All published articles of this journal are available on ScienceDirect.
Application of MALDI-TOF MS for the Identification of Food Borne Bacteria
Abstract
Matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS) has recently emerged as a powerful tool for the routine identification of clinical isolates. MALDI-TOF MS based identification of bacteria has been shown to be more rapid, accurate and cost-efficient than conventional phenotypic techniques or molecular methods. Rapid and reliable identification of food-associated bacteria is also of crucial importance for food processing and product quality.
This review is concerned with the applicability of MALDI-TOF MS for routine identification of foodborne bacteria taking the specific requirements of food microbiological laboratories and the food industry into account. The current state of knowledge including recent findings and new approaches are discussed.
INTRODUCTION
In recent years Matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS) has revolutionized routine identification of bacteria. Rapid and reliable identification of food-associated bacteria is of crucial importance for product quality. The challenges for food analytical laboratories are as varied and numerous as are the requirements for bacterial identification methods. Food spoilage bacteria, food-borne pathogens, starter cultures, and probiotics must be among other requirements, accurately identified. Only in this way can fermentation failures be prevented and routes of contamination be uncovered in order to prevent recontamination and to improve the hygiene of production plants. The requirements for identification methods concerning taxonomic resolution and sensitivity differ considerably depending on the task. Whereas it is generally sufficient to identify food spoilage bacteria to the species level, the complex technological features of starter cultures are strain specific. According to statutory provisions and the type of food analysed, highly sensitive detection methods are required for the detection of hygienic indicator bacteria, while the assignment to a certain group like coliforms or enterococci may be sufficient in other cases.
Currently, traditional culture-based methods are still used to identify the majority of food-associated bacteria in the daily routine of food microbiology laboratories. Complete identification requires at least two days, or more for fastidious organisms. In addition, differentiation of isolates with a different taxonomic background but similar physiological characteristics is limited using these phenotypic methods. Therefore, traditional phenotypic methods are - to some extent - supplemented with serological and genotyping methods. Generally, serological methods do not require pure cultures, but have a low-level of discriminatory power and are restricted to species for which antisera are available. In contrast, genotyping methods enable rapid identification and are highly sensitive and specific, but require high levels of technical expertise and remain expensive. Therefore, they are not suitable for routine identification.
MALDI-TOF MS, a chemotaxonomic method, also allows rapid identification of bacteria. In contrast to genotyping methods, it can be easily implemented into routine analysis. All types of food-associated microorganisms can be processed with the same protocol. Reliable data are available within a total turnaround time of 16 hours. Due to short turnaround times, low sample volume requirements and low reagent costs, MALDI-TOF MS has recently emerged as a powerful tool for the identification of clinical isolates. The application areas of MALDI-TOF MS-based microbial identification reach far beyond human clinical use. Recently, the platform has successfully been used to improve veterinary diagnostics [1-3] and to classify environmental isolates [4, 5]. This paper evaluates the applicability of MALDI-TOF MS to food-associated bacteria, while taking the specific requirements of food microbiological laboratories and the food industry into account.
FUNDAMENTALS OF THE MALDI-TOF MS ANALYSIS
MALDI-TOF MS Technology
MALDI-TOF MS detects many different biomolecules, such as nucleic acids, peptides, proteins, sugars and small molecules. This technology identifies microorganisms via the generation of fingerprints of highly abundant proteins followed by correlation to reference spectra in a database. The basic principle of all mass spectrometric methods is the ionization of a neutral molecule and the subsequent accurate determination of the resulting primary ions and their decay products in high vacuum. A typical mass spectrometer is composed of three components: an ion source, a mass analyzer, and the detector.
LDI-MS
Laser desorption ionization (LDI) MS techniques have been used since the 1960s [6]. Initially LDI MS was limited to the analysis of small molecules. The high levels of direct irradiation in combination with the low volatility and thermal instability of biomolecules caused excessive fragmentation. Therefore, larger molecules, such as bacterial membrane proteins, could not be clearly identified. The crucial breakthrough came in the mid-1980s with the development of matrix-based LDI technology [7, 8].
MALDI-MS
MALDI ionizes biomolecules without degradation by embedding them within crystals of small organic compounds. The analyte is first co-crystallized with a matrix compound in high molar excess. Suitable matrices for protein analysis are: 2,5-dihydroxybenzoic acid (DHB), α-cyano-4-hydroxycinnamic acid (CHCA) or 3,5-dimethoxy-4-hydroxycinnamic acid (sinapinic acid, SA). The small organic compound absorbs the radiation from a UV laser and transfers the energy to the proteins. The proteins become ionized without degradation. The ionization step in MALDI can be described as a two-step mechanism consisting of a primary and a secondary ionisation [9]. Ideally, the energy of the laser pulse is exclusively absorbed by the matrix. The matrix vaporizes and transfers ions (generated by the irradiation) to the analyte, resulting in desorption of the analyte. The gentle ionization produces only a singly-charged ion from each type of molecule (Fig. 1). Once in the gas phase, the charged molecules are then accelerated in an electric field, separated in a mass analyzer according to their mass-to-charge value (m/z), and quantified by a detector.
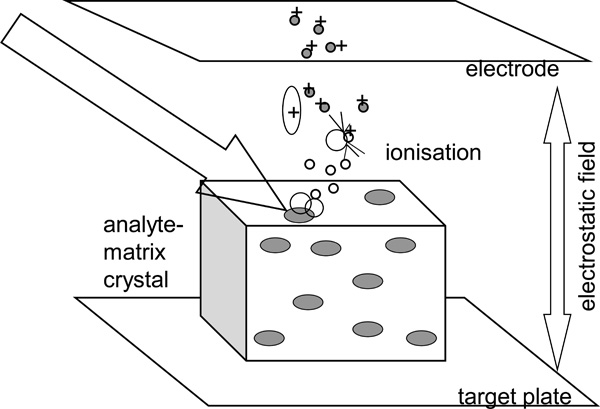
Principle of the MALDI method. The analyte-matrix crystals are irradiated with a pulsed laser. This leads to the desorption of the analyte molecules and protons are transferred from the matrix to the analyte (modified according to [10]).
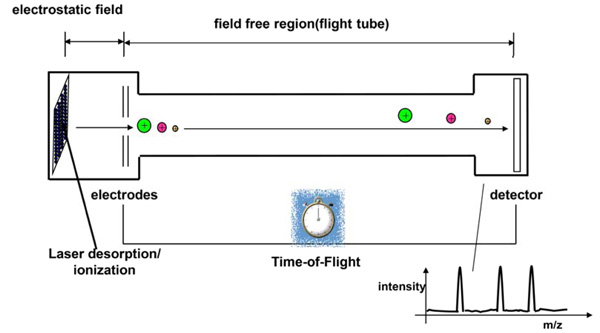
MALDI analyses with a time of flight mass analyzer (modified according to [10]).
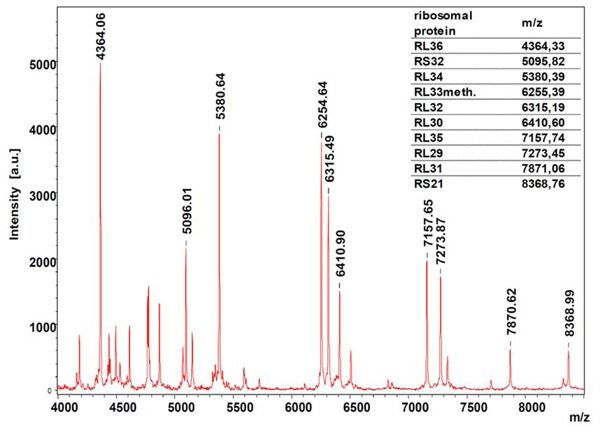
Protein fingerprint of an Escherichia coli isolate. The assignment of the main peaks to the m/z ratios of the ribosomal proteins of E. coli indicates that ribosomal proteins are principal components in the MALDI-TOF MS based identification of bacteria (modified according to [12]).
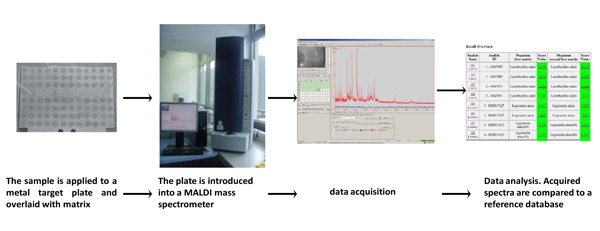
Identification of bacteria on the basis of mass spectrometric protein fingerprints. Microorganisms are pre-treated as described or directly applied onto a target plate and overlaid with matrix. The target is introduced into the mass spectrometer to record the fingerprints in the range of about 2 000-20 000 m/z. A typical mass spectrum is shown. Data are analysed automatically.
TOF Analyzers
Different types of detectors are available. In MALDI-TOF MS, time-of flight (TOF) analyzers are used. TOF analyzers exploit the fact that all ions accelerated in the same electric field have the same kinetic energy. After leaving the electric field they enter a field-free section (the flight tube) and have different speeds depending on their mass. Large ions take more time to traverse the flight tube than smaller ions. The construction of a TOF mass spectrometer is shown in Fig. (2).
PROTEOMIC PROFILING
Mass spectrometric identification of microorganisms is based on their proteome. The proteome is defined as the complete set of proteins synthesized by the cell at a certain moment. The proteome changes with changing growth conditions. Incubation temperature, culture media as well as incubation times should be kept as stable as possible for reliable, reproducible identification results. For bacteria, most of the biomarkers used for identification are ribosomal proteins [11-12; Fig. (3)].
As the content of ribosomal proteins is known to change under nutrient deficient or cold-stress conditions, fresh samples should be used for mass spectrometric analysis. In order to avoid misidentification, cultures should not be exposed to cold storage. Microorganisms that cannot be identified immediately by MALDI-TOF MS should be stored in 70% ethanol.
WORKFLOW FOR MALDI-TOF MS ANALYSIS
Sample preparation is a crucial factor for accuracy and repeatability of MALDI MS measurements. Variations and abbreviations in the protocol can affect the outcome. As for other culture-based identification methods, MALDI-TOF MS analysis can include a conventional enrichment step in order to increase the recovery of injured cells and to obtain pure cultures for subsequent analysis. If no pre-enrichment is needed, MS analysis can be run directly from single colonies. Generally there are three possible approaches for cell disruption. Samples can be either prepared by using the direct smear method, on target-extraction, or an acetonitrile / formic acid extraction after an ethanol purification step.With the direct smear method, a single colony is picked and deposited on the target plate as a thin layer. The sample is allowed to dry and then covered with the matrix. After drying, MALDI-TOF analysis can be performed.
Some isolates might require additional cell disruption. Addition of 1 µl of 25% formic acid to the smeared isolate on the target plate enhances extracted protein amounts and peak intensities, resulting in more successful outcomes for certain groups of microorganisms such as fungi or Bacillus species. Isolates which are difficult to smear due to their texture on solid media, including some Corynebacterium and Arcobacter strains, or microorganisms grown in liquid media should be prepared according to the ethanol-formic acid extraction. A small volume of the isolate is first suspended in 300 µl of distilled water before 900 µl of absolute ethanol are added, mixed and centrifuged. The resulting pellet is allowed to dry completely before the extraction is carried out. For cell disruption 70% formic acid and acetonitrile are added to the pellet in a 1:1 ratio and centrifuged again. One microliter of the supernatant is added to the target plate, allowed to dry and overlaid with one microliter of matrix solution. The matrix type depends on the analyte. For protein analysis, CHCA and SA have become favoured matrices.
After matrix and analyte co-crystallization on target, the target plate is inserted in the MALDI-TOF MS instrument and the protein fingerprints are generated. Depending on the quality and purity of the sample, the database complexity, and the number of reference spectra in the database, the identification of the isolate is completed within a few seconds to a few minutes after a culture isolate is obtained. The identification is based on protein fingerprints. Certain components of the spectra are specific for a certain genus, species or even a subspecies. The fingerprints of the sample are then compared with reference spectra in the database. The integrated software generates an outcome list, in which species with the most similar fingerprints are ordered according to their logarithmic score value (log (score value)). For the MALDI-Biotyper™ system log(score values) higher than 2.0 indicates species identification, while log(score values) between 1.7 and 2.0 are sufficient for identification of the genus. For values below 1.7, no isolate identification is possible. A typical result list is shown in Fig. (4). The robustness of the identification process is increased by using only a limited number of reproducible peaks with high intensities. In contrast, if all recorded peaks were used then the outcomes might be more influenced by growth media or variations in the exact growth temperature.
The quality of the database is a further critical factor influencing the accuracy of the identification. The use of reference strains with a well-defined taxonomic status is a prerequisite for a reliable identification. Furthermore, the accuracy of the species identification increases with the number of reference spectra present for each species [13].
REQUIREMENTS APPLICABLE TO METHODS FOR ROUTINE IDENTIFICATION OF FOOD-ASSOCIATED MICROORGANISMS
The applicability of MALDI-TOF MS for routine identification of food-associated microorganisms depends on the following factors: performance of the method, economic considerations, and organisational criteria. First of all, the identification method should provide reliable and reproducible identification results for a broad spectrum of food-specific microorganisms. The sample preparation and analytical procedure should be as simple and consistent as possible for different types of microorganisms and for the degree of taxonomical resolution required.
Economic considerations include all aspects affecting cost and expenditure of the method. For example, routine assays should be automatable to increase throughput, thus reducing labour costs and improving reproducibility. Rapid analysis results in fast turn around times, which benefits overall food quality and safety, particularly in situations where a food poisoning outbreak requires rapid identification. Low costs of consumables per sample also helps to reduce the cost to the food safety supply chain. Organisational criteria include the ease of implementation of the analysis platform into operational structures. Accreditation is also an important factor for science-based organisations.
In the face of these demands, the MALDI-TOF MS based identification has a considerable edge over common phenotypic methods. First of all, MALDI-TOF MS can provide more rapid results. These are generally available within a total turnaround time of 16 hours, while phenotypic methods require at least 48 hours from sample to identification.
The time factor plays a key role in food processing. Contaminations during the production process might go unnoticed for days and might adversely affect product quality before information about hygiene is available. Large and expensive storage capacities have to be provided until the products are released to the market.
While MALDI-TOF MS based identification of microorganisms provides the advantage of a universal sample preparation platform for bacteria, fungi, and yeasts, traditional biochemical assays require organism specific procedures, reagents, and kits. These differences in sample preparation procedures are reflected in the direct and indirect costs for consumables, making biochemical assays considerably expensive. Additionally, MALDI-TOF based identification is remarkably low in operational costs - minimal requirement of matrix and reagents for the optional extraction (ethanol, formic acid, and acetonitrile). Storage costs associated with fridges and freezers are significantly reduced compared to traditional phenotypic methods. In addition, simple sample handling during analysis enables greater automation and high throughput.
MALDI-TOF MS identification results can be directly transferred into a Laboratory Information Management System (LIMS). Complete and reliable sample traceability can be easily achieved. Sample and batch traceability is crucial to ensure and monitor product quality in terms of an effective quality management for well-organized recalls with limited product loss.
Variable multifaceted tasks and challenges for food microbial laboratories require flexible methods specific for food-associated microorganisms. While common biochemical systems like API® kits were designed for clinical purposes, current database versions for MALDI-TOF MS based identification include a growing number of food specific species and variants. The databases can be easily expanded to meet the specific requirements of particular laboratories.
One potential drawback of MALDI-MS analysis is the instrument cost. The MALDI platform may find favour in higher throughput laboratories or laboratories that employ MALDI-TOF instruments for analysis of other food components. However, considering the extremely low cost of consumables and the potential to reduce labour costs, the total per sample cost may actually be reduced in the long run, compared to other methods.
MALDI-TOF MS is based on the principle of chemotaxonomic methods because it is based on differences and similarities in certain biomarkers. For a reliable identification result, comparison with reference data is always necessary. Much depends on the quality of the reference data, reference protein fingerprints, and the algorithm for identification. The algorithm for automated identification of isolates in the Biotyper™ application was developed in terms of a robust and reliable identification procedure. A limited number of high intensity peaks, mainly assigned to ribosomal proteins [11] are chosen as biomarkers. Closely related species may be delineated with Biotyper™ compatible software tools, such as ClinProTools (Bruker Daltonics) or BioNumerics (Applied Maths, Belgium). Our own experience demonstrates that the same spectra generated for Biotyper™ analysis can be fed into the above-mentioned software tools [14] for further differentiation.
EXPERIMENTAL EXPERIENCE IN IDENTIFICATION OF FOOD ISOLATES
Numerous studies demonstrate, that MALDI-TOF MS based identification is a rapid and reliable method for routine identification of bacteria [13, 15-17], yeast and fungi [18-21] from clinical samples. Indeed, information concerning the general performance of the MALDI-TOF platform can be readily evaluated, but the specific requirements of food microbial laboratories are generally not considered. Until now the focus of MALDI-TOF MS based identification of food-associated bacteria has been for food pathogens like Campylobacter spp., Cronobacter spp., Listeria spp., Salmonella spp. and Vibrio spp. [22-29] and their clinical isolates. Only a few reports show MALDI-TOF MS for the identification of isolates from food and beverage sources. These reports evaluated multiple aspects of the applicability of MALDI-TOF to food microbiology and ranged from the classification of lactic acid bacteria in fermented meat [30] and the surveillance of probiotics in yoghurt [31], to strain identification and characterisation of biogenic amine-producing bacteria [32].
The very different requirements for the discriminatory power of MALDI-TOF MS become apparent regarding these studies. For example, reliable species identification for foodborne pathogens in seafood was possible, while other studies addressed the need to identify starter cultures like lactic acid bacteria beyond the species level. However, subspecies identification was not achieved using the automated identification algorithms. Specific biomarkers had to be identified. Zeller-Péronnet et al. [14] used BioNumerics and ClinProTools for reliable and reproducible classification of subspecies of Lactococcus lactis. Some of the relevant biomarkers were already described by Tanigawa et al. [33]. Cluster analysis of the m/z peaks list for the probiotic B. animalis identified potential markers for its subspecies [34]. Beyond subspecies level, Kuhns et al. [35] recently demonstrated that MALDI-TOF MS is promising for discrimination of Salmonella enterica serovars. In our laboratory, we have been able to differentiate 20 Enterococcus faecium strains on the basis of 23 peaks (unpublished work). However, MALDI-TOF MS already allows for rapid and reliable identification of species, which are hard to delineate using geno- and phenotypic techniques as shown for Cronobacter spp. [29].
Our own studies revealed that major obstacles concerning the mass spectrometric identification of food isolates using the Biotyper™ application software are attributed to the database. An update of the database with reference spectra from food specific variants and strains improved the identification rate.
An innovative approach expands the possible applications of generated fingerprints in food microbiology. Nicolaou et al. [36] very recently successfully quantified the microbial spoilage load in milk and pork meat using MALDI-TOF MS in combination with multivariant analyses. Quantification was directly performed in the food matrix without subculturing. Both types of food were allowed to naturally spoil for 168 and 78 h before MALDI-TOF analysis.
CONCLUSION
In summary MALDI-TOF MS is a promising platform for quick, flexible, and reliable identification of food microbial isolates. The method complies with a variety of requirements for food microbial laboratories. Particularly the simple protocol and shortened analysis time assist in the maintenance of high levels of food safety. Exact knowledge of the potentials and pitfalls of mass spectrometric microbial identification is a prerequisite for successful implementation into and substitution of existing processes in food microbial laboratories. As a standard application for typing and identification at the subspecies, strain, and serovar level, mass spectrometric protein fingerprinting has great potential. Database entries will continue to include more organisms associated with food microbiology and analysis methods should continue to be simplified. The approach of direct quantification of microbial spoilage load, using MALDI-TOF analysis as a useful platform in food processing promises to simplify microbial food analytics.
ACKNOWLEDGMENTS
The authors would like to acknowledge the financial support of the Bavarian State Ministry of the Environment and Public Health. We thank Dr. Azuka Iwobi for critically reading our manuscript.


