All published articles of this journal are available on ScienceDirect.
Phage Therapy: Clinical Applications, Efficacy, and Implementation Hurdles
Abstract
The increasing resistance of bacteria to antibiotics has brought back attention to phages with the need to determine their efficacy and possible use in treating antibiotic-resistant infections. Over the years, there has been a gradual uptick in the integration of phages for treatment, although primarily centered around compassionate usage. Despite the recognition of phage’s capacity to counter bacterial infections over a century ago, utilizing with this therapy has encountered numerous impediments. Currently, the efficacy of phages finds validation in numerous favorable case studies centered on individual patients. Nonetheless, a necessity exists for conducting clinical trials and streamlining the research and development procedures to ease the assimilation of these experimental treatments into a widely accepted adjunct to antibiotics. Integrating bacteriophage therapy into standard clinical practice will necessitate substantial, dedicated investments from research institutions and healthcare establishments, demanding collaboration with the international scientific and medical communities. This endeavor should encompass consensus among regulatory agencies and an adaptable perspective that evolves alongside our deepening understanding of the nuances of this promising therapeutic approach.
1. INTRODUCTION
Therapeutic options available to the medical community have been significantly reduced due to antibiotic resistance (AR). Arguably, one of the foremost global health issues today centers on the daunting challenge posed by antibiotic resistance. An increasing array of infections is steadily growing more difficult to address, presenting a significant hurdle in effective treatment [1]. The infections associated with antibiotic-resistant pathogens are usually accompanied by substantial morbidity and mortality, along with a massive economic burden on global healthcare [2].
The frightening rate of the evolution, persistence, and spread of antibiotic resistance demands novel and effective therapies to treat multidrug-resistant infections. With AMR becoming a global health threat, phage therapy has experienced a resurgence of interest. The growing AMR crisis has fueled research into alternatives, with one of the most prominent approaches being bacteriophage therapy. Several alternative therapies have been put forward in treating antibiotic-resistant infections, including exploring phytochemicals, silver nanoparticles, phage-encoded enzymes, immunotherapy, antimicrobial peptides, probiotics, fecal microbiota transplantation (FMT), and bacteriophage therapy. Phage therapy is particularly promising since it can target specific bacterial strains, minimizing damage to beneficial bacteria.
Phage therapy is defined as the targeted application of virulent phages, which are viruses proficient in infecting and reproducing within bacterial cells, into a patient’s system. This intervention is undertaken to disrupt the bacterial pathogen responsible for a clinically significant infection [1]. Phage therapy involves using strictly lytic phages in treating bacterial infections [3]. Bacteriophages are inherent bacterial predators that have undergone co-evolution alongside bacteria for nearly 4 billion years [4].
As far back as 1896, researchers had the perception of the bacteriolytic activity of phages. It was not until 1917 that the French Microbiologist Felix D’ Herelle while working at Institut Pasteur in Paris, observed that phages recovered from the stool of patients who suffered from Shigella-mediated bacillary dysentery demonstrated a specific bacteriolytic activity against Shigella [5]. He later prepared phage solutions for treating shigellosis and digestive and skin infections caused by Shigella, Staphylococcus and Streptococcus. He prepared cocktails of bacteriophages to treat different clinical syndromes. However, the phage stability within the cocktail was obscure. By the end of the 1920s, d’Herelle had developed phage therapy for the treatment of staphylococcal infections, furunculosis, phlegmons, and osteomyelitis at Yale University, USA. However, between 1930 and 1940, in the USA, two challenges prevented the adoption of phage therapy. These challenges had to do with the quality of the medication (phage cocktail) and the poor stability of the phages. In some instances, phages were undetectable in phage preparations to be used for treatment.
The present review discusses the clinical applications, effectiveness, and implementation challenges of phage therapy. Emphasis will be on skin and soft tissue infections, respiratory tract infections, recurrent urinary tract infections, and gastrointestinal tract infections.
2. MECHANISM OF PHAGE ACTION
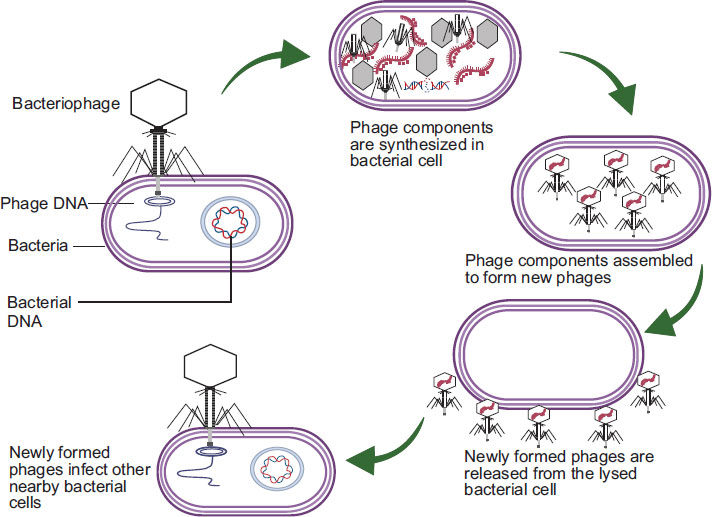
The initiation of infection by lytic phages involves binding to specific receptors located on the surface of the host bacteria. These receptors may be found on cell walls, either Gram-positive or Gram-negative, as well as on polysaccharide capsules or even on various cellular appendages like pili and flagella [6]. After establishing attachment, the virus proceeds to release its genetic material into the host. Subsequently, the virus takes control of the bacterial replication machinery, facilitating the production of the succeeding generation of phage progeny. This replication process persists until phage-encoded proteins are activated to induce cell lysis, ultimately terminating the host cell, allowing the newly synthesized viruses to escape and initiate the cycle once more, as shown in Fig. (1).
3. CLINICAL APPLICATIONS OF PHAGE THERAPY
Antibacterial Resistance Leadership Group (ARLG) Phage Task Force was convened in 2020 by the National Institute of Allergy and Infectious Diseases (NIAID). The Phage Task Force is comprised of experts in the field of phage therapy, clinical microbiology, antimicrobial resistance, and pharmacology. The report from the ARLG Phage Task Force suggests that experimental phage therapy can be considered for a variety of infections, including respiratory tract infections, infections involving devices that cannot be removed, osteoarticular infections, recurrent urinary tract infections (UTIs), gastrointestinal infections, endovascular infections, chronic rhinosinusitis and otitis media, skin and soft-tissue infections, bone and joint infections, cardiac device-associated infection, and sepsis [7].
4. SKIN AND SOFT-TISSUE INFECTIONS
A nick on an intact skin surface induced by either trauma, accident, surgical operation, or burn provides an open door for bacterial infections [8]. Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, Streptococcus pyogenes, Proteus sp., Streptococcus sp., Enterococcus sp., Citrobacter, E. faecalis, and coagulase-negative staphylococci are bacterial pathogens commonly associated with wound infections. These organisms display inherent resistance to numerous antibiotics and antiseptics, enabling them to survive for long periods and even proliferate in the presence of minimal nutrients to colonize traumatized skin. Clinical infection occurs in approximately 50% of wounds contaminated with bacteria [8]. Many of these bacteria appear on the WHO’s list of priority pathogens for research and development of new antibiotics, thus highlighting the importance of new antibacterial treatments for wound-associated infections. Using phage therapy to eradicate wound-associated infections shows interesting therapeutic potential for clinical applications [9].
Phage therapy's potential for treating skin infections has been studied, with results summarized in Table 1. In a study by Soothill, the role of phages in preventing infections during skin grafting was examined. Guinea pig models with wounds were infected with P. aeruginosa and/or phage (BS24) or control suspension before grafting. Phage BS24 effectively protected grafts against P. aeruginosa infection, showcasing its potential in preventing wound-associated infections [10].
Diabetic foot infection, a severe complication of diabetes mellitus patients, is typically managed using debridement and antibiotics to prevent and treat infections [11, 12] conducted a comparative in-vivo analysis using phage AB-SA01 (a cocktail of Myoviridae phages targeting S. aureus) and vancomycin in diabetic mice with wound infections. There are variations in skin wound healing between rodents and humans, as rodents incorporate an additional stage in their wound-healing mechanism known as skin contraction [12]. A splint wound model was employed to align rodent and human wound healing, neutralizing the differences. Results showed similar wound healing for both treatments, while AB-SA01 exhibited comparable or better bacterial load reduction than vancomycin. A clinical trial evaluating topical AB-SA01 administration demonstrated safety and tolerability with positive patient feedback [13].
| Phage Used | Target Bacteria | Potential Clinical Application | Models Used | References |
|---|---|---|---|---|
| BS24 | P. aeruginosa | Skin grafting procedure | Guinea pig | [10] |
| F44/10 and F125/10 | S. aureus | Wound treatment of patients suffering from diabetes mellitus | Wistar rats | [11] |
| F770/05 and F510/08 | P. aeruginosa | Wound treatment of patients suffering from diabetes mellitus | Yorkshire pigs | [11] |
| F1245/05 | A. baumannii | Wound treatment of patients suffering from diabetes mellitus | Yorkshire pigs | [11] |
| AB-SA01 (cocktail of 3 S. aureus Myoviridae phages) | S. aureus | Wound treatment of patients suffering from diabetes mellitus | BALB/c mice | [12] |
| Cocktail of S. aureus MR-5 and MR-10 | S. aureus | Wound treatment of patients suffering from diabetes mellitus | BALB/c mice | [24] |
| PP1131 (phage cocktail made of 12 anti P. aeruginosa phages) | P. aeruginosa | Burn wound infections | Humans | [18] |
| Cocktail of three customized bacteriophages | Escherichia coli, Staphylococcus aureus, and Pseudomonas aeruginosa | Chronic, non-healing wound | Humans (20 patients) | [25] |
Phages are administered to infected wounds through topical application, including gels, creams, and ointments [14]. Wound dressing is a beneficial option for topical treatment of wounds as it keeps adequate concentrations of the treatment at the site of infection, preventing wound exudate from washing away [15]. Table 1 provides a list of phages analyzed in vivo for possible clinical application in skin and wound treatments.
Several clinical trials of phage therapy in the treatment of skin infections have consistently demonstrated the safety and, to varying degrees, efficacy of phage therapy [16-18]. In all these studies, nearly 100% success was achieved in therapy with no side effects. Although, in some cases, the phage therapy was used in combination with antibiotics, the success of the treatment is still largely dependent on the phage used. Up-to-date systematic analysis of observational clinical data encompassing 2,241 cases indicated that 79% of patients undergoing phage therapy experienced clinical enhancement, with 87% eradicating bacterial infections [19]. This depicts that phage therapy was well tolerated. This is consistent with a systematic review by [20], which found that 86.1% of 310 individuals with chronic wounds achieved clinical resolution or improvement with phage therapy without evidence of adverse effects. Two out of seven modern trials have demonstrated evidence of efficacy [13, 21].
In the US, a 2006 report from the Southwest Regional Wound Care Centre disclosed using phages to treat 17 individuals with chronic wound infections, including cases linked to Pseudomonas aeruginosa. Positive outcomes were observed in all patients [19, 22]. In 2009, a safety trial involving phages for chronic venous ulcers was published, but due to exclusion criteria, no efficacy conclusions could be drawn [16]. The trial administered phages into wounds weekly for 12 weeks, establishing safety without adverse effects, as phages are common in the environment. A 2016 case series also highlighted nine cases of antibiotic-resistant diabetic foot infections caused by Staphylococcus aureus [23].
In a recent UK study, ten individuals with diabetic foot infections (DFIs) at high amputation risk despite antibiotics received topical anti-staphylococcal phage therapy. Expert clinical teams found that 9 out of 10 patients benefited from this additional phage treatment, with no reported adverse effects. In 6 cases, phage therapy contributed to infection resolution and limb preservation. One patient with unresolved osteomyelitis required amputation, while another saw Staphylococcus aureus eradication from a polymicrobial infection. Clinical improvement was noted in a ninth patient, but therapy ceased due to an unrelated event. However, a patient with weakly susceptible S. aureus did not respond significantly [19].
Encouragingly, the existing evidence indicates that phage therapy is secure and lacks significant adverse effects when utilized for treating skin infections.
5. RESPIRATORY TRACT INFECTIONS
Respiratory tract infections (RTIs) persist as ailments affecting the airways and lungs, emerging as a prominent global cause of suffering and mortality. The attention directed towards RTIs is a result of their significant impact on public health, reflecting their profound threat. RTIs rank among the most lethal and widespread types of infectious diseases, causing more than 3.8 million deaths globally in 2017. This accounted for 46% of the total infectious disease-related deaths during that year [26, 27].
There has been an upsurge of novel bacterial, viral, and fungal respiratory pathogens that are becoming increasingly challenging to treat, with respiratory tract infections (RTIs) being exacerbated by antibiotic resistance of Gram-positive and Gram-negative bacteria [28]. A respiratory infection caused by antibiotic-resistant bacteria can be life-threatening, and with the dearth of new alternatives and present attrition rate in new antibiotic molecule developments by pharma companies, an alternative approach, such as phage therapy is being explored. The use of phages in the treatment of various pulmonary infections like pneumonia, tuberculosis, cystic fibrosis, and pulmonary arterial hypertension, chronic obstructive pulmonary diseases caused by antibiotic resistant bacteria is being explored.
Various delivery methods are utilized for administering phages to treat pulmonary infections, including nebulization [29], inhalation therapy [30, 31], endotracheal tube administration [32], bronchoscopy [33], intravenous administration, direct instillation [32], and encapsulation [30, 14].
Nebulization involves converting phages into aerosols using devices like nebulizers and aerolizers, generating a mist that patients inhale for direct respiratory tract targeting. This technique employs vibrating mesh, compressed air (jet nebulization), or ultrasound to create aerosols from liquid suspensions. Notably, air-jet nebulization damages phage structure more than mesh nebulization, with the extent tied to tail length [29]. Nebulization approaches have demonstrated the ability to diminish infectivity, quantified by titer reduction, which aligns with morphological damage in the phages. This method is standard in respiratory medicine, delivering therapies to deep airways.
Inhalation therapy employs inhalers, akin to asthma medications, to directly deliver phage preparations into the lungs for infection combat. Intubated or ventilated patients can receive phages through the endotracheal tube, facilitating targeted access within the respiratory tract. Bronchoscopy introduces phages via a flexible tube, directly spraying infected areas. Direct instillation via a catheter delivers phage preparations into lung airways. Phages encapsulated within liposomes enhance stability and targeted lung delivery, leveraging lipid-based vesicles (liposomes).
It has been shown in most cases that phages used in the treatment of respiratory tract infections have demonstrated high efficacy either when used alone or in combination with antibiotics, as shown in Table 2.
Table 2.
| Phage Used | Target Bacteria | Potential Clinical Application | Models Used | Outcome | References |
|---|---|---|---|---|---|
| Cocktail of five phages comprising three Myoviridae pseudomonal phages and two Podoviridae pseudomonal phages designated PP1450, PP1777, PP1902, PP1792 and PP1797 | P. aeruginosa | Ventilator associated pneumonia (VAP) | Pig | 1.5-log reduction in Pseudomonas aeruginosa density | [31] |
| AB-PA01 AB-PA01-m1 |
Pseudomonas aeruginosa Burkholderia dolosa |
Lung transplant patients | Human | Phage therapy was well tolerated with no adverse events. Successful although treatment was combined with antibiotics | [35] |
| A first cocktail (APC 1.1) containing three lytic phages (JWDelta, JWT and 2-1) A second cocktail (APC 2.1) containing four lytic phages (JWDelta, JWT, 2-1 and JWalpha |
Pandrug-Resistant Achromobacter xylosoxidans | Lung-transplanted cystic fibrosis patient | Human | Despite initial persisting airway colonization, the final clinical and microbiological outcome was favorable. Low-grade airway colonization by A. xylosoxidans persisted for months before samples turned negative. No re-colonisation occurred more than two years after phage therapy was performed | [36] |
| Three-phage cocktail comprising (PaAH2ΦP (103), PaBAP5Φ2 (130), and PaΦ 134 | Pseudomonas aeruginosa (PsA UNC-D) | Pneumonia | Immunosuppressed BALB/cJ female mice | A one-time administration of phage via IMIT delivery was 100% efficacious in preventing mortality in an aggressive lethal murine model. Intraperitoneal phage administration alone did not forestall mortality but when combined with meropenem, therapy was more effective. | [32] |
| PEV20 | Pseudomonas | Lung infection | Mouse | Exhibited significant lethal potency against Pseudomonas in-vivo when used alone or when combined with ciprofloxacin | [37] |
Studies dating back to 1936 highlight phage administration against pulmonary pathogens like E. coli, Klebsiella, Streptococcus, Staphylococcus, and Pseudomonasvia inhalation. Some studies show 80-100% efficacy with inhaled phage therapy, yet occasional treatment failures arise due to gaps in phage specificity, quality control, and stability. Integrating modern phage therapy with inhalation and aerosolization techniques has advanced pulmonary phage treatment [33, 34, 30].
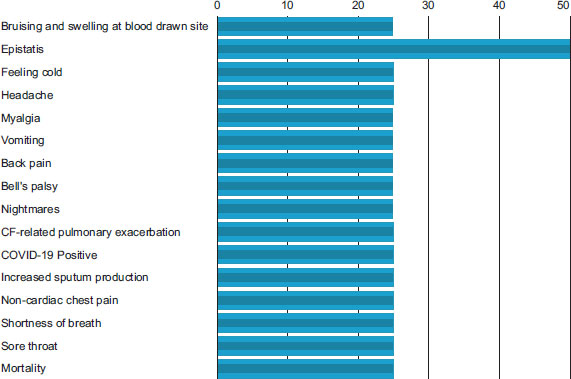
In the US, a clinical trial involving cystic fibrosis patients with chronic Pseudomonas aeruginosa airway infections utilized nebulized anti-Pseudomonas phage (YPT-01) to reduce sputum bacterial load in a cystic fibrosis bacteriophage study at Yale (CYPHY) https://clinicaltrials.gov/ct2/show/ NCT04684641. The results indicated successful treatment, with few experiencing mild adverse effects (Fig. 2). Recent progress, such as targeted delivery using electrospray, improved nebulization techniques, software-controlled individualized inhalation, and liposome-encapsulated nebulized phages, is poised to elevate pulmonary phage delivery, enhancing outcomes in antibiotic-resistant respiratory infections.
6. RECURRENT URINARY TRACT INFECTIONS (rUTIs)
Urinary tract infections (UTIs) are microbiologically defined as the urothelium's inflammatory response to microbial pathogens [38, 39]. Although there is no universally accepted definition for rUTIs, it is commonly defined as at least two infections with associated symptoms (acute bacterial cystitis, dysuria, pyuria) within the last six months or three infections in the previous 12 months [38].
Urinary tract infection is one of the most common bacterial infections, affecting an estimated 150 million people each year. It is a major public health concern in terms of morbidity and financial cost [39]. The cost of treating urinary tract infections in the United States alone is about 3.5 billion dollars a year [40].
Among all ages and genders, the urinary tract is the leading site of infections. Recurrent UTIs are prevalent among children and females. Worldwide, approximately 50% of women are anticipated to encounter a urinary tract infection (UTI) at least once during their lifetime, with UTIs being most observed among individuals aged between 16 and 64 years [41]. It is prevalent as a community and healthcare-related infection and affects both immunocompetent and immunocompromised hosts.
Urinary tract infections are caused by uropathogens, which are either community or hospital-acquired pathogens. Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, Staphylococcus saprophyticus, or Enterococcus faecalis induce community-based infections, whereas Escherichia coli, Pseudomonas aeruginosa, Proteus, Enterobacter, Serratia, or Enterococcus faecalis cause nosocomial infections [41]. Among the pathogens that cause urinary infections, E. coli is the dominant pathogen that causes nearly 80% of infections and infects 8–10 million people in the USA annually [42].
Antibiotics that were once effective now have minimal effect on bacteria that cause urinary tract infections, primarily due to the emergence and spread of bacteria-resistant strains, population growth, travel, and uncontrolled and excessive use of antibiotics [42]. Many uropathogens have developed low sensitivity to routinely used antibiotics like ciprofloxacin (frequently used), amoxicillin, cotrimoxazole, cephradin, nalidixic acid, ciprofloxacin, and azithromycin.
Bacteriophage therapy is one of the most outstanding alternatives to antibiotics being explored for the treatment of UTIs. In a study by [43], a successful clinical outcome was achieved when a cocktail of phages comprising HP3, HP3.1, ES17–1, and ES19–3 was used in treating a 56-year-old male liver transplant patient with complex, recurrent prostate and urinary tract infections caused by an extended-spectrum beta-lactamase (ESBL)-producing Escherichia coli (E. coli) (UCS1). The patient received two weeks of intravenous phage cocktail with concomitant ertapenem for six weeks.
A 72-year-old woman with chronic bacterial cystitis, suffering from recurrent UTIs caused by Klebsiella pneumoniae, sought treatment at the Eliava Phage Therapy Center (EPTC) in Tbilisi, Georgia, aiming to eliminate the pathogens [44]. The patient underwent a 20-day phage therapy course in June 2018, consisting of custom and Intesti phages. After 11 days, Klebsiella pneumoniae was no longer detected. However, on October 9, 2018, the last day of the treatment, the bacteria reappeared in her urine and vaginal swabs. A similar recurrence occurred in January 2019. In April 2019, the patient received another custom phage, advised to administer it twice daily for 20 days alongside a once-daily oral Intesti phage for a month. However, follow-up tests in July 2019 confirmed Klebsiella pneumoniae's presence in urine and vaginal samples, indicating unsuccessful eradication. Dissatisfied with the results, the patient did not return to EPTC in 2019 or subsequent years.
A 58-year-old renal transplant recipient developed recurrent urinary tract infections (UTIs) involving an extended-spectrum β-lactamase (ESBL)-positive Klebsiella pneumoniae strain within a month post-transplant. Seeking treatment, the patient turned to the Eliava Institute in Tbilisi, Georgia, for bacteriophage therapy. Following urine analysis, the patient received vials containing anti-Klebsiella pneumoniae bacteriophages. The prescribed regimen included twice-daily oral consumption of one vial content and bladder irrigation using a vial twice daily for two weeks. This was followed by two weeks of oral bacteriophage intake alongside once-daily bladder irrigation and, subsequently, one vial each orally and intravesically every second day for eight weeks. The patient administered all bladder installations through intermittent catheterization, ensuring maximum dwell time. No adverse effects were observed from oral or intravesical administration. After unsuccessful 10-day to 4-week meropenem therapy had been administered seven times, a combination of meropenem and bacteriophages successfully treated the persistently recurrent UTI caused by the ESBL-positive Klebsiella pneumoniae strain. Rapid alleviation of urethritis symptoms occurred upon bacteriophage initiation, indicating their contribution to treatment success [45].
7. GASTROINTESTINAL INFECTIONS
Gastrointestinal infections are among the most common infectious diseases found worldwide, varying depending on the etiological agent. Symptoms usually include diarrhea, vomiting, and abdominal pain. Water and electrolyte imbalances are the main consequences of gastrointestinal infections [46].
Every minute, there are over 50,000 cases of gastrointestinal illnesses; some individuals, especially children, will die from these infections. The estimated number of deaths worldwide from diarrheal diseases alone is over 2 million, and this estimate may not be entirely accurate as it does not include mortality from other manifestations of enteric infections like hepatitis, encephalitis, hemolytic uremic syndrome, meningitis, and others [47].
Emerging literature suggests that phages may play a key role in restoring the gut microbiome balance and controlling disease (gastrointestinal infections) progression either with exogenous phage intervention, filtered fecal transplantation, or engineered phages [48].
This is a syndrome characterized by nausea, vomiting, diarrhea and abdominal discomfort. Gastrointestinal infections are a significant cause of morbidity and mortality worldwide. Studies conducted in 2006 found that globally, severe diarrhea and dehydration are responsible each year for the death of 1,575,000 children under the age of five. This represents 15% of the 10.5 million deaths per year of children in this age group. According to recent estimates, acute gastroenteritis causes as many as 770,000 hospitalizations per year in the United States (Britton and Versalovic, 2008). Gastrointestinal infections can be caused by a large number of microorganisms present in the water, food, or hospital environment, that cause inflammation of different segments of the gastrointestinal tract.
According to clinicaltrials.gov, five ongoing clinical trials are focused on the gastrointestinal (GI) tract, with two trials already completed. One trial investigated a phage cocktail that targeted E. coli in the gut, assessing its impact on inflammation and the gut microbiome in healthy adults [49, 50]. Findings indicated that the phage did not influence overall microbiome diversity. Notably, there was a 40% average reduction in E. coli levels, accompanied by no changes in stool fatty acid production, lipid metabolism, or inflammatory markers. Additionally, a decrease in circulating interleukin-4 (IL4) was observed, possibly linked to allergic or autoimmune responses.
The second GI phage trial results were presented as a poster at the 2021 AASLD meeting. This trial focused on the safety, tolerability, and pharmacokinetics of an orally administered Klebsiella pneumoniae phage, given twice daily for three days to fourteen participants, compared to four placebos. The study revealed a sustained increase in phage presence from 0 to 103 PFU up to day 6. The treatment showed tolerability and safety, with a treatment-emergent adverse effect (TEAE) rate of 42.9% for treatment and 50% for placebo, and no adverse effects related to treatment were identified [51].
Furthermore, three additional GI clinical trials are currently ongoing. These trials are evaluating the safety and efficacy of a phage cocktail (ShigActive) for treating shigellosis, exploring fecal bacteriophage transfer to support GI maturation in preterm infants, and investigating the safety and efficacy of an adherent invasive E. coli-specific phage cocktail in patients with inactive Crohn’s Disease (University of Baltimore - https://clinicaltrials.gov/ct2/show/NCT05182749; https://www.clinicaltrials.gov/ct2/show/NCT03808103 [52].
Phage therapy promises to be a valuable tool in the treatment of gastrointestinal infections. However, a lot of work still needs to be done to understand the dynamics between phages, bacterial hosts, bacteriomes, and the mammalian host.
8. IMPLEMENTATION HURDLES
Numerous phages show promise in inhibiting antibiotic-resistant bacteria, yet their limited usage persists due to specific challenges. The deployment of phage therapy has been chiefly based on compassionate use. In August 2021, the FDA made available transcripts from a meeting discussing “Science and Regulation of Bacteriophage Therapy,” during which prominent scientists and medical professionals extensively debated the practicalities of applying phage therapy in clinical settings [53]. The U.S. Food and Drug Administration (FDA) has issued emergency use authorization (EUA) for bacteriophage therapy in response to the significant impact of antibiotic-resistant bacterial infections. This authorization permits compassionate use of this therapy on a case-by-case basis. Compassionate use, also known as expanded access, involves offering investigational therapies to patients with no other available treatment options (as determined by a physician), even when these therapies have not received full regulatory approval. While these compassionate care instances transpire within controlled clinical environments, they are distinct from clinical trials and are assessed through individual patient case series rather than large cohorts (selected populations).
Several challenges are associated with the utilization of phage therapy. These include the development of phage resistance, the potential transfer of virulence or antimicrobial resistance genes via horizontal transfer, and the absence of randomized controlled trials to effectively translate in vitro findings to clinical settings, necessitating cautious application. Other obstacles include the limited host range of phages, the time-consuming process of characterizing and selecting the most suitable phages for therapy, the inability of phages to infect intracellular pathogens, and the recognition of phages as foreign entities by the immune system, leading to subsequent destruction [51].
Additionally, phages must endure various stress conditions to remain effective, and regulatory agencies lack standardized and comprehensive guidelines regarding their application and administration. Moreover, the absence of the inclusion of phage therapy in public health insurance in many countries contributes to increased healthcare costs [54]. To promote the widespread adoption of phage therapy in clinical practice, future research should address these challenges and find solutions.
Phages multiply on their own, complicating dosage determination. Actual site concentration may differ from administered amounts, especially when combined with antibiotics that boost multiplication via increased cell lysis [55]. Further studies are crucial to understand and standardize dosing practically.
Data on public awareness and patient perceptions of phage therapy are scarce due to its emerging status in clinical infection management. Further research is crucial in understanding patient perspectives and worries to reintroduce and progress phage therapy effectively. Patient-centered insights will guide the development and application of these therapies. Additionally, assessing the thoughts and concerns of participants in phage-related clinical trials is essential for a comprehensive understanding.
A well-regulated, regularly updated resource is essential for efficient global transportation of phages. This resource should encompass isolated phages stored in various banks worldwide and their host ranges. Presently, phage updates are irregularly scattered across diverse scientific journals, needing a standardized platform for comprehensive phage information [56, 57]. Although databases like the Phage Directory exist, voluntary contributions are an incomplete representation of global phage labs and scientists. The future of phage therapy relies on a structured, standardized, and institution-independent database for accurate information dissemination.
9. GLOBAL EFFORTS IN PHAGE THERAPY INITIATIVES AND ADOPTION
The use of phage therapy in compassionate cases aligns with the Declaration of Helsinki, Article 37, addressing unproven interventions in clinical practice. Each country's regulatory authority, like the Food and Drug Administration (FDA) in the US, the Therapeutic Goods Administration (TGA) in Australia, the European Medicines Agency (EMA) in the EU, and the Brazilian National Health Surveillance Agency (Anvisa) in Brazil, governs the process and criteria for phage therapy. Unlike formal clinical trials, compassionate use focuses on patient care, diagnosis, and treatment rather than gathering data for investigational product approval [51].
The pursuit of unapproved treatments via compassionate use has steadily gained traction over time. Different countries have rules guiding the adoption of phage therapy based on compassionate use. In some countries, like the United States, legislation such as the “Right-to-try” law supports compassionate use care for critically ill individuals [58]. Conversely, in Poland, phage therapy is categorized as an “Experimental Treatment,” governed by a law adopted on December 5, 1996, and available to medical professionals [59]. The Hirszfeld Institute of Immunology and Experimental Therapy (HIIET) has led the way in the compassionate use of therapeutic phages, significantly advancing phage therapy research [60]. Likewise, in response to the absence of dedicated regulations regarding phage utilization, the Belgian Federal Agency for Medicines and Health Products adapted its licensing procedures to accommodate phages as active components in pharmaceutical compounding. This permits individual phage prescriptions following internal guidelines [23, 61]. Furthermore, phage therapy is not universally standardized but is personalized based on patient requirements, complicating the establishment of uniform regulations [62].
Efforts like the Africa Phage Forum show promise in regional collaboration. However, more work is needed to foster collaboration across different global regions, particularly involving Low- and Middle-Income Countries (LMICs) [63]. Bacteriophage research is heavily concentrated in the U.S. and China, along with productive phage therapy research in countries, such as Canada, India, Poland, Spain, Australia, and South Korea. Current international collaboration patterns often exclude LMICs despite their potential to enhance global health equity through phage therapy.
Countries like Brazil hold untapped phage resources, yet investment in phage research is limited. Nevertheless, Brazil is working to establish a phage bank for therapeutics and international research collaboration. As a prominent South American nation with close geographic proximity and trade ties to neighboring LMICs, Brazil can establish itself as a crucial center for phage research.
Physical phage conferences and personnel/equipment exchanges enhance global phage research. Teleconference frameworks like Project ECHO enable experts to share knowledge even in remote areas, facilitating information exchange across institutions regardless of location. This collaborative approach is vital, as it empowers LMICs to establish their phage banks, reducing reliance on phages from industrialized countries [64].
CONCLUSION
Bacteriophages offer a compelling and essential therapeutic potential, particularly considering the escalating worldwide antimicrobial resistance crisis. Up to this point, the safety record and patient tolerance of phage therapy have shown promising results. Although phage therapy is a promising treatment option, it requires time, funds, and dedication to pursue. Even more importantly, it must be administered correctly. Like any other medical technology, it is unlikely to be end-all and be-all treatment.
LIST OF ABBREVIATIONS
| ARLG | = Antibacterial Resistance Leadership Group |
| NIAID | = National Institute of Allergy and Infectious Diseases |
| UTIs | = Urinary Tract Infections |
| RTIs | = Respiratory Tract Infections |
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The author declares no conflict of interest financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


