All published articles of this journal are available on ScienceDirect.
Characteristic of Extended Spectrum β-Lactamase-Producing Enterobacteriaceae from Fecal Carriage Isolates of Intensive Care Unit Patients at Sanglah Hospital, Bali, Indonesia
Abstract
Background:
The increasing Extended-Spectrum β-Lactamases-producing Enterobacteriaceae (ESBL-PE) infections in the Intensive Care Unit (ICU) needs an early warning system for the detection of these bacteria. The ESBL-PE fecal carriage analysis is a screening method that can be used to detect and characterize these bacteria. Furthermore, it aids in assessing an ICU patient’s risk of possible infection and prevent its transmission to the other patients within the period of hospitalization; therefore, enhancing the quality of patient care while alsoreducing morbidity and mortality due to ESBL-PE infection in ICU.
Objective:
The study aimed to determine the antibiogram and molecular characteristics of ESBL-PE fecal carriage from ICU patients at Sanglah Hospital, Denpasar, Bali.
Methods:
This cross-sectional retrospective study involved 30 stored-bacterial isolates of ESBL-PE from a rectal swab of ICU patients who had just been admitted to the ICU of Sanglah General Hospital from February to March 2019, consecutively. The identification and antimicrobial susceptibility test of the isolates were conducted using Vitek-2 Compact (bioMérieux®, Marcy-l'Etoile, France), while genotype identification was conducted using PCR for the detection of blaTEM, blaSHV, blaCTX-M genes.
Results:
Thirty bacterial isolates were identified as Escherichia coli (24/30) and Klebsiella spp. (6/30) and detected as ESBL-producing isolates by Vitek-2 Compact. All isolates were susceptible to piperacillin-tazobactam, meropenem, and amikacin. Twenty-two (73.3%) isolates harbored ESBLs blaTEM, blaSHV, blaCTX-M genes, either individually or in combination. Most of the isolates had the combination of ESBL genes. About 20% (6/30) of isolates had a combination of blaTEM and blaCTX-M, while 10% (3/30) of them possessed all of the three genes detected in this study. Only 3.3% (1/30) of the isolates had each combination of blaTEM and blaSHV as well as blaSHV and blaCTX-M. Meanwhile, 16.7% (5/30) of the isolates were detected to have each single gene of blaCTX-M or blaTEM, and only one isolate (3.3%) harbored blaSHV.
Conclusion:
High prevalence of blaTEM, blaSHV, and blaCTX-M ESBL genes harbored by fecal flora of patients who had just been admitted in ICU give rise to the risk for transmission among critically ill patients in ICU. Fecal screening of ESBL-PE besides infection control can be considered for those patients who have a risk factor of ESBL-PE colonization before they are being admitted to the ICU.
1. INTRODUCTION
The emergence of Multi-Drug Resistance Organisms (MDROs) has become a major health problem worldwide during the last decade. Highly-resistant Gram-negative bacterial infections increase morbidity, mortality, length of stay, and cost in Intensive Care Units (ICUs) [1, 2]. The prevalence of ESBL-PE in ICUs is increasing worldwide [3, 4]. A review of epidemiology and molecular characterization of multidrug-resistant Gram-negative bacteria in Southeast Asia reported that ESBL-producing bacteria were commonly isolated from ICU patients than non-ICU patients in the Philippines and Vietnam [5]. The most prevalent ESBL-encoded genes are blaTEM, blaSHV, and blaCTX-M, especially there has been a significant increase in the prevalence of blaCTX-M in certain regions around the world within the past decade [6, 7]. In Indonesia, several studies have shown a high prevalence of ESBL-PE, in which the CTX-M type was predominantly spread in a hospital setting [8, 9]. ESBL-PE infections in ICUs are commonly associated with fecal colonization of these bacteria, which have been considered as a reservoir of ESBL-PE, including Escherichia coli and Klebsiella pneumoniae [10-12]. Early detection through fecal screening is a method that can detect the colonization of ESBL-PE in the gastrointestinal tract. Furthermore, the molecular epidemiological data of these resistant bacteria in ICU is extremely valuable for efficient containment of the emergence of MDROs. There are only limited studies, so far that determined the molecular characteristics of ESBL-PE among ICU patients in Indonesia, including Bali. Therefore, this study aimed to determine the antibiogram and molecular characteristics of ESBL-producing E. coli and Klebsiella spp. in the ICU of Sanglah Hospital, Denpasar, Bali.
2. MATERIALS AND METHODS
2.1. Study Design and ESBL Identification
The design of this study was a retrospective cross-sectional study. Thirty glycerol stocked- ESBL-PE isolated from thirty ICU patients’ rectal swabs were used in this study. The rectal swabs of the ICU patients were collected in up to 4 hours after admission in the ICU of Sanglah General Hospital from February to March 2019, consecutively. The isolates were identified and tested for antimicrobial susceptibility test using Vitek-2 Compact System (bioMérieux®, Marcy-l'Etoile, France).
2.2. Molecular Detection of blaTEM, blaSHV, blaCTX-M Genes using PCR
The genotypes of ESBL-PE isolates were detected using Polymerase Chain Reaction (PCR). ESBL producingEscherichia coli and Klebsiella spp. (Klebsiella pneumoniae and Klebsiella oxytoca) were cultured on MacConkey agar and incubated for 24 hours at 37°C. DNA was extracted from bacterial colonies using a plasmid DNA extraction kit (QIAprep® Spin Miniprep Kit, Qiagen, Gmbh), following the manufacturer's instructions. DNA template was subjected to PCR to detect blaTEM, blaSHV, blaCTX-M genes with primer sets, as shown in Table 1 [13]. Briefly, the PCR mix (GoTaq® Green master Mix, Promega, Madison, WI, USA) was prepared based on the manufacturer's protocol. The final concentration of the primer set was 0.8 µM each. PCR cycle consisted of pre-denaturation at 95°C for 5 minutes; 35 cycles of denaturation at 94°C for 1 minute, primer annealing at 55°C for 30 seconds, extension at 72°C for 1 minute, and a final extension at 72°C for 4 minutes (Biometra®, TPersonal thermal cycler). Amplicons were electrophoresed in 1.5% agarose gel stained with GelRed® Nucleic Acid Stain (Biotium, Hayward, CA, USA) in TBE buffer. The PCR product was UV-visualized and captured.
3. RESULTS
Thirty bacterial isolates, which consisted of 24 isolates of Escherichia coli and six isolates of Klebsiella spp. (K. oxytoca and K. pneumoniae) were successfully isolated from rectal swabs of ICU patients that were just admitted to the ICU from February to March 2019.
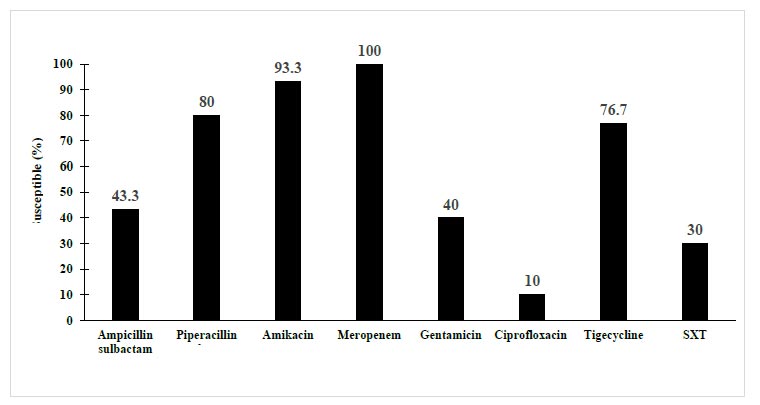
The antibiogram profiles showed reduced susceptibility to gentamicin, ciprofloxacin, and trimethoprim-sulfamethoxazole. This ESBL-producing E. coli and Klebsiella spp. isolates were susceptible to meropenem, amikacin, piperacillin-tazobactam, and nitrofurantoin (Fig. 1). All isolates were successfully grown on MacConkey media. The molecular characteristics of ESBL-producing E. coli and Klebsiella spp. were determined from the results of PCR (Fig. 2) and (Table 2). In this study, twenty-two (73.3%) isolates harbored gene types of ESBLs. Most of the isolates were dominated by a combination of blaTEM and blaCTX-M genes (20%). A single gene of blaCTX-M or blaTEM was detected in 16.7% of isolates, whereas only 1 isolate (3.3%) had a single gene of blaSHV.
| Identified Genes | No. (%) |
| blaTEM | 5 (16.7) |
| blaSHV | 1 (3.3) |
| blaCTX-M | 5 (16.7) |
| blaTEM and blaSHV | 1 (3.3) |
| blaTEM and blaCTX-M | 6 (20.0) |
| blaSHV and blaCTX-M | 1 (3.3) |
| blaTEM, blaSHV, and blaCTX-M | 3 (10.0) |
| Negative | 8 (26.7) |
| Total | 30 (100) |
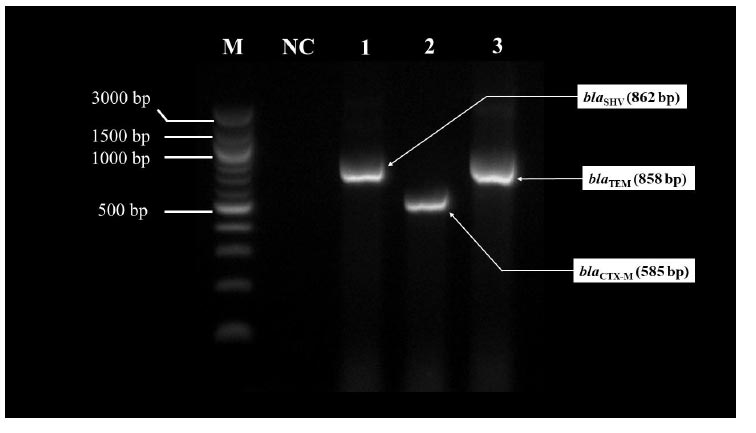
The amplicons were electrophoresed on 1.5% of agarose gel stained with GelRed® Nucleic Acid Stain (Biotium) in TBE buffer at 100 volt, 45 minutes. The gene encoded blaSHV was 862 bp (1), blaCTX-M was 585 bp (2), while blaTEM gene was 858 bp (3). (M = 100 bp DNA Ladder, New England Biolabs; NC = negative control).
In this study, the ESBL-producing E. coli and Klebsiella spp., which possessed blaTEM and blaCTX-M genes, showed similar antibiotic resistance phenotype to ampicillin, cephalosporins, aztreonam, and ciprofloxacin (Tables 3 and 4).
| Organism (Number) | Numbers of Isolates with bla Type | ||
| CTX-M | SHV | TEM | |
| E. coli (n=24) | 12 | 6 | 12 |
| K. pneumoniae (n=5) | 2 | 0 | 2 |
| K. oxytoca (n=1) | 1 | 0 | 1 |
| Isolates that Resistant to Antibiotic | Number of Isolates (Resistant/Total) | Susceptibility (%) | Numbers of Isolates with bla Type (s) | ||
| CTX-M | SHV | TEM | |||
| Ampicillin | 30/30 | 0.0 | 15 | 6 | 15 |
| Ampicillin-sulbactam | 17/30 | 43.3 | 6 | 2 | 8 |
| Piperacillin-tazobactam | 6/30 | 80.0 | 4 | 1 | 6 |
| Cefazolin | 30/30 | 0.0 | 15 | 6 | 15 |
| Ceftazidime | 30/30 | 0.0 | 15 | 6 | 15 |
| Ceftriaxone | 30/30 | 0.0 | 15 | 6 | 15 |
| Cefepime | 30/30 | 0.0 | 15 | 6 | 15 |
| Aztreonam | 30/30 | 0.0 | 15 | 6 | 15 |
| Amikacin | 2/30 | 93.3 | 1 | 0 | 1 |
| Meropenem | 0/30 | 100.0 | 0 | 0 | 0 |
| Gentamicin | 18/30 | 40.0 | 7 | 4 | 7 |
| Ciprofloxacin | 27/30 | 10.0 | 14 | 6 | 15 |
| Tigecycline | 7/30 | 76.7 | 3 | 0 | 3 |
| Nitrofurantoin | 4/30 | 86.7 | 2 | 0 | 4 |
| Trimethoprim-sulfamethoxazole | 21/30 | 30.0 | 11 | 4 | 8 |
4. DISCUSSION
ESBL-producing E. coli and Klebsiella spp. shows reduced susceptibility to cephalosporin and monobactam, common antimicrobial agents used in healthcare facilities [3, 4]. ICU is a critical care unit in which many critical patients are treated and prone to MDROs, including ESBL-PE [14]. Detection of the antibiogram and the molecular characteristics of ESBL-PE from the fecal material of ICU patients, can prevent the spreading of ESBL-PE among ICU patients [15]. In this study, we identified the antibiogram and genotype of ESBL-producing E. coli and Klebsiella spp., isolated from a rectal swab of ICU patients in Sanglah Hospital, Bali, Indonesia. The antibiogram profiles of ESBLs isolates in this study were consistent with a previous study in Indonesia reported by Hasibuan et al., Hayati et al., and Anggraini et al. in which the isolates were sensitive to meropenem, amikacin, and piperacillin-tazobactam [8, 9, 16]. A difference in amikacin susceptibility was found in a study from Thailand by Kiddie et al. [17]. They reported that only 41.9% of ESBL-PE isolates were susceptible to amikacin. ESBL-PE isolates showed less susceptibility to aminoglycosides, chloramphenicol, fluoroquinolones, tetracyclines, and trimethoprim-sulfamethoxazole [8, 9, 12].
The majority of the blaTEM gene found in this study was consistent with a previous study in Malaysia conducted by Thong et al. in which the majority of the ESBL-positive isolates harbored blaTEM (88%) followed by blaCTX-M (20%) [18]. The gene type of blaTEM has been reported to be responsible for 90% of ampicillin resistance in E. coli, while this study also showed that the isolates were resistant to ampicillin with 50% (15/30) detected blaTEM [18]. Multiple genes patterns of blaTEM and blaCTX-M were predominantly found in this study, which is consistent with previous studies in Surabaya, conducted by Naelasari et al. (24%) and in Denpasar, conducted by Fatmawati et al. (15.5%) [19, 20] This pattern was also consistent with another study in Iraq, reported by Pose et al. that blaTEM and blaCTX-M were dominant (30.9%). Such molecular patterns also showed similar antibiotic sensitivity patterns, which were mostly sensitive to meropenem and amikacin [21].
The prevalence of ESBLs type, blaCTX-M was still more dominant than other types in Surabaya that reported 80% and 94.5% in E. coli and 55.6% in K. pneumonia [19, 22]. Other studies in Singapore, Vietnam, and Thailand also showed a prominent spreading of CTX-M type ESBLs. A study in Singapore showed that CTX-M type ESBLs were currently a major resistance contributor, especially in nosocomial infections with Klebsiella spp. and E. coli [23]. Similar to those studies, in this study, CTX-M type prevalence was also as high as TEM type, and combination genes of TEM and CTX-M (20%) were also highly prevalent. This study found that 26.6% (8/30) of ESBLs producing E. coli and Klebsiella spp. were undetected for the three resistant genes. These ESBLs isolates might have other types of ESBLs-encoded genes; therefore, they could not be detected by specific primer sets used in this study.
ESBLs genes, included in this study, could be mostly plasmid-encoded, indicating that these genes were transferable to other Enterobacteriaceae that may give significant problems in infection prevention and control in ICU [24].
The sequence grouping and subtyping of blaTEM, blaSHV, and blaCTX-M, as well as detection of other ESBLs genes, were not performed in this study. Therefore, to confirm our results, a future study for gene typing is needed to be performed. Also, detection of other genes that encode ESBLs is important forfurther characterization. Further investigation of molecular characteristics of ESBL-PE before ICU admission and after discharge from the ICU would be promising to get a good database for risk factor assessment of ESBL-producing bacterial infection and its dissemination. Clonal transmission of the ESBL-PE needs further investigation.
CONCLUSION
In conclusion, our study shows that the gastrointestinal flora of patients who have just been admitted to ICU harbored blaTEM, blaSHV, and blaCTX-M ESBL genes, which increased the risk of transmission among critically ill patients in ICU. Faecal screening of ESBL-PE may be considered for patients who have a risk factor of ESBL-PE colonization before being admitted to ICU. Considering the antibiogram and genotype characteristics of ESBL-PE among screening isolates, especially in a patient who has just been admitted to ICU, is necessary to adopt using suitable controlling tools to prevent further spread of resistance genes in this organism in the hospital setting.
LIST OF ABBREVIATIONS
| ESBLs | = Extended-Spectrum β-Lactamases |
| ESBL-PE | = Extended-Spectrum β-lactamases Producing Enterobacteriaceae |
| ICU | = Intensive Care Units |
| DNA | = Deoxyribonucleic Acid |
| PCR | = Polymerase Chain Reaction |
| MDRO | = Multi Drug Resistance Organisms |
| CLSI | = Clinical and Laboratory Standard Institute |
| bp | = Base Pair |
| TBE | = Tris-Borate-EDTA |
| S | = Sample |
| M | = Marker |
| NC | = Negative Control |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study has been approved by the Committee of Ethical Research of Udayana University, Indonesia (Number 36/UN14.2.2.VII.14/LP/2019).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures were followed in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
All patients provided informed consent in accordance with the relevant regulations in Sanglah general hospital, Denpasar, Bali.
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included within the article.
FUNDING
None.
CONFLICT OF INTEREST
There is no conflict of interest for any of the authors regarding this article.
ACKNOWLEDGEMENTS
The authors thank Ni Made Adi Tarini, (Clinical Microbiology Medical Doctor, Head of Clinical Microbiology Laboratory Department Sanglah General Hospital), Nyoman Sri Budayanti, Clinical Microbiologist, Ni Wayan Nilawati (Clinical Microbiology Laboratory Sanglah General Hospital Staff), Wahyu Hidayati, Ni Wayan Eka Putri Gayatri Kastawa, and Gede Ngurah Rsi Suwardana, (Molecular Microbiology Staff) for their technical support.


