All published articles of this journal are available on ScienceDirect.
Prevalence of High-Risk Antibiotic Resistant Acinetobacter baumannii in the Holy Cities of Makkah and Al-Madinah
Abstract
Background:
Acinetobacter baumannii strains resistant to carbapenems are a global public health problem.
Objectives:
The aim of the present study is to evaluate the prevalence of genetic fingerprints associated with Metallo β-lactamases in A. baumannii in addition to the clonal diversity of A. baumannii in Makkah and Al-Madinah regions of Saudi Arabia, which receive a high number of international visitors.
Methods:
Multi-antibiotic resistant A. baumannii isolates were investigated. Bacterial isolation was conducted employing a basic bacteriological technique after confirming the ID of isolates. The antimicrobial susceptibility test was carried out using the Vitek 2 compact system. The molecular clonal diversity of the isolates was determined by Pulse Field Gel Electrophoresis (PFGE). Clusters were analyzed with BioNumerics software version 6.5. Dice coefficient was used for calculating the similarities.
Results:
The results indicated resistance in 82.5% of A. baumannii isolates against the carbapenems. All the isolates were found to be sensitive to colistin, while 5% of isolates were resistant to tigecycline. The screening of carbapenem-resistant A. baumannii isolates showed that the dissemination of imipenem and meropenem resistance was 81 and 84%, respectively, while the majority of the strains were susceptible to tigecycline and colistin. The blaOXA and blaVIM were the most encountered genes in A. baumannii isolates, while ISAba1 was the prominent insertion sequence. The genetic fingerprinting results (PFGE) revealed two types of epidemic clones: monoclonal and polyclonal models of 17 clusters.
Conclusion:
The current investigation indicates the diversity in genetic fingerprints of carbapenem-resistant A. baumannii in Makkah and Al-Madinah region of Saudi Arabia, and that two types of epidemic clones are present. It has also been demonstrated that such clones create serious infection dissemination to other parts of the world as heavy pilgrimage traffic is received throughout the year in Makkah and Al-Madinah, especially in the Haj season.
1. INTRODUCTION
Infections resulting from Acinetobacter baumannii continue to receive global attention by researchers, more so as such infections have become extremely difficult to treat. These infectios have led to an increase in the literature available globally, which aims to conduct the molecular analysis of the bacterium in order to better understand the antimicrobial resistance exhibited by A. baumannii [1]. Generally, settings associated with infections of Acinetobacter include the following: ICUs acquired infections by patients, those from other healthcare settings, community-acquired as well as those resulting in trauma patients [2]. Moreover, infections are commonly associated with immunocompromised as well as immunosuppressed patients [3].
The 21st century witnesses the threat of a possible pre-antibiotic era due to the evolution of multi-antibiotic resistant bacteria. In terms of susceptibility to existing antibiotics, the resistance of A. baumannii has progressively increased since the 1970s, differing among the geographical regions of countries, hospitals, medical departments, and clinical samples [3]. Multi-antibiotic resistance includes resistance to the carbapenems, as reported globally by researchers [4-6]. The mechanism of resistance by A. baumannii to antimicrobials has been receiving attention from researchers leading tomuch available literature on the genomics and evolutionary aspects of carbapenem-resistant A. baumannii [6-8]. Understanding the differences in clonal strains can help understand variability in pathogenicity and mortality, with the possibility of a subsequent curb on the spread of resistance genes [9]. The production of Metallo-β-lactamases (MBLs) is one of the primary causes of resistance to β-lactam antibiotics [10]. A wide range of β-lactamases that contribute to carbapenem resistance in Saudi Arabia have received continuous attention along with IMP-like, SIM-like and VIM-like carbapenemases reported in A. baumannii [11]. The available literature attributes antimicrobial resistance exhibited by this bacterium to a diverse number of genetic determinants, of which OXA-23, OXA-40 and OXA-51 β-lactamases are the most commonly reported genes [1]. The dominance of blaOXA23 and blaOXA-40 has been reported for carbapenem-resistant A. baumannii from the Gulf Cooperation Council States (GCC) [12]. The MBLs genes are located on integrons, and thus, can be transmitted from one bacterial species to another [2, 13]. In fact, the rise in multi-antibiotic resistant phenotypes is due to the acquisition of mobile genetic elements (MGEs), such as integrons (Int) and genomic islands (GEIs) [14]. The evolution of high levels of multi-antibiotic resistance in A. baumannii over the years is attributed to a number of factors, such as the intrinsic carriage of resistance genes by the bacterium as well as being able to acquire resistance genes through genetic elements from other bacteria [15-17]. There are different types of integrons, with class 1 reported to be the most commonly associated with the spread and emergence of multi-antibiotic resistance [18]. The class 1 integron and its associated cassettes are the main contributors to the multi-antibiotic resistance exhibited by A. baumannii isolates globally [19]. However, molecular analysis of CRAB isolates from the region focused in the present study with origins from different states shows different clones to be clustered together, implying the transference of genes responsible for multi-antibiotic resistance [12].
Colistin remains the last line antibiotic available for treating multi-antibiotic resistant gram-negative bacterial infections. There are, however, periodic global reports of resistance to colistin, thus pointing at strains that are resistant to all available antibiotics [7]. Plasmid-mediated colistin resistance gene has been isolated from E. coli in GCC countries between 2012 and 2016 [20, 21]. Thus, there is the possibility that colistin might soon fail to be the last line of drug choice. There is a high rate of research investigations and reporting on multi-antibiotic resistant A. baumannii originating from hospitals in Saudi Arabia, highlighting the public health problem faced globally and regionally by multi-antibiotic resistant gram-negative bacteria inclusive of A. baumannii [1].
2. MATERIALS AND METHODS
2.1. Bacteria Isolates and Antimicrobial Susceptibility
Thirty-seven Acinetobacter baumannii isolates collected from patients in the western region of Saudi Arabia (Makkah and Al-Madinah) were used for the investigation. All bacterial samples in this investigation were identified phenotypically as well as genotypically. Identification of strains as Acinetobacter baumannii complex as well as minimum inhibitory concentration (MIC) of imipenem, meropenem, tigecycline, and colistin were determined by using the VITEK 2 compact system (BioMerieux, Marcy L Etoile, France) with the GN ID and Antibiotic Susceptibility Testing (AST) cards, according to the guidelines of the manufacturer.
The presence of blaOXA−51−like, blaOXA−23−like and blaOXA−40−like genes was detected by PCR reactions using specific primers. Production of a blaOXA−51−like amplicon confirmed the species identification. ATCC 19606 A. baumannii was used as a positive control, while molecular grade water was used as a negative control in PCR assays to detect contamination.
The presence of acquired metallo-β-lactamase genes (MBLs) of the blaIMP, blaVIM, blaSIM, blaSPM and blaGIM families and the insertion sequences ISAba1, ISAba2, ISAba3, and IS18 in the isolates were screened by PCR using consensus primers.
The molecular typing of the isolates was carried out by PFGE using the CHEF-RDII apparatus (Bio-Rad). The methodology is as earlier described by AlSultan et al. [22], Evans et al. [23], and Miranda et al. [24], according to the guidelines of the manufacturers. All the A. baumannii clinical isolates were typed by PFGE analysis in the following way. DNA obtained from bacteria was digested using the ApaI restriction endonuclease (Promega), and DNA fragments were separated on 1 % agarose gel in 0.5× TBE buffer using the CHEF-RDII apparatus (Bio-Rad). Pulse time of 3–35 s at a field strength of 6 Vcm−1 at 14 °C for 24 h was used. The gel was stained by ethidium bromide, and the digital images were captured by Gel doc2000 (Bio-Rad). All isolates were analysed using Bionumerics software version 6.5. Isolates that clustered together with a similarity of greater than 85 % were considered as belonging to the same PFGE type.
3. RESULTS
3.1. Isolates and Antimicrobial Susceptibility
Of the total 37 A. baumannii isolates, 25 (68%) were from the Al-Madinah district, while 12 (32%) of the isolates were from the Makkah district of Saudi Arabia.
| District | Antimicrobial agents [n, %] | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Imipenem | Meropenem | Tigecycline | Colistin | |||||||||
| S | I | R | S | I | R | S | I | R | S | I | R | |
| Al-Madinah (n=25) |
2 (8) | 2 (8) | 21 (84) | 0 (0) | 3 (12) | 22 (88) | 23 (92) | 0 (0) | 2 (8) | 25 (100) | 0 (0) | 0 (0) |
| Makkah (n=12) |
3 (25) | 0 (0) | 9 (75) | 2 (17) | 1 (8) | 9 (75) | 12 (100) | 0 (0) | 0 (0) | 12 (100) | 0 (0) | 0 (0) |
| Total (%) (n=37) |
5 (14) | 2 (5) | 30 (81) | 2 (5) | 4 (11) | 31 (84) | 35 (95) | 0 (0) | 2 (5) | 37 (100) | 0 (0) | 0 (0) |
The results presented in Table 1 show the antimicrobial susceptibility of all groups of isolates against tested antimicrobials.
84% and 88% resistance to imipenem and meropenem, respectively, was observed to be exhibited by A. baumannii isolated from patients in Al- Madinah. For A. baumannii isolated from patients in Makkah, there was a 75% resistance observed to both imipenem as well as meropenem. A 100% sensitivity to colistin and tigecycline was seen to be exhibited by the A. baumannii isolates from Makkah. However, there was an 8% resistance observed to tigecycline of the isolates from Al-Madinah. There was, however, a 100% sensitivity observed of all isolates to colistin (Table 1).
3.2. Molecular Genotyping (PFGE)
In this study, the fingerprints of genomes of A. baumannii strains were compared following digestion with the restriction enzyme Apa1 and the restriction analysis of the generated fragments has been conducted as reported by Seifert et al. [25]. PFGE typing was used to analyze 37 clinical isolates of A. baumannii obtained from various deep-site infections of different patients located in different major hospitals in Western Saudi Arabia.
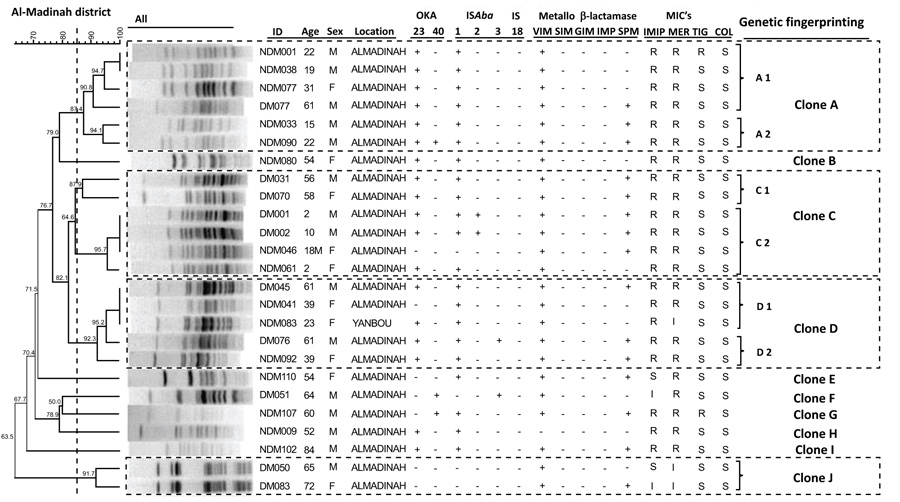
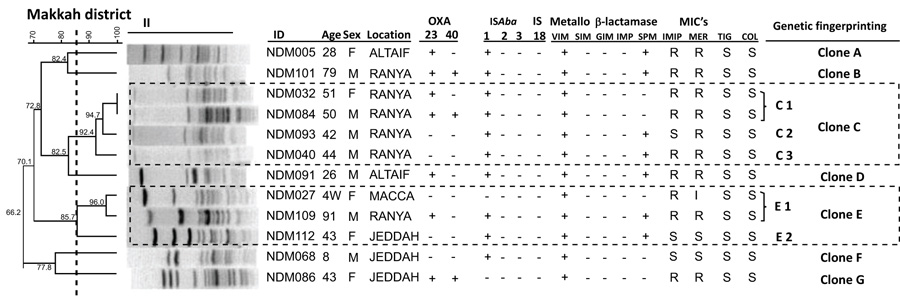
The clustering results by PFGE for Acinetobacter baumannii isolates from Al-Madinah and Makkah districts are presented in Figs. (1 and 2), respectively. Each dendrogram for the districts details the sample ID code, the patient's age and gender. Also included are the presence or absence of transferable OXA-23, OXA-40, and the insertion sequences ISAba1, ISAba2, ISAba3, and IS18. The results of five Metallo β-lactamases (VIM, SIM, GIM, IMP, and SPM) for each A. baumannii isolate from both of the districts are also included in the respective dendrograms (Figs. 1 and 2).
The discrimination power of the PFGE technique was expressed by the dice coefficient with (BioNumeric) software system version 6.5. All detected PFGE patterns showed a genetic fingerprinting similarity coefficient ranging from 63.5% to 100%; however, all samples with a similarity of 85% and more were considered to compromise the same genetic fingerprint. In the Al-Madinah district (Fig. 1), the PFGE clones clustered into 10 groups from A to J. The genetic fingerprints grouped into A, C and D consisted of 17 different strains. The fingerprinting groups A and C comprised 6 strains each, while the fingerprinting group D possessed 5 strains. Each group consisted of 2 subgroups (A1, A2), (C1, C2) and (D1, D2), respectively. The other 7 groups B, E, F, G, H, I and J consisted of a single clone each, as shown in Fig. (1).
In Makkah district (Fig. 2), the PFGE clones were clustered into 7 groups from A to G. The genetic fingerprinting groups C and E compromised 7 strains, group C comprised 4 strains in 3 sub-groups C1, C2 and C3, whereas group E possessed 3 strains in 2 sub-clusters, E1 and E2. The remaining five groups A, B, D, F and G comprised single clone each, as shown in Fig. (2).
3.3. Statistical Analysis of the Gene Expression
Among the OXA carbapenemases, the OXA-23 was more prevalent (56.7%) among the isolates than OXA-40 (5.4%) from this western region of Saudi Arabia. There was an overall 11% coexistence of OXA-23 and OXA-40 among the A. baumannii isolates from both the districts of Makkah and Al-Madinah, and the difference was not statistically significant (p<0.05) (Table 2).
Of the three classes of insertion sequences (ISAba 1, 2, and 3), the most frequently encountered was ISAba1, with an overall percentage of 84%. The carriage of ISAba1 was high at 83% and 84% for isolates from Makkah and Al-Madinah, respectively. Insertion sequences ISAba2 and ISAba3 were less with a 5.4% of each detected in the isolates from Al-Madinah only. The sequence IS18 was absent in all the A. baumannii isolates from this Western region of Saudi Arabia. The coexistence of ISAba1 and ISAba2 was seen in 8% of A. baumannii isolates from Al-Madinah alone, while there was no coexistence of ISAba2 and ISAba3 (Table 2). The absence of ISAba1, ISAba2, and ISAba3 was found in 2 (17%) of the isolates from Makkah and 3 (12%) of the isolates from Al-Madinah. Also, of the five investigated MBL (VIM, SIM, GIM, IMP, and SPM) genes, only VIM and SPM were detected, with VIM being predominant at 97% than the SPM carriage of 57% in the isolates, as shown in Table 2. Twenty-one (57%) of the total A. baumannii isolates were found to involve both VIM and SPM Metallo β-lactamase genes (Table 2).
| Genotype Expression | Number of Isolates, n (%) | ||
|---|---|---|---|
| - | Makkah (n=12) | Al-Madinah (n= 25) | Total Number of Isolates (n= 37) |
| OXA-23 | 4 (33.3%) | 17 (68%) | 21 (56.7%) |
| OXA-40 | 0 (0) | 2 (8%) | 2 (5.4%) |
| Isolates with both OXA-23 and OXA-40 |
3 (25%) | 1 (4%) | 4 (11%) (0.8501) † |
| Absence of both OXA-23 and OXA-40 |
5 (41.7%) | 5 (20) | 10 (27) (0.8240) † |
| ISAba 1 | 10 (83) | 21(84) | 31 (84) (0.6307) † |
| ISAba 2 | 0 (0) | 2 (8) | 2 (5.4) |
| ISAba 3 | 0 (0) | 2 (8) | 2 (5.4) |
| Co-existence of ISAba 1 and ISAba 2 |
0 (0) | 2 (8) | 2 (5.4) |
| Co-existence of ISAba 2 and ISAba 3 |
0 (0) | 0 (0) | 0 (0) |
| Absence of ISAba 1, ISAba 2 and ISAba 3 |
2 (17) | 3 (12) | 5 (13.5) |
| VIM | 12 (100) | 24 (96) | 36 (97) |
| SPM | 7 (58) | 14 (56) | 21 (57) |
| Co-existence of VIM and SPM | 7 (58) | 14 (56) | 21 (57) |
4. DISCUSSION
Antimicrobial resistance to A. baumannii has led to significant challenges associated with treating infected patients, especially those who are immunocompromised. Excluding effective therapy, like carbapenem antibiotics, leaves the options of treatment very limited. In the current study, A. baumannii isolates obtained from the Western District of Saudi Arabia were examined to elaborate the prevalence of genetic fingerprinting associated with metallo β-lactamases and the clonal diversity in such busy districts with international visitors. The significant difference in resistance to both imipenem and meropenem by isolates indicates that the isolates are of different strains, and also that A. baumannii isolates in circulation carry different genes having multi-antibiotics resistance to the carbapenems. Comparable to these results are those of earlier studies that reported a clonal diversity in Acinetobacter baumannii isolates in hospitalized ICU patients in other regions of Saudi Arabia [22]. They reported a 92% and 96% resistance by the ICU isolates against imipenem and meropenem, respectively. The 82.5% carbapenem resistance seen in patients in this study, although less than that of their earlier report [22], points out that there are clones of A. baumannii in the Western2 district of Saudi Arabia that are different in terms of the carriage of resistance gene strains. On the other hand, the 2014 study for ICU isolates could explain the 92% and 96% imipenem and meropenem resistance, while the isolates in this study were not from ICU in origin. Therefore, the differences could be a result of the different regions of the isolate’s origin and associated patient risk factors, such as whether the patient had an extended stay in the hospital or otherwise. The 86% CRAB for the Al-Madinah district encountered in the present report is contrary to those studies that reported a 100% resistance each to meropenem and imipenem for isolates from hospitalized patients [25, 26]. This, while pointing again to clonal differences in the Western district of Saudi Arabia, also calls for intense and close monitoring of CRAB isolates in the region. In the present report, for the Makkah district, imipenem as well as meropenem resistance was found to be 75%. The result shows a rise in resistance to these antibiotics as compared with reports from other researchers. Previously, the percentage resistance to imipenem for Makkah district for the years 2004 to 2005 was found to be at 14% [27]. This percentage in resistance rose to 45.9% between 2005 and 2006 [28]. For meropenem, from a reported 28% resistance covering the years 2005 and 2006, there was an increase in resistance to 64% by A. baumannii by the year 2015 for Makkah district [29]. Reports while differing do point that high CRAB isolates in circulation in these western districts of Saudi Arabia carry different clones.
The most prominent insertion sequence in the A. baumannii isolates from this Western region of Saudi Arabia was found to be ISAba1 (84%). This insertion sequence has also been found to be the most prevalent in other studies [30] though differing in percentages as previously reported. Al Sultan et al. [22] and Villalo´n et al. [30] recorded a prevalence of 90.6% (Saudi Arabia) and 93.2% (Spain), respectively. Regardless of differences in percentages, these insertion elements contribute to the ability of A. baumannii to be resistant to the carbapenems [31]. In the present study, the genetic fingerprinting similarity in PFGE was found to be very high, at 87.4 -100%. The genetic fingerprinting diversity revealed two models of epidemic clones: monoclonal and polyclonal. The monoclonal type was the most common fingerprinting gene and affected 11 patients in both districts. This type of clonal outbreak was caused by one or more epidemic clones. On the other hand, the polyclonal strains affected 26 patients in both districts. Patients harbored six polyclones out of 17 different clones, four of which (A, C, D and J) were detected in the Al-Madinah district, while the remaining polyclonal models (C and E) were detected in the Makkah district. Detection of such pathogenic clones could mean an explosive outbreak at an unexpected time. Makkah and Al-Madinah districts are very busy and always crowded with local and international visitors that can lead to a very quick clonal transmission not only from one patient to another, but also from one district to another, as well as to a distant country. The admixture and out-of-control transmission of A. baumannii have been well documented [32-34]. The reports mentioned above and those of the present findings show that there has to be a continuous and close monitoring, generally of Gram-negative bacteria and their resistance to available antimicrobials in Saudi Arabia, in order to meet with global and regional requirements of continuous surveillance aimed at control.
One limitation of this study is that low number of isolates (37 isolates) have been reported. Although a large number of samples were screened, duplicates were frequently encountered. All duplicates were excluded from the study.
CONCLUSION
The problem of the evolving difficult-to-treat A. baumannii infection carrying multi-antibiotic resistance genes is highlighted in the present study. The significant difference in resistance to both imipenem and meropenem by isolates indicates that the isolates are of different strains, and also A. baumannii isolates in circulation carry different genes having multi-antibiotic resistance to the carbapenems. The current investigation indicates the diversity of genetic fingerprinting of carbapenem-resistant A. baumannii in Western Saudi Arabia, in addition to the capability of such clones to create serious infection dissemination to many cities in Saudi Arabia and the rest of the world. The continuous surveillance of a wide range of antimicrobial-resistant genes is especially required for cities of Makkah and Al-Madinah, considering the heavy pilgrimage traffic that is received throughout the year in these regions, especially in the Haj season.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The data that supports the findings of this study is available with in the article.
FUNDING
This research was funded by King Abdulaziz City for Science and Technology (KACST), grant number BIO-1093-06-10.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The author would like to thank King Faisal University for its support. The author also acknowledgesthe following: Dr. Lorina Badger-Emeka for the critical review of the article write up, Dr. P. Emeka for his help with statistical analysis, and Dr. Ghazala Muteeb and Dr. Yousif Elmosaad for the great help and support. The author also shows gratitude to Mr. Hani Alrasasi, Mr. Hani Alfarhan, Mrs. Hajar Al-Dohilan, Mrs. Nouf Al-Homaini, Abdulqader Abouli, and Fatemah Al Najar for their technical assistance.


