All published articles of this journal are available on ScienceDirect.
A Case Series and Review of Bacillus Cereus Endocarditis from India
Abstract
Introduction:
Bacillus cereus is a gram positive bacilli found commonly in the soil and environment. It is a bacteria rarely associated with endocarditis.
Case History:
Intravenous drug abuse, presence of valvular defects, pacemakers, immunodeficiency are some of the known risk factors for B.cereus endocarditis. We present here a case series of two patients with B.cereus endocarditis along with a review of the literature.
Conclusion:
This is the first report of B.cereus endocarditis from India to the best of our knowledge.
1. INTRODUCTION
Bacillus cereus is an aerobic gram positive bacilli which is most often considered as a contaminant when isolated from blood culture. This bacteria can also cause deadly diseases in the presence of risk factors such as immunodeficiency history of drug abuse and pacemakers. The bacteria is known for its association with food poisoning, but it can also be responsible for infections such as osteomyelitis, respiratory tract infections, urinary tract infections, cutaneous infections, endophthalmitis, meningitis and rarely endocarditis [1]. Hence the isolation of B.cereus from blood culture must be given due importance in the presence of significant risk factors in the patient’s clinical history.
2. CASE HISTORY
2.1. Case 1
A five month old female baby was admitted for ventricular septal defect (VSD) repair for which she underwent VSD closure with a polytetrafluoroethylene (PTFE) (Gore-tex®) patch. On post operative day two, the baby developed persistent diarrhoea with occasional spikes of fever. Her clinical investigations showed normal haemoglobin(12.6g/dl) total white cell count (14.7K/µl) and erythrocyte sedimentation rate (12 mm/hr) but raised C- reactive protein(CRP)(121.2mg/dl). Sepsis was suspected and blood culture done in pediatric BacTAlert bottle (bioMerieux) yielded gram positive bacilli with central spores which was initially considered as a contaminant.
Two subsequent blood cultures also yielded the same Gram positive bacilli with central spores. The Gram positive bacilli was identified as B. cereus based on morphology on bacterial culture plates and standard biochemical tests. Antimicrobial susceptibility of the isolate was performed using Kirby Bauer disc diffusion testing according to CLSI guidelines [2] and were interpreted using breakpoints for Staphylococcus aureus . The isolate was found to be sensitive to vancomycin, amikacin, teicoplanin, piperacillin/tazobactam, cefoperazone/sulbactam, clindamycin and linezolid. It was resistant to penicillin G and trimethoprim-sulfamethoxazole. The identification of the isolate was confirmed by sequencing the 16SrRNA gene using universal primers [3].
Echocardiography (ECHO) performed on postoperative day four revealed an intact VSD patch with a thin strand like freely mobile hyper-echoic structure attached to the atrial tricuspid leaflet and choroid papillary muscle junction. An additional tiny apical muscular ventricular septal defect was also seen. Patient was thus diagnosed to have infective endocarditis and managed with intravenous vancomycin for ten days and continued with oral linezolid(40mg thrice daily) for 4 weeks. Patient improved with therapy and a repeat ECHO performed at the time of discharge and two weeks after discharge showed no vegetations. The patient continues to remain healthy and asymptomatic at 18 months follow-up.
2.2. Case 2
A 19 year old male presented with high grade fever for the past two weeks. He had been diagnosed with urinary tract infection and was on antibiotic therapy for the past one week with no improvement of symptoms. On clinical examination, the patient had a pansystolic murmur on the mitral area. All other system examinations were within normal limits. His lab investigations revealed normal hemoglobin(10.9g/dl), total white cell count(8400 K/µl) and platelet count(198 K/µl) with raised ESR(59 mm/hr), procalcitonin(0.392 ng/ml) and CRP(32.7mg/l). Paired aerobic blood cultures done in BactTAlert bottles were sterile.
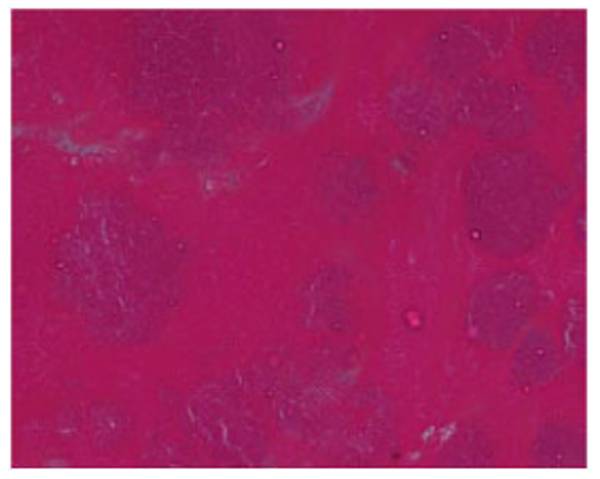
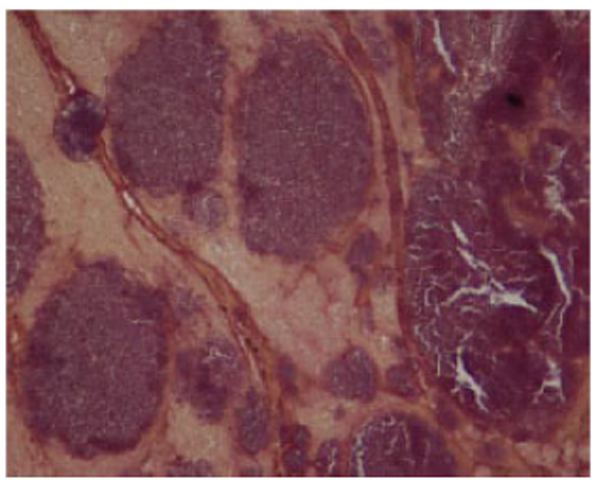
Transoesophageal ECHO showed that the patient had an atrial myxoma measuring 2.16 x 0.8 cm probably arising from the lower part of the inter-atrial septum. The mass was protruding into the mitral valve causing severe eccentric mitral regurgitation. The patient was operated and the mass excised and sent for culture and histopathology. Histopathological examination of the vegetation revealed presence of amorphous eosinophilic material with nodular collections of basophilic round and rod like structures resembling bacterial colonies using Hematoxylin and Eosin stain and nodular colonies of Gram positive cocci and bacilli using Gram stain Figs. (1 and 2).
The excised mass on culture grew a gram positive bacilli which was identified as B.cereus using colony morphology and standard biochemical tests. The isolate was sensitive to gentamicin, vancomycin, tigecycline, and teicoplanin. It was resistant to penicillin G and trimethoprim -sulfamethoxazole. The bacteria was confirmed as B.cereus by matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS) (Bruker Daltonics, Bremen, Germany).
Post-operative period was uneventful. He was started on intravenous vancomycin 1 gm twice daily for 6 weeks. He responded to therapy and at the time of discharge patient had no vegetations on ECHO and subsequent blood cultures were also negative. He continues to remain asymptomatic without any evidence of recurrence at five months of follow-up.
3. DISCUSSION
Bacillus cereus is a a facultative anaerobic, spore forming gram positive bacilli which is found in soil, environment and hospital surroundings. The bacteria when cultured on 5% sheep blood agar produces dull opaque colonies with swarming and beta hemolysis [4]. It produces tissue destructive exoenzymes which are responsible for its pathogenicity. The bacteria is known to cause food poisoning and endophthalmitis. They can rarely cause severe infections such as bacterial pneumonia, brain abscess, meningitis, osteomyelitis, ocular keratitis, necrotizing skin and soft tissue infection . It is known to cause nosocomial acquired bacteremia and wound infections in postsurgical patients with intravascular devices such as catheters [1]. B.cereus isolated from blood culture is most often considered as a contaminant [5]. According to Centres for Disease Control and Prevention(CDC), common skin contaminants such as B.cereus when isolated from blood can be considered as a pathogen if the patient exhibits signs and symptoms of sepsis and two or more blood culture drawn on separate occasions yield the same bacteria [6].
B.cereus is rarely associated with endocarditis. A review of cases reported in literature clearly shows that B.cereus endocarditis is most often seen in patients with history of intravenous drug abuse, central venous catheters, prosthetic heart valves, valvular heart disease, malignancy and immunosuppression. There is one case report of B.cereus endocarditis in a pregnant female with history of intravenous drug abuse [7].
The first case of B.cereus endocarditis was reported in a heroin addict in 1974. Since then there have been 24 reports of B.cereus endocarditis Table (1) [8-27].
| S.No. | Author | Place |
Age/ Gender |
Predisposing Factor | Valve | Treatment | Surgery | Clinical Outcome |
|---|---|---|---|---|---|---|---|---|
| 1 | Craig et al 1974 [8] | USA | 18/F | ASD/DA | TV | Cli/Lm (5 weeks) | No | Recovered |
| 2 | Block et al 1978 [9] | SA | 51/F | PV | MV | Tob/Chl(NR) | No | Died |
| 3 | Tuazonet al 1979 [10] | USA | NR | DA | NR | Naf(NR) | NR | Recovered |
| 4 | Tuazonet al 1979 [10] | USA | NR | DA | NR | Cli | NR | Recovered |
| 5 | Tuazonet al 1979 [10] | USA | NR | DA | NR | Chl, Ery,Gen (NR) | NR | Recovered |
| 6 | Wanvarie et al 1979 [11] | Thailand | NR | RHD | AV | Pen, Gen, Str(NR) | No | Died |
| 7 | Weller et al 1979 [12] | USA | 50/F | DA | None | Cli/Kan(4 weeks) | No | Recovered |
| 8 | Oster et al 1982 [13] | USA | 55/M | PV | AV | Cli/Gen (6 weeks) | Yes | Recovered |
| 9 | Slimanet al 1987 [14] | USA | 43/F | PM, RHD | PM Wire | Cli(6 weeks) | Yes | Recovered |
| 10 | Steen et al 1992 [15] | USA | 34/M | PV | AV | Van(6 weeks) | Yes | Recovered |
| 11 | Tomomasaet al 1993 [16] | Japan | 12 month/F | IS, Ins | MV | NR | No | Recovered |
| 12 | Yamamura et al 1994 [17] | Japan | 43/M | PV | MV | Amk,Min(NR) | Yes | Recovered |
| 13 | Martin et al 1998 [18] | Spain | NR | PV | MV | Gen, Rif,Van(NR) | Yes | Recovered |
| 14 | Castedoet al 1999 [19] | Spain | 45/F | PV | MV | Gen, Rif,Van(6 weeks) | Yes | Recovered |
| 15 | Cone LA et al 2005 [20] | USA | 38/M | ALL | MV | Pen, Van(3 weeks) | No | Died |
| 16 | Abusin S et al 2008 [21] | USA | 69/F | PM | Wire | Cfz(6 weeks) | No | Recovered |
| 17 | Thomas et al 2012 [22] | USA | 42/M | None | AV | Cro(6 weeks) | Yes | Recovered |
| 18 | Barraud O et al 2012 [23] | France | 65/M | PM | Wire | Ofx, Pip(4 weeks) | Yes | Recovered |
| 19 | Oh DH et al 2012 [24] | Korea | 54/M | VHD | MV | NR(6 weeks) | Yes | Recovered |
| 20 | Sharma et al 2013 [25] | USA | 5month/F | ALL | Junction of SVC and RA | Van, Mero(6 weeks) | No | Recovered |
| 21 | Ngow HA et al 201326] | Malaysia | 31/M | DA | AV | Cxm(6 weeks) | No | Recovered |
| 22 | Kitazawa et al 2015 [27] | Japan | 66/M | None | AV | Van(9 weeks) | Yes | Recovered |
| 23 | Shah M et al 2015 [7] | USA | 30/F | DA, Preg | TV | Van(6 weeks) | No | Recovered |
| 24 | Wright et al 2016 [4] | USA | 27/M | MP-CVC | TV | Van(6 weeks) | Yes | Recovered |
| 25 | Present study | India | 5 month/F | VSD | TV | Van, Lin (4 weeks) | No | Recovered |
| 26 | Present study | India | 19/M | Nil | MV | Van (6 weeks) | Yes | Recovered |
ALL, acute lymphoblastic leukemia; ASD, atrial septal defect; AV, aortic valve; DA, drug abuse; IS, immunosuppression; Ins, instrumentation;MP-CVC, medical port central venous catheter; MV, mitral valve; Preg, Pregnant; PM, pacemaker; PV, prosthetic valve; TV, tricuspid valve; RA, right atrium; RHD, rheumatic heart disease; SVC, superior vena cava; VHD, valvular heart disease; VSD, ventral septal defect
Amk, amikacin; Cfz, cefazolin; Chl, chloramphenicol; Cip, ciprofloxacin; Cli, clindamycin; Cro, ceftriaxone; Cxm, cefuroxime; Ery, erythromycin; Gen, gentamicin; Kan, kanamycin; Lm, lincomycin; Mero, meropenem; Min, minocycline; Naf, nafcillin;Ofx, ofloxacin; Pen, penicillin; Pip, piperacillin; Rif, rifampicin; Str, streptomycin; Sxt, trimethoprim sulfamethoxazole; Tob, tobramycin;Van, vancomycin.
B.cereus endocarditis affects males more than females and is seen most often among the age group 41-60 years. Among the cardiac valves, mitral valve is more commonly involved followed by aortic and tricuspid valves. B.cereus endocarditis in majority of cases occurs after an episode of bacteremia, 88% of patients with endocarditis present with fever. Cutaneous colonisation, contamination of drug agents and injection equipment are the causative agents of bacteremia [4]. Kato et al. report 20% mortality among patients with B.cereus BSI [28]. Mortality rate of B.cereus bacteremia is higher in neonates compared to adults [29].Patients with prosthetic valve experience higher morbidity and mortality compared to those with native valve [22].
In most of the previous studies, prosthetic valve associated B.cereus endocarditis were treated using a combination of surgery and antimicrobial therapy and native valve endocarditis were treated with antimicrobial therapy alone. In our study, we report one native valve endocarditis which was treated with surgery and antimicrobial therapy. Surgery was necessitated due to the large size of the mass which was initially diagnosed as an atrial myxoma. Oh et al, also report a case of native mitral valve endocarditis caused by B.cereus where surgery and antibiotics were used for therapy [24].
Antimicrobial therapy for B.cereus endocarditis is given for a period for 6 weeks. Monotherapy as well as combined therapy depending on the antimicrobial susceptibility of the isolate can be used. Though the bacteria is generally sensitive to vancomycin, aminoglycosides, imipenem, tigecycline, ciprofloxacin, chloramphenicol and linezolid, vancomycin appears to be the drug of choice [1, 4].The bacteria secretes beta-lactamase enzyme which is responsible for its resistance against penicillin and cephalosporins [1].
CONCLUSION
This study underscores the significance of clinical correlation of B.cereus isolation from specimen cultures before reporting them as contaminants. B.cereus, which was so far believed to be hospital and environmental flora have now emerged as pathogens which can cause invasive disease.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Consent for publication obtained.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
All authors have contributed substantially to the reporting of the study.


