All published articles of this journal are available on ScienceDirect.
Temporal Variation in Antibiotic Resistance of Acinetobacter baumannii in a Teaching Hospital in Tunisia: Correlation with Antimicrobial Consumption
Abstract
Background:
To investigate the potential correlation between the rates of antimicrobial drug consumption and the prevalence of antimicrobial resistance among clinical Acinetobacter baumannii recovered in a tertiary care hospital in Tunisia.
Methods:
The microbiological and epidemiological profiles of A. baumannii infections at the Hospital Sahloul, Sousse, were investigated between 2001 to 2004 and 2012 to 2015 along with the consumption record of broad-spectrum antibiotics.
Results:
Our data showed that extensively drug-resistant A. baumannii (XDRAb) isolates increased from 11.2% to 30.5% between 2012 and 2015 and disseminated endemically for a long time. Furthermore, we evidenced a drastic increase of carbapenem-resistant A. baumannii isolates from 29.5% in 2001 up to 88.6% in 2015 (612/691). This rise could be paralleled with a significant increase in antibiotic consumption over the last 15 years, especially with the sharp increase in the annual consumption of imipenem (r = 0.816 and p <103). A noteworthy correlation between carbapenem use and resistance rate (r = 0.778, p<0.001) was evidenced.
Conclusion:
Feedback of these data to clinicians and decision-makers in the local setting was crucial to promote the rational use of antimicrobials and to raise awareness to strictly implement hygiene measures to limit the spread of these XDRAb isolates, to prevent colonization and subsequent infection.
1. INTRODUCTION
Acinetobacter baumannii has emerged as one of the most troublesome pathogens for health care settings worldwide [1]. The World Health Organization (WHO) declared it as one of the most serious ESKAPE organisms in public health [2]. They have been involved in a variety of Health Care Associated Infections (HCAIs), associated with extremely high crude mortality rates mainly among patients in intensive care units (ICUs).1,3 Its clinical significance, has been propelled by its capacity to tolerate desiccation and its remarkable propensity to accumulate enzymatic and non-enzymatic resistance mechanisms [1]. The combination of several of these resistance mechanisms, especially those conferring resistance to carbapenems, will limit the therapeutic options for severe infections. Carbapenem-resistant A. baumannii (CRAb) infections are usually resistant to all antibiotics, except colistin [3]. In the present study we (i) describe the microbiological and epidemiological characteristics of A. baumannii isolates identified in the clinical microbiology laboratory in a Tunisian teaching hospital between 2012-2015; (ii) present the trend of the antibiotic resistance rate and correlate them with the antibiotic consumption over a 15-year-period.
2. MATERIALS AND METHODS
2.1. Clinical Settings, Study Design and Bacterial Isolates
A descriptive study was performed at the Sahloul Hospital, Sousse, Tunisia, a 629-bed teaching hospital,-over a four-year period, from 2012 to 2015. A baumannii isolates were identified through the clinical microbiology laboratory data base. Duplicate isolates were excluded, based on antibiotic patterns and time period. Isolates were considered different when complying the following criteria one major and three minor differences in antibiotic patterns and a 30-day time period between two isolates. According to these criteria, a total of 691 isolates were retained and recovered from 573 hospitalized patients.
For further statistical analysis, we retrieved data concerning the resistance features from period ranged between 2001 and 2004 in order to highlight the global evolution of the antibiotic resistance. We considered the period between 2001 and 2004 as period 1 (270 isolates) and that between 2012 and 2015 as period 2 (691 isolates).
2.2. Microbiological Methods
The isolates were identified by conventional methods and using the Vitek 2 automatic system (BioMérieux, Tunis, Tunisia). Antimicrobial susceptibility of A. baumannii isolates was assessed by a disc diffusion method based on MH agar which was performed as recommended by the European Committee on Antimicrobial Susceptibility Testing (EUCAST, 2015, www.eucast.org).
2.3. Definitions
According to the work of a group of international experts following definitions were used in the medical literature to characterize the different patterns of resistance found in healthcare-associated, antimicrobial resistant bacteria [4]:
MDR (for Multidrug-Resistant) was defined as acquired non-susceptibility to at least one agent in three or more antimicrobial categories listed in the worksheet for categorizing isolates.
XDR (for Extensively Drug-Resistant) was defined as non-susceptible to all but two or fewer antimicrobial categories (i.e. bacterial isolates remain susceptible to only one or two categories listed in the worksheet for categorizing isolates).
PDR (for Pandrug-Resistant) was defined as non-susceptible to all agents in all antimicrobial categories listed in the worksheet for categorizing isolates.
2.4. Antibiotic Consumption
Data regarding antibiotic consumption and the occupancy from 2001 to 2015 were collected from the Pharmacy department and statistics office of the hospital, respectively. We focused on broad-spectrum antibiotics, used to treat severe infections with Multidrug resistant A. baumannii isolates (MDRAb). The extracted data were converted to DDD is defined daily dose per 1,000 inpatient-days used according to the Anatomic Therapeutic Chemical classification system defined by WHO Collaborating Centre for Drug Statistics Methodology according to the ATC/DDD Index 2017 (https://www.whocc.no/atc_ddd_index/).
2.5. Statistical Analysis
Descriptive statistics was used for all the studied variables. A chi-squared test was used to compare susceptibility rates between period 1 and 2. A P-value (P) below 0.05 was deemed statistically significant. A linear regression was applied to assess local trends in the consumption of antibacterial agents (P ≤ 0.05 considered for statistical significance). Normality was tested using the Shapiro Wilk test. For the correlation analysis, the nonparametric Spearman correlation coefficient was used. Statistical analyses were performed using SPSS 17.0 Statistical package (SPSS Inc, Chicago, USA).
3. RESULTS AND DISCUSSION
3.1. Demographic and Microbiological Data
A total of 691 A. baumannii were collected over four years in a teaching hospital in Sousse-Tunisia. The majority 536/691 (77.6%) of the isolates were recovered from patients hospitalized in the Intensive Care Units (ICU). The predominant clinical samples that were positive for A. baumannii were blood 264/691 (38.2%), urines 149/691 (21.6%), respiratory tract 78/691 (11.3%) and biopsies, urethral samples, articular fluids, ascites, pleural, sinusual, and synovial fluids 200/691 (28.9%).
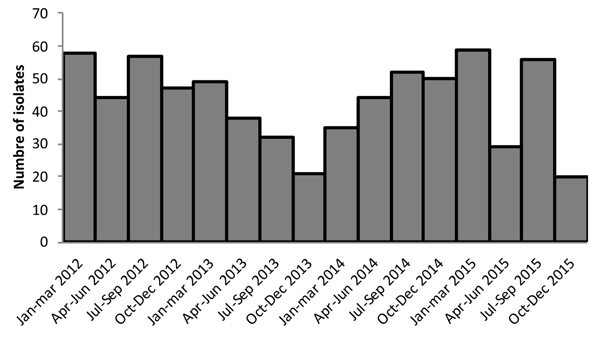
The clinical epidemiologic data obtained by retrospective chart review clearly revealed the endemicity of A. baumannii over a 2012-2015 4-year period, as shown by the epidemic curve (Fig. 1). In fact, A. baumannii can assemble and modulate a host of antimicrobial resistance mechanisms to survive the selective pressure they encounter in the hospital environment, providing them with a strong ecological advantage [1].
Antimicrobial susceptibility testing results revealed a high rate of resistance to most antimicrobials tested, exceeding 80% in most cases. Carbapenem resistance was observed in 612 (88.6%) A. baumannii isolates. Only four antibiotics still revealed some activity (> 50% susceptibility) against these A. baumannii isolates: Co-trimoxazole (Sulphamethoxazole/ trimethoprim), Netilmicin, rifampicin and colistin (Table 1, Table S1).
| Antibiotic |
Period 1 2001-2004 |
Period 2 2012-2015 |
P-value |
|---|---|---|---|
| TIC | 64.6 | 93 | <106* |
| PIP | 85.75 | 91 | 0.019* |
| CAZ | 90 | 89.4 | 0.796 |
| IMP | 40 | 88.6 | <106* |
| GM | 71 | 85.2 | <106* |
| TM | 29 | 62 | <106* |
| NET | 24.5 | 40.6 | <104* |
| AN | 79 | 79.2 | 0.925 |
| OFX | 88 | 91.8 | 0.083 |
| CIP | 88 | 91.7 | 0,083 |
| SXT | 89 | 51.5 | <106* |
| RA | 91.6 | 26.3 | <106* |
| COL | 0 | 0.3 | -- |
| Total | 270 | 691 |
According to the definitions cited below 69.6% (481/691) were MDRAb and 22.6% (156/691) were XDR A. baumannii (XDRAb) (Table S2). The prevalence of XDRAb significantly increased throughout the duration of the study (p < 10-3). The rate of the MDRAb and XDRAb isolates in the ICU is significantly higher as compared to the general hospital setting showing 83% and 79% (p < 10-3) respectively. There were 42 isolates, which remained susceptible only to colistin (27% of the total XDRAb). Only two isolates from 2012 were colistin resistant with MICs > 128mg/L.
In the last decade, the empirical therapy for treatment of A. baumannii infections includes the aminoglycosides, fluoroquinolones, and carbapenems owing to their efficiency and safety against this organism [3, 5]. Our data clearly show an increasing trend of resistance to these antibiotics over the time course of this study, with a resistance rate of 88%. The expanded surveillance network, which encompasses about ten teaching Tunisian hospitals (LART: L’AntibioRésistance en Tunisie) [6] reported 75.7% in 2014. The same high rate was reported in Europe, with generally higher resistance percentages observed in countries in the east and south of Europe than in the north. The combined resistance to all three groups ranged from 83% to 90% in Greece and Italy [7]
To highlight the evolution of resistance locally, we compared susceptibility rates between the 2001-2004 (period 1) and 2012–2015 (period 2) periods. The most remarkable finding of our study was a statistically significant increase of imipenem resistance from 40% (period 2001-2004) to 88.6% (period 2012-2015) (p < 10-6) (Table 1). Similarly, a significant increase in the rate of resistance (p < 0.05) for all the aminoglycosides was observed. Whilst, there was a remarkable decrease (p < 10-6) in sulfamethoxazole and rifampin resistance rate. Only colistin remained constantly efficient against A. baumannii isolates during the study period.
Already, in 2006, the multicenter pilot survey of resistant bacteria in the Mediterranean area brings up the fact that imipenem-resistant A. baumannii seems to be a particularly prevalent organism in Tunisia with 60% of resistance to imipenem especially in ICU [8]. The same alarming findings were reported by Thabet et al. [9] in Tunisia, where the resistance rate to imipenem raised from 63.9% between 2005-2008 to 89.3% between 2008-2011 in the intensive care burn unit and a teaching hospital in the capital. The national survey (LART) [6] reported the rates to imipenem ranging from 44.9% in 2008 to 86.9% in 2014. The European Antimicrobial Resistance Surveillance Network (EARS-Net) report the same high rate in Greece (88-95%) and Italy (87–92%) [7]. Resistance to carbapenems is still lower in Spain (64.4%) in 2014 [7].
Globally, data reported from studies all over the world show that with the exception of polymyxins and tigecyclin, high rates of resistance to all classes of antibiotics have been observed [1, 10, 11]. Indeed, tigecyclin is a broad-spectrum modified minocycline derivative considered as therapeutic alternative showing excellent in vitro activity against MDRAb [12]. Nevertheless, we’ve noted in our series a high rate of resistance ranged from 61.1% to 89.4% and this value is alarming as compared to worldwide studies results. Mendes et al. [11] conducted a large assessment of tigecyclin activity tested against 5127 Acinetobacter spp. They noted a slight increase in tigecyclin resistance rates occurred in Europe (from 1.3-1.5% in 2005-2006, to 3.2-5.8% in 2007- 2008) and Latin America (from 0.4-0.9% in 2005-2006, to 5.3-6.9% in 2007-2008).
| Antibiotic | Antibiotic Consumption (DDD/1000 patient-days) by Year | Correlation | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | r | p | R2 | |
| CAZ | 3.8 | 3.2 | 2.8 | 1 | 3 | 6 | 4 | 3.6 | 3 | 3.5 | 4 | 7 | 3.5 | 7 | 5.5 | 0.574 | 0.025 | 0.330 |
| IMP | 12.9 | 14.5 | 12.6 | 13 | 22 | 27 | 27.5 | 25.6 | 19 | 24 | 25.5 | 23 | 29 | 27 | 32.5 | 0.816 | <103 | 0.665 |
| AN | 9 | 9 | 8 | 4 | 33 | 10 | 11 | 8 | 6.6 | 7.3 | 9 | 8 | 8.5 | 10 | 7 | -0.17 | 0.539 | 0.030 |
| GM | 43.5 | 45.2 | 42.5 | 14 | 33 | 27 | 28 | 24 | 23 | 19.5 | 19 | 16 | 19 | 23 | 23.5 | 0.71 | 0.003 | 0.502 |
| LVX | 0 | 0.1 | 2 | 1 | 3.5 | 5 | 4.5 | 2 | 4 | 3 | 2 | 3 | 4.5 | 3.5 | 3.2 | 0.56 | 0.030 | 0.314 |
| CIP | 17.9 | 23 | 22 | 11 | 34.5 | 37 | 34 | 36.5 | 35.7 | 39 | 33 | 44.5 | 31.5 | 41 | 17 | 0.46 | 0.085 | 0.211 |
| FOS | 1.3 | 3.4 | 4.5 | 2 | 5 | 2.6 | 4 | 7 | 6.5 | 6.3 | 7 | 5 | 10.5 | 8 | 8 | 0.834 | <103 | 0.696 |
| SXT | 7.6 | 5.8 | 5 | 1.5 | 4 | 3.5 | 2 | 4.5 | 3.5 | 3 | 3 | 2.5 | 3 | 4 | 6 | -0.3 | 0.280 | 0.089 |
| RA | 53 | 53.8 | 53 | 23 | 23 | 45 | 46 | 38.6 | 40 | 24.5 | 35.5 | 41 | 44 | 49 | 50 | -0.05 | 0.865 | 0.002 |
| COL | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.3 | 0.2 | 0.3 | 0.2 | 0.3 | 1.3 | 1.3 | 2 | 2 | 0.821 | <103 | 0.675 |
The decrease of resistance of co-trimoxazole was remarkable reaching 50% in periods 2. These rates were lower than several studies where resistance rate to co-trimoxazol within XDRAb were ranged from 97.9% to 100% [13]. We also noted in our study a significant correlation between resistance to co-trimoxazole and phenotype. It shows that resistance for non-MDRAb isolates was 5.5% versus 40.2% for the MDRAb and 91.7% for XDRAb isolates.
*Correlation is significant at the 0.05In this study, colistin has shown a good in vitro activity against A. baumannii isolates. Only two isolates from 2012 were colistin resistant with MICs > 128mg/ml. This is a noteworthy finding since colistin has been revisited as the last defense line against MDR Gram-negative bacilli [16].
The patterns of susceptibility to rifampin in our series reflected a good potency against MDRAb, even though it is not recommended to use rifampin in monotherapy because of the rapid development of resistance. This effect can be blunted by the association with imipenem or sulbactam/ampicillin or colistin [14, 15].
3.2. Trends in Antimicrobial Consumption
To allow comparison of our findings with the previous study we focused only on studies using the same measurement unit (i.e., DDD per 1 000 inhabitants and per day) for reporting antimicrobial consumption. The most prominent finding during the study period was a significant increase in the annual DDD/1000 patient-days of IMP (r = 0.816 and p < 10-3 and a remarkable 20-fold increase in the colistin consumption (r = 0.816 and p < 10-3) (Table 2, Table S3, Fig. S1). Our findings are in line with the ESAC-Net results [17] between 2008 and 2012 that showed a significantly increasing trend in the consumption of imipenem in seven countries (Denmark, Estonia, France, Ireland, Norway, Slovenia and Sweden).
A remarkable 20-fold increase of colistin consumption (r = 0.821, p < 10-3) was noted. Indeed, the dramatic rise of XDR strains may further explain the increase in consumption of so-called “last resort” antimicrobial agent. However, the ESAC-NET reported that there is no significant change at European country level in polymyxin consumption during 2010‒2014, but it increased significantly in three individual countries (Denmark, Hungary and Italy) [17].
3.3. Correlation Between Antimicrobial Consumption and Resistance Rate
Unlike several published studies [18, 19], only imipinem consumption and resistance to this subgroup showed a strong positive correlation (r > 0.72 and P < 0.05) (Figure S1). In fact, carbapenems remained for a long time the only active beta-lactam against MDRAb; this has led to an increase in their use not only for documented infections but also for empirical treatment of acquired hospital infections especially in ICU setting [3, 5]. Our finding support a recent study by Munoz-Price et al. [20] who demonstrated that the risk for acquisition of carbapenem resistant A. baumannii (CRAb) quadrupled with carbapenem exposure and that every additional carbapenem DDD increased the chance of acquiring CRAb by 5.1%. Wide-ranging approach has yielded varying results worldwide, but it is generally assumed that the selective pressure due to the excessive use over the last 50 years of antimicrobials has a significant upshot on bacterial resistance.
CONCLUSION
The escalating problem of antimicrobial resistance in our health care setting is the consequence of the overuse and misuse of antimicrobials leading to increasing selection pressure, and the likely spread of antimicrobial-resistant microorganisms, as recently shown for A. baumannii [21]. Feedback of these data to clinicians and decision-makers at the hospital were crucial to promote the rational use of antimicrobials and to improve compliance with standard and transmission-based precautions and other infection prevention strategies.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
This work was funded by a grant from the Laboratory of Clinical Microbiology and from the UR 12 SP 37 “Emerging Bacterial Resistance and Safety of Care”, Department of Clinical Microbiology, University Hospital of Sahloul, Sousse, Tunisia
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
CONFLICTS OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We thank many dedicated staff who have helped with the data collection and technical issues, in particular: Mourad Achouri (Statistics office of University Hospital of Sahloul, Sousse, Tunisia), Houyem Said (Department of prevention and safety of care of University Hospital of Sahloul, Sousse, Tunisia), Lotfi ben Romdhane (MARS (Modelling of automated Reasoning Systems) Research Lab, Director) ISITCom, University of Sousse, Jihene Maatoug (Department of Epidemiology, University Hospital Farhat Hached, Sousse, Tunis, Tunisia).
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publishers Website along with the published article.


