All published articles of this journal are available on ScienceDirect.
Incidence and Bacterial Etiologies of Surgical Site Infections in a Public Hospital, Addis Ababa, Ethiopia
Abstract
Background:
Surgical Site Infections (SSIs) are among the frequently reported healthcare-acquired infections worldwide. Successful treatment of SSIs is affected by the continuous evolvement of drug-resistant microbes. This study investigated the incidence of SSIs, identifying the major etiologic agents and their drug resistance patterns in Yekatit 12 Hospital, Ethiopia.
Methods:
A cross-sectional study was conducted on 649 patients who underwent surgery at Yekatit 12 hospital from April 2016 to April 2017. Socio-demographic and clinical data were collected from each patient on admission. After surgery, they were followed for SSI occurrence. SSI was initially diagnosed by a senior surgeon based on standard clinical criteria and then confirmed by culture. Isolates were tested for drug resistance according to the clinical and laboratory standards institute guideline.
Results:
Of the 649 study participants, 56% were females. Their age ranged from 9 months to 88 years with a median age of 37 years. The incidence of culture-confirmed SSI was 10.2% (66/649) where 73 isolates were recovered. About two-third of the isolates were Gram-positive bacteria. Staphylococcus aureus was the most frequently isolated (27/73, 37%) followed by Coagulase-negative staphylococci (18/73, 24.7%), Escherichia coli (11/73, 15.1%) and Klebsiella species (10/73, 13.7%). About 89% and 44% of S. aureus isolates were resistant to penicillin and trimethoprim-sulfamethoxazole, respectively. MRSA constituted 11% of the S. aureus isolates. All the Gram-negative isolates were resistant to ampicillin and trimethoprim-sulfamethoxazole but susceptible to amikacin and meropenem. Klebsiella species showed 70-100% resistance to ceftazidime, cefuroxime, augmentin, chloramphenicol, ciprofloxacin, cefepime and gentamicin. About 82% of E. coli isolates were resistant for chloramphenicol, cefepime, ceftazidime, augmentin, cefuroxime and 64% for gentamicin and ciprofloxacin.
Conclusion:
The incidence of surgical site infection in Yekatit 12 hospital is 10.2%. Most of the SSIs were due to Gram-positive bacteria. Gram-negative isolates showed high resistance to the most commonly prescribed drugs. No resistance was found for meropenem indicating the absence of carbapenem-resistant bacteria. SSI treatments should be guided by culture and drug resistance test. Better infection prevention practices and continuous surveillance of antimicrobial resistance in the hospital are recommended for better patient care.
1. BACKGROUND
Healthcare-Associated Infections (HCAIs) remain a significant cause of morbidity and mortality [1]. It is estimated that about 10% of hospitalized patients in developing countries acquire at least one healthcare-associated infection. The most common of these is surgical site infection. A surgical site infection is an infection that occurs after surgery in the part of the body where the surgery took place [2]. Besides causing substantial morbidity and mortality, SSI has an economic impact due to prolonged hospitalization, additional diagnostic tests, treatment and sometimes additional surgery [3-5].
Most surgical site infections are preventable through the implementation of proper infection prevention and control measures [6]. For these measures to be successful, local data regarding the SSI burden, etiologies and their drug resistance pattern need to be identified before the intervention [7].
2. METHODS
2.1. Study Area and Period
A cross-sectional study was conducted on 649 study participants who underwent surgery at Yekatit 12 hospital from April 2016 to April 2017. Yekatit 12 Hospital is a public general hospital with about 500 beds and annual patient flow of greater than 150, 000. It is located in Addis Ababa, the capital city of Ethiopia.
2.2. Data and Sample Collection
Information regarding socio-demographic characteristics of study participants (age, sex, marital status, etc.) was collected during admission by trained nurses. The participants were then followed for the occurrence of surgical site infections for 30 days after surgery. From those who developed SSIs based on clinical criteria, swab samples were collected from the infected incision site by trained nurses. Briefly, the infected site was cleansed using normal saline and samples were collected by rotating a sterile culture swab over viable wound tissue with sufficient pressure [8]. The swab samples were placed into asterile tube containing Amie’s transport medium and transferred immediately to the Microbiology laboratory of the hospital for analysis.
2.3. Culture and Identification
The samples were directly inoculated on to blood, MacConkey, and Mannitol salt agar media. The inoculated plates were then incubated aerobically at 35-37oC for 24 hours and then examined for bacterial growth. Bacterial identification was performed based on colony morphology, appearance, color, hemolytic activity, Gram reaction, and different biochemical tests.
2.4. Antimicrobial Susceptibility Testing
Antimicrobial susceptibility testing was performed by the Kirby–Bauer disc diffusion method [9]. Briefly, standardized suspensions of the bacterial isolates were prepared using normal saline and the turbidity was matched with the turbidity standard McFarland 0.5. The standardized suspension was streaked onto to the Muller-Hinton Agar and allowed to air dry. Then antibiotic discs were placed on to the medium and then incubated at 35-37°C for about 18 to 24 hours. The zones of inhibition were measured using a caliper and interpreted according to the Clinical Laboratory Standards Institute, CLSI 2014 [10] criteria as sensitive, intermediate and resistant. The quality of the culture media, gram stain, and antimicrobial discs were checked using standardized reference strains of Escherichia coli (ATCC 25922) and Staphylococcus aureus (ATCC 25923).
2.5. Data Analysis
Data was entered into an excel spreadsheet, cleaned and exported to SPSS software version 20 for analysis. P-value<0.05 was considered as cut off point for the significant association.
2.6. Ethical Consideration
This study was conducted after approval by the Institutional Review Board of Yekatit 12 Hospital Medical College. Written informed consent was also obtained from adults, parents/guardians (for study participants less than 18 years old). Also, consent was obtained for those between 12 and 18 years old. The aim of the study, its significance, confidentiality, their rights of participation, the procedure and associated risks were explained through an information sheet. Results of the isolated organisms and antimicrobial susceptibility testing were promptly reported to the attending physician for better patient care.
3. RESULTS
3.1. Socio-demographic Characteristics
The 649 study participants enrolled in this study were recruited from the department of general surgery, Gynecology and Obstetrics, and ENT. Females constituted a higher proportion (364/649, 56%) compared to males (285/649, 44%). The age of the participants ranged from 9 months to 88 years, with a median age of 37 years. About 61.5% (399/649) of the study participants had elective surgery whereas 38.5% (250/649) had emergency surgery. Overall, 71/649 (10.9%) of them had clinically (SSA) and 66 (66/71, 93%) were culture-confirmed. This resulted in an overall culture-confirmed SSI incidence of 10.2% (Table 1).
3.2. Culture Findings
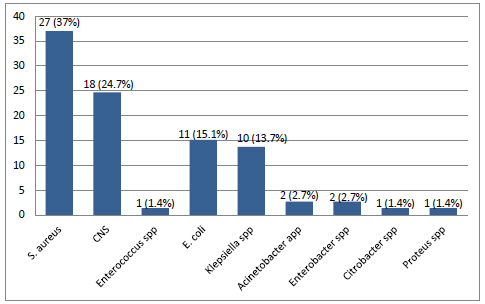
There were a total of 73 bacterial isolates from the 66 culture-confirmed SSIs. Double isolates were detected from seven of the 66 study participants. Among the isolates, Gram-positive bacteria constitute a higher percentage (46/73, 63.1%). Staphylococcus aureus was the most frequently isolated bacteria (27/73, 37%) followed by Coagulase-negative staphylococci (18/73, 24.7%), E. coli (11/73, 15.1%) and Klebsiella species (10/73, 13.7%) (Fig. 1).
3.3. Antimicrobial Susceptibility Profile
S. aureus showed the highest resistance to penicillin (89%) followed by trimethoprim-sulfamethoxazole (44.4%), clindamycin (14.8%), erythromycin (8.5%) and ciprofloxacin (3.7%). Three of the 27 S. aureus isolates (11.1%) were MRSA based on cefoxitin resistance. No S. aureus isolate was found resistant to gentamicin and vancomycin (Table 2).
All the Gram-negative isolates were resistant to ampicillin and trimethoprim-sulfamethoxazole but susceptible to amikacin and meropenem.
In addition, E. coli showed higher resistance to chloramphenicol (81.8%), cefepime (81.8%), ceftazidime (81.8%), augmentin (81.8%), cefuroxime (81.8%), gentamicin (63.6%) and ciprofloxacin (63.6%). The Klebsiella species showed increased resistance to ceftazidime (100%), cefuroxime (100%), augmentin (100%), chloramphenicol (100%), ciprofloxacin (90%), cefepime (80%), and gentamicin (70%). Both of the Acinetobacter species were resistant to chloramphenicol, ceftazidime, cefuroxime, augmentin, trimethoprim-sulfamethoxazole, and ampicillin but susceptible to gentamicin, meropenem, and amikacin. In addition, both the Enterobacter species were resistant to gentamicin, trimethoprim-sulfamethoxazole, cefuroxime, augmentin, and ampicillin (Table 2).
| Variables | Frequency (n) | Percentage (%) | |
|---|---|---|---|
| Hospital department | General surgery | 604 | 93.1 |
| Gynecology and Obstetrics | 23 | 3.5 | |
| ENT | 22 | 3.4 | |
| Sex | Male | 285 | 43.9 |
| Female | 364 | 56.1 | |
| Procedure type | Elective | 399 | 61.5 |
| Emergency | 250 | 38.5 | |
| Clinically suspected SSI | Yes | 71 | 10.9 |
| No | 578 | 89.1 | |
| Culture confirmed SSI | Yes | 66 | 10.2 |
| No | 583 | 89.8 | |
| Antibiotic discs (Oxoid, UK) | Susceptibility pattern | S. aureus (n=27) | Enterococcus spp (n=1) | Klepsiella spp (n=10) | E. coli (n=11) | Acinetobacter spp (n=2) | Citrobacter spp (n=1) | Proteus spp (n=1) | Enterobacter spp (2) |
|---|---|---|---|---|---|---|---|---|---|
| Penicillin, 10 units | S | 3 (11.1) | 0 (0) | NT | NT | NT | NT | NT | NT |
| R | 24 (88.9) | 1 (100) | NT | NT | NT | NT | NT | NT | |
| Erythromycin, 15 µg | S | 22 (81.5) | NT | NT | NT | NT | NT | NT | NT |
| R | 5 (8.5) | NT | NT | NT | NT | NT | NT | NT | |
| Clindamycin, 2 µg | S | 23 (85.2) | NT | NT | NT | NT | NT | NT | NT |
| R | 4 (14.8) | NT | NT | NT | NT | NT | NT | NT | |
| Cefoxitin, 30 µg | S | 24 (88.9) | NT | NT | NT | NT | NT | NT | NT |
| R | 3 (11.1) | NT | NT | NT | NT | NT | NT | NT | |
| Vancomycin, 30 µg | S | 27 (100) | 1 (100) | NT | NT | NT | NT | NT | NT |
| R | 0 (0) | 0 (0) | NT | NT | NT | NT | NT | NT | |
| Gentamicin, 10 µg | S | 27 (100) | NT | 3 (30) | 4 (36.4) | 2 (100) | 1 (100) | 1 (100) | 0 (0) |
| R | 0 (0) | NT | 7 (70) | 7 (63.6) | 0 (0) | 0 (0) | 0 (0) | 2 (100) | |
| Trimethoprim/sulfamethoxazole, 1.25/23.75 µg | S | 15 (55.6) | NT | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| R | 12 (44.4) | NT | 10 (100) | 11 (100) | 2 (100) | 1 (100) | 1 (100) | 2 (100) | |
| Ciprofloxacin, 5 µg | S | 26 (96.3) | 0 (0) | 1 (9) | 4 (36.4) | 1 (50) | 0 (0) | 1 (100) | 1 (50) |
| R | 1 (3.7) | 1 (100) | 9 (90) | 7 (63.6) | 1 (50) | 1 (100) | 0 (0) | 1 (50) | |
| Chloramphenicol, 30 µg | S | NT | 1 (100) | 0 (0) | 2 (18.2) | 0 (0) | 0 (0) | NT | NT |
| R | NT | 0 (0) | 10 (100) | 9 (81.8) | 2 (100) | 1 (100) | NT | NT | |
| Meropenem, 10 µg | S | NT | NT | 10 (100) | 11 (100) | 2 (100) | 1 (100) | 1 (100) | 2 (100) |
| R | NT | NT | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | |
| Cefepime, 30 µg | S | NT | NT | 2 (8) | 2 (18.2) | 1 (50) | 0 (0) | 1 (100) | 2 (100) |
| R | NT | NT | 8 (80) | 9 (81.8) | 1 (50) | 1 (100) | 0 (0) | 0 (0) | |
| Ceftazidime, 30 µg | S | NT | NT | 0 (0) | 2 (18.2) | 0 (0) | 0 (0) | 1 (100) | 2 (100) |
| R | NT | NT | 10 (100) | 9 (81.8) | 2 (100) | 1 (100) | 0 (0) | 0 (0) | |
| Amikacin, 30 µg | S | NT | NT | 10 (100) | 11 (100) | 2 (100) | 1 (100) | 1 (100) | 2 (100) |
| R | NT | NT | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | |
| Cefuroxime, 30 µg | S | NT | 0 (0) | 0 (0) | 2 (18.2) | 0 (0) | 0 (0) | NT | 0 (0) |
| R | NT | 1 (100) | 10 (100) | 9 (81.8) | 2 (100) | 1 (100) | NT | 2 (100) | |
| Augmentin (amoxicillin/clavulanate, 20/10 µg) | S | NT | NT | 0 (0) | 2 (18.2) | 0 (0) | 0 (0) | 1 (100) | 0 (0) |
| R | NT | NT | 10 (100) | 9 (81.8) | 2 (100) | 1 (100) | 0 (0) | 2 (100) | |
| Ampicillin, 10 µg | S | NT | NT | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| R | NT | NT | 10 (100) | 11 (100) | 2 (100) | 1 (100) | 1 (100) | 2 (100) |
4. DISCUSSION
Surgical site infections continue to be associated with substantial mortality, morbidity and additional cost [3-5]. It is even worse in resource-limited settings where infection prevention and control measures are not adequate. Having local data regarding the incidence of surgical site infections, major etiologies, and their drug resistance pattern is of great importance whether to implement a better infection prevention practice and guide empirical therapy of SSIs.
The current study was conducted on 649 study participants who underwent surgery at Yekatit 12 Hospital. Sociodemographic characteristics of the study participants were collected during admission and patients were followed for the occurrence of surgical site infection for 30 days after surgery. Surgical site infection was initially diagnosed clinically by a senior physician and confirmed by bacterial culture. Then, isolates were tested for their drug resistance patterns.
In the present study, a total of 71 patients (10.9%) developed SSI based on clinical criteria. However, only 66 of them were confirmed by culture. The possible explanation for this could be because the etiologic agents might be non-bacterial or anaerobic bacteria as this study addressed only aerobic ones. The overall culture-confirmed incidence of surgical site infection was 10.2%. This is comparable with previous findings of 9.6%-14.8% in Ethiopia [11-13] and 10.6% in Spain [14]. In contrast, the current finding is lower than 20.6% of SSI incidence in Spain [15], 23% in Nepal [16], 17% Egypt [17], and 17%-21% in Ethiopia [18-20]. However, it is higher than 2.5% incidence rate of SSI in Peru [7] and Saudi Arabia [21], 3.3% in China [22], 5% in India [23] and 5.9% in Italy [24]. Such differences could be explained by variations in the distribution of pathogens and infection prevention and control practices.
The majority of the etiologies of SSIs in Yekatit 12 Hospital (63%) were Gram-positive bacteria. Among the etiologies, S. aureus was the most common constituting 37% of the overall isolates. This is in agreement with previous studies in Italy [25], Iran [26], Saudi Arabia [21], Egypt [27] and Kenya [28]. Similarly, previous studies in different parts of Ethiopia had also reported S. aureus as the most frequent isolate from SSIs [19, 20, 29]. These studies found S. aureus comprising from 29% to 40% of the total SSI isolates which is comparable to the finding of the present study; i.e. 37%.
In addition, the present study found E. coli and Klebsiella species from a significant number of patients next to S. aureus which constituted 15% and 14% of the isolates, respectively. Similarly, these bacteria were reported among the frequent isolates from surgical site infections elsewhere. For example, E. coli was the second most isolates constituting 23% of the total isolates in Iran [26]. However, in Egypt Klebsiella pneumonia was the second common SSI isolate [27]. Previous studies in Ethiopia also reported E. coli [19] and Klebsiella species [20] as the most frequent isolates next to S. aureus constituting 27% and 25% of total isolates respectively.
About 90% of S. aureus isolates in the present study were resistant to penicillin. This is in line with previous studies in Ethiopia that reported penicillin resistance ranging from 82-91.5% [29-31]. This is in line with published reports elsewhere indicating that penicillin-resistant S. aureus affected the continent [32, 33].
To overcome the increasing penicillin resistant S. aureus, methicillin was introduced in 1959. However, the first methicillin-resistant S. aureus (MRSA) was reported after two years of its introduction [34]. Shortly thereafter, MRSA became pandemic in many healthcare institutions worldwide [35]. The present study found MRSA in 11% of the S. aureus which is much lower than reports elsewhere that found 60-85% of S. aureus isolates as MRSA [36-38]. This highlights drugs in the oxacillin group could still treat most surgical site infections in the study area. Interestingly, the present study did not find any vancomycin resistance despite its use in the treatment of SSIs in the study area. This drug is recommended to treat MRSA infections [39]. The modestly lower MRSA and no vancomycin resistance in the present study urge to decrease vancomycin prescription in the treatment of staphylococcal infections in the study area.
The current investigation also found that >80% of E. coli isolates were resistant to chloramphenicol, cefepime, ceftazidime, augmentin and cefuroxime, and 63.6% were resistant to both gentamicin and ciprofloxacin. This is in line with previous studies by Giri et al. in Nepal [16] that reported 69.5% of ciprofloxacin, Hafez et al. in Egypt [17] reported >60% ceftazidime resistance, and Mengesha et al. in Northern Ethiopia [20] reported 100% ampicillin resistance by E. coli. In contrast, Mengesha et al. [20] found 0% and 100% gentamicin and augmentin resistance in E. coli, respectively.
The present study found that Klebsiella species showed alarmingly high resistance for multiple drugs; including, ceftazidime (100%), cefuroxime (100%), augmentin (100%), chloramphenicol (100%), ciprofloxacin (90%), cefepime (80%), and gentamicin (70%). A comparable finding was reported by Hafez et al. in Egypt where all Klebsiella isolates were resistant to ceftazidime. Similarly, Mengesha et al. [20] in Northern Ethiopia reported that 100% of the Klebsiella species were resistant to amoxicillin, 93.1% for tetracycline and 86.2% for ceftriaxone. The present study hasn’t found any meropenem and amikacin resistance.
CONCLUSION
The incidence of surgical site infection in Yekatit 12 hospital was 10.2% which was predominantly caused by Gram-positive bacteria. The most common SSI etiology was S. aureus. Escherichia coli and Klebsiella species were the dominant gram negative bacteria. S. aureus isolates showed very high resistance (89%) to penicillin, low resistance (11%) to oxacillin and no resistance to vancomycin. Most Gram-negative isolates were resistant to multiple drugs, including those commonly used in the study area. However, no resistance was documented for amikacin and meropenem. Therefore, SSI treatments should be guided by culture and drug resistance tests or at least current data should be used to guide empirical therapy in the study area. In addition, the implementation of antimicrobial stewardship program is strongly recommended for the prevention and containment of drug-resistant organisms.
LIST OF ABBREVIATIONS
| ATCC | = American Type Culture Collection |
| CLSI | = Clinical and Laboratory Standards Institute |
| CoNS | = Coagulase-negative Staphylococci |
| MRSA | = Methicillin-resistant Staphylococcus aureus |
| SSI | = Surgical site infection |
| WHO | = World Health Organization |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Ethical approval was obtained by the Research Ethics Review Committee of Yekatit 12 Hospital medical college before the conduct of the study and the approval number is 010/2008.
HUMAN AND ANIMAL RIGHTS
No Animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Informed consent was obtained from all the participants prior to publication.
STANDARD OF REPORTING
STROBE guidelines and methodology were followed.
AVAILABILITY OF DATA AND MATERIALS
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
FUNDING
The entire fund of this study was covered by Yekatit 12 Hospital Medical College and the grant number is YK12-336/300/7604.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We would like to thank all the study participants for their kind collaboration to participate in this study. We appreciate nurses, physicians and laboratory personnel of Yekatit 12 Hospital for their unreserved support.


