate should be investigated as it could be related to the course and severity of the infection caused by this organism, as well as the expression of the virulence factors of both septic and non-septic A. baumannii isolates could differ and may be dependent on the host’s neutrophils and pathogen interaction as well as on the patient’s condition. Clarifying this relation may have a great value in predicting the course of infection and influence the therapeutic choices undertaken by physicians.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Ethics Committee of the University of Jordan, Jordan (Project identification code 16/2012/1613).
HUMAN AND ANIMAL RIGHTS
No Animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
All patients participated on a voluntary basis and gave their informed consent.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
The research was made possible by grant No. (19/2018/244) from the School of Graduate Studies, The University of Jordan, and by a grant No. “Excom-01-2017” from MedLabs Consultancy Group.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors are grateful to the healthy immunocompetent and immunodeficient individuals for providing peripheral blood samples. Also, the authors are thankful to Dr. Feras Hawari who in collaboration with the co-author Prof. Asem Shehabi facilitated collecting the clinical bacterial isolates from KHCC.


