All published articles of this journal are available on ScienceDirect.
Molecular-based Survey of Rickettsia spp. and Coxiella burnetii in Mosquitoes (Diptera: Culicidae) from Fars Province, Southern Iran, during 2017-18
Abstract
Objectives:
Since there have not been any studies on the roles of the Iranian mosquitoes in the transmission of Rickettsia spp. and Coxiella burneti, the present study investigates the roles of mosquitoes in the transmission of the pathogens using the PCR techniques for the first time in Iran.
Methods:
The present study was conducted in Fars province during the activity seasons of mosquitoes in 2017-18. The primer design was done to investigate the probability of mosquito’s contamination with Rickettsia spp. and Coxiella burnetii. The conventional PCR was used after the extraction of DNA from mosquitoes to study the contamination.
Results:
A total of 1103 adult mosquitoes were collected and identified. Among them, 3 genera and 11 species were identified, including Anopheles (25.74%), Culex (51.84%) and Culiseta (22.39%) genera. All tested mosquitoes were negative in terms of contamination to Rickettsia spp. and Coxiella burnetii.
Conclusion:
Based on the results, mosquitoes are not considered as vectors of Rickettsia spp. and Coxiella burnetii in this part of the country currently. Further studies on a larger scale are needed to examine the exact role of mosquitoes (as a possible vector with high abundance and mobility) in the transmission of these pathogens in tropical areas of Iran.
1. BACKGROUND
Mosquito-borne diseases are the main public health problems worldwide, especially in tropical and subtropical regions [1-5]. Despite the fact that their bites are widely annoying, they are probably the most dangerous vectors of disease or cause of mortality in humans [1]. Their infection issues are related to their ability to transmit various types of pathogens, such as filarial nematodes, Plasmodiums and Arboviruses. Among them, malaria, dirofilariasis, dengue fever, west nile virus (WNV), yellow fever and some other viral diseases are the most pathogenic [1, 6]. Due to climatic conditions in tropical and subtropical regions, the invasion of mosquitoes, which bite the human, may be high in urban and rural places leading to the increase of mosquito-borne diseases [7]. Among international travelers, malaria, dengue, and Rickettsial diseases are the most common causes of febrile illnesses; and the exposure to mosquitoes is reported as the most common source of fever [2, 8]. Most vector mosquitoes, which can cause human diseases, are divided into three genera, namely Anopheles, Aedes and Culex. These genera can transmit human filariasis, arboviruses and dirofilariasis; however, only Anopheles mosquitoes can transmit five known Plasmodium parasites that cause human malaria [7]. Despite a great number of researches on mosquito-borne infections, there is no study on the probable transmission of Rickettsia spp. and Coxiella burnetii by mosquitoes to humans in Iran, and there are a few studies worldwide.
These are bacteria of the Rickettsiales order and are referred to as short, gram-negative and rod-shaped bacteria and need eukaryotic cells for growth and proliferation [9]. In terms of history, Rickettsiales order is divided into three families, namely Rickettsiaceae, Bartonellaceae and Anaplasmataceae. Rickettsiaceae Family consists of three tribes, namely Rickettsieae, Ehrlichieae and Wolbachieae; and rickettsieae consists of three genera, namely Coxiella, Rickettsia, and Rochalimaea. Based on morphological, antigenic and metabolic characteristics, Rickettsia genus is divided into three groups: the typhus group (TG) consisting of R. typhi, R. prowazekii, and R. Canada members [10-15].
The spotted fever group (SFG) contains 20 different species of Rickettsia spp. (R. felis) [11, 13, 14, 16]. The scrub typhus group includes Orientia tsutsugamushi (Rickettsia tsutsugamushi until 1995) [12-15, 17]. Rickettsia spp. are transmitted through infected arthropods such as ticks, mites, fleas and lice.
Coxiella burnetii is also a small gram-negative bacterium that cannot grow in the medium [18, 19]. This bacterium is the cause of Q fever disease that is common worldwide. Rickettsial diseases of Iran include endemic typhus (murine typhus) [20-24], epidemic typhus (louse-borne typhus) [21, 24, 25] and Q fever [26-29].
In 1924, the presence of intracellular microorganisms of Rickettsia was reported in ovarian and testicle cells in Culex pipiens [30]. Later, this bacterium was introduced as Wolbachia pipientis, a member of Anaplasmataceae family in Rickettsiales order. In addition, Yen et al. introduced another type of transovarially transmitted Rickettsia through the electron microscope in 1975 [31]. Therefore, we assumed that there are probably other unknown species of Rickettsia in mosquitoes. In sub-Saharan Africa, R. felis is the cause of 15% of the unknown fever [32]. It has been recently found that Anopheles gambiae, the main vector of malaria in sub-Saharan Africa, acts as a vector of R. felis [2, 30, 31]. Interestingly, both cells of Anopheles gambiae and Aedes albopictus help to develop R. felis [7, 32-34]. The research indicates that mosquitoes can play roles in transmitting Rickettsia, especially R. felis. Since there have not been any studies on the roles of the Iranian mosquitoes in transmitting Rickettsia and Coxiella burnetii, the present study investigates the roles of mosquitoes in the transmission of these pathogens using the PCR (polymerase chain reaction) technique in an endemic area in south of Iran (near Persian Gulf with subtropical weather) for the first time.
2. METHODS
2.1. Study Area
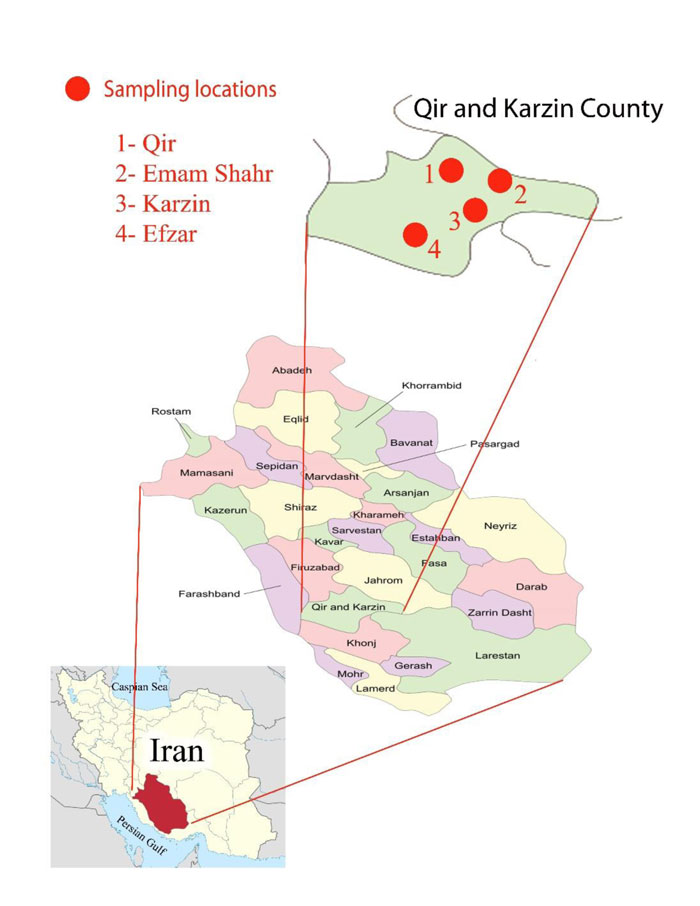
Fars Province: Fars is one of 31 provinces of Iran and is located in the south of Iran (29.62°N, 52.53°E). According to topographic characters, there are three distinct climatic regions in this province. Qir and Karzin County is located in the southwest of Fars province. It is between N 28° 32' to 28° 54' latitude and E 52° 6' to 53° 13' longitude of the Greenwich meridian (N 28.48415°, E 52.99710°) with an area of about 98.3402 km2 based on the coupon classification in the semi-arid areas of the earth with rainy winter and hot dry summer. The maximum temperature is 46 °C in the summer and the minimum temperature is 25.1 °C in the winter. The mean precipitation of this city is 270 mm per year. Qir and Karzin County has borders with Firuzabad, Jahrom, Khonj and Farashband counties. Sampling was carried out in four regions (including Qir, Efzar, Karzin and Emam Shahr) in the mosquito activity seasons every 2 months (Fig. 1).
2.2. Mosquitoes Collecting and Identifying
Mosquitoes were collected by hand-catch, total catch from indoor and outdoor places (human and animal) [7, 35, 36]. All needed data like date, location and hour of collecting were recorded along with mosquito species. Adult specimens of mosquitoes were pinned and kept at the Museum of Medical Entomology (Shiraz University of Medical Sciences, Department of Medical Entomology). In this study, another innovative and effective method was used for catching mosquitoes. In this method, wasted tires were placed in the target places and filled with water to attract mosquitoes for resting in these moist environments. Two to three days after using tires, they were covered with nets with small pores. Then they were shaken so that mosquitoes, which were resting, flied upwards and got caught by the net. The aspirator was then inserted slowly from the corner of net, and mosquitoes were collected and transported into the cups. The caught mosquitoes were transferred alive to the Medical Entomology Laboratory of Shiraz University of Medical Sciences for further studies. After pinning, the mosquitoes were identified using a valid identification key [37].
Adult mosquitoes were collected by simple random sampling. Since there was no research on this field, and the prevalence was not known in Iran, the sample size was calculated according to the statistical consultant's opinion based on similar studies using the sample size formula; and 600 mosquitoes were necessary for this project. For this study, about 1103 specimens were caught, and then Anophelinae and Culicinae mosquitoes were identified using valid identification keys [37, 38]. Molecular tests were performed on both Anophelinae and Culicinae mosquitoes.
2.3. Molecular Experiments
2.3.1. Designing Primer for Molecular Detection of Rickettsia spp. and Coxiella burnetii
The primers were designed to identify Rickettsia spp. and Coxiella burnetii in mosquitoes. Therefore, the DNA sequences of Coxiella burnetii (MH920311.1 and CP018150.1) and various Rickettsia species (CP002912.1, CP002912.1, CP002912.1 and MG906669.1) were obtained from the NCBI website; and the alignment was done using Mega6 software, and then the primer was designed based on Conserve points using Oligo7.0 and Generunner4.0 software according to gltA (Rickettsia spp. citrate synthase kinase) and IS1111 A Transposase genes to identify Rickettsia and Coxiella burnetii species (Table 1).
| Expected Size | (5’_3’) Primer Sequence | Primer Name | Gen |
|---|---|---|---|
| 828 bp | TCT CAT CCT ATG GCT ATT ATG C CTT CGT GCA TTT CTT TCC ATT |
Glt F Glt R |
gltA (Rickettsia spp. citrate synthase kinase) |
| 813 bp | TAT GTA TCC ACC GTA GCC AGT C CCA ACA ACA CCT CCT TAT TCC C |
Cox F Cox R |
IS1111 A Transposase gene |
2.3.2. DNA Extraction from Mosquitoes
After the identification of mosquitoes in the present study, the DNA extraction was performed using Gene all kit (cat.No. 106-101 South Korea). Procedures of this method were carried out in accordance with the manufacturer's instructions. Due to the small sizes of mosquitoes, about 20 mosquitoes of the same genera and species were extracted for each time (pooled). Concentrations of prepared DNA were measured using the NanoDrop machine; and necessary amounts (100-200ng) for each of them were calculated to enter the PCR.
The quality of DNA extraction using the mentioned kit was very good. DNA samples extracted from mosquitoes in most cases had such characteristics (OD= 680ng/µl DNA purify=1.82). In addition, to ensure the presence of the desired DNA, two microliters of it were run on 1% gel, and the band was checked for sharpness.
2.3.3. Conventional PCR
Materials with listed concentrations in Table 2 with a final volume of 20μl were used to do the conventional PCR.
Variables of conventional PCR during the research were obtained from using specific forward and reverse primers of the gene as well as the PCR at the specific binding temperature (Annealing) relating to the same primers. All PCRs were done using the Analytikjena machine made in Germany. The following program was used to do the conventional PCR (Table 3).
The present study used ready tubes of Taq DNA Polymerase 2x Master Mix RED of Amplicon Cat. No.: A190303 (Taq 2x master mix, red, and 1.5mM mgch2) for doing the PCR. The tubes had all necessary materials to do the PCR, including Buffer 10X, MgCl2, dNTPs and Taq DNA polymerase solution; and specific primers, Template DNA and adjusted the volume to 20 μl by adding double-distilled water (DDW). To do the test, 1 μl of DNA template (at a concentration of 100 to 200 ng/ μl), 12.5 μl of master mix, 1 μl of both primers, and DDW for adjusting up to 20 μl entered the microtube 0.2 then the materials were slowly pipetted and then a small Spin was done. The 0.2 microtube was put in the thermal cycler device, and the schedule was given to the device. After the required time and the device turned-off, PCR product was run on the 1% gel agarose; the electrophoresis tank was adjusted to a voltage of 100 volts. After 1 hour, the gel was put in the Gel Documentation System to see DNA bands, and the attached monitor to the system showed sizes of bands by turning on the UV button [34, 39, 40].
Positive control of Coxiella burnetii and Rickettsia spp. were prepared from Pasteur Institute of Iran (IPI) and used for the molecular assay.
3. RESULTS
3.1. Entomological Study
In the present study, a total of 1103 adult culicidae mosquitoes were collected and identified from four regions of Qir and Karzin County, including Qir, Efzar, Emam Shahr, and Karzin during the activity seasons of mosquitoes (spring, summer and autumn of 2017-18). Among them, 3 genera and 11 species were identified, including Anopheles (25.74%) (An. dthali, An. stephensi, An. superpictus), Culex (51.84%) (Cx pipiens, Cx sinaiticus, Cx bitaeniorhynchus, Cx theilers, Cx laticinctus, Cx tritaeniorhynchus, and Cx torrentium) and Culiseta (22.39%) (Cu. longiareolata) genera (Table 4).
| Final Concentration | Amount | PCR Mix Component |
|---|---|---|
| 1 X | 2 µl | PCR buffer 10X |
| 400 nM | 1.0 µl | Forward primer |
| 400 nM | 1.0 µl | Reverse primer |
| 1.5 mM | 0.75 µl | Mg Cl2 (50 mM) |
| 1 unit | 0.2 µl | Taq DNA polymerase |
| 0.2 mM | 0.5 µl | dNTPs (10 mM) |
| 100-200 ng/reaction | Variable | DNA |
| - | adjust to 20µl | Water(DDW) |
| Final Extension | Extension | Annealing | Denaturation | Initial Denaturation | Stages |
|---|---|---|---|---|---|
| 72 °C | 72 °C | 54 °C Glt 58 °C Cox |
94 °C | 94 °C | Temperature |
| 10 min | 60 seconds | 30 seconds | 30 seconds | 5 seconds | Time |
| 1 cycle | 35 Cycle | 1 cycle | Number of cycles |
| Genus | Species | Sampling Locations | No. of Collected Samples |
Percentage (%) |
|||
|---|---|---|---|---|---|---|---|
| Qir | Efzar | Karzin | Emam Shahr | ||||
| Culex | Cx. pipiens | 69 | 26 | 17 | 25 | 137 | 12.42 |
| Cx. sinaiticus | 55 | 220 | 30 | 102 | 407 | 36.90 | |
| Cx. bitaeniorhynchus | 0 | 3 | 0 | 0 | 3 | 0.27 | |
| Cx. theileri | 5 | 11 | 0 | 0 | 16 | 1.45 | |
| Cx. laticinctus | 3 | 6 | 0 | 0 | 9 | 0.82 | |
| Anopheles | An. stephensi | 22 | 28 | 33 | 41 | 124 | 11.24 |
| An.dthali | 58 | 52 | 15 | 29 | 154 | 13.96 | |
| An. superpictus | 0 | 3 | 0 | 3 | 6 | 0.54 | |
| Culiseta | Cu. longiareolata | 123 | 55 | 38 | 31 | 247 | 22.39 |
| Total | 335 | 404 | 133 | 231 | 1103 | 100 | |
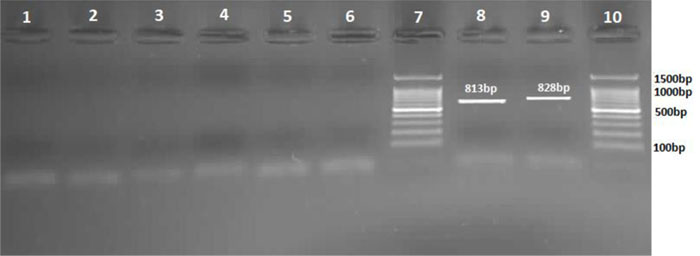
3.2. The Possible Contamination of Adult Mosquitoes with Rickettsia sp. and Coxiella burnetii
The collected adult mosquitoes in this county were examined in terms of possible contamination with Rickettsia spp. and Coxiella burnetii using the conventional PCR in the medical entomological lab of the Faculty of Health at Shiraz University of Medical Sciences. After doing molecular assays, samples were all negative for Rickettsia spp. and Coxiella burnetii. Positive control of Coxiella burnetii and Rickettsia spp. were detected in this assay with the same procedure (Fig. 2).
4. DISCUSSION
In general, there are few studies on Rickettsial diseases in Iran. A study was recently conducted by Ghavami et al. who investigated an ITS1 part of gltA gene and Rickettsia contamination in human flea (Pulex irritans) in Iran. Their results indicated that 4.9% out of 182 samples of human flea were contaminated with Rickettsia sp [41]. Azizi et al. conducted an overview of epidemic Typhus in the world in the 19th and 20th centuries and stated that typhus was a major Rickettsial disease in Iran [24]. Historically, typhus (Rickettsial disease) was a major health problem with an economic impact on Iran; however, there is limited published information on the prevalence of typhus over the centuries. Typhus has been an endemic disease in Iran, but its prevalence has been rare. In the Qajar period (1796-1925), infectious diseases, including typhus were common. In the twentieth century, the prevalence of typhus occurred in Iran [24].
Spotted Fever Group (SFG) Rickettsioses occur worldwide and may cause serious illnesses in humans. They are transmitted to humans by vector arthropods such as ticks, fleas and lice [42-47]. In the Middle East, a number of known species of Rickettsia genus have been reported, but some studies are suspicious [48]. Al-Deeb et al. reported the first case of spotted fever group of Rickettsia sp. and Theileria annulata in Hyalomma dromedarii tick that was collected from camels in the United Arab Emirates [49]. Furthermore, Rickettsia sibrica mongolitimonae was diagnosed in a male patient in Turkey in 2016 [50]. Mediterranean Spotted Fever (MSF) is caused by Rickettsia conorii subsp. Israelensis is the main cause of fever and chills from spotted fever group rickettsiosis in collected ticks from mice, deers, goats, red foxes and wild boars [48, 51].
According to conducted studies in the Middle East, the presence of different types of ticks, which can transmit Rickettsia in several countries including Yemen, Saudi Arabia, and Oman throughout the Middle East, can cause Rickettsial diseases in the Middle East [48, 52, 53]. Despite the evidence of the presence of Rickettsial SFG pathogens in the Middle East, there is no study on this disease in Iran. Therefore, it is necessary to study the SFG Rickettsiae to have a better understanding of the epidemiology of this disease in Iran [48]. There is only a paper on the spotted-fever group rickettsioses, and it investigates 40 samples of human serum collected from 4 countries, including Iran, for the presence of spotted fever group rickettsia antibodies among other pathogens by Kovacova et al. From 40 human laboratory samples, 45% were positive in terms of SFG rickettsia by the ELISA test, and 27.5% were positive using the IFA test [48, 54].
In the study of Idris et al. [2000], 347 human serum samples for Rickettsia infections were obtained in Dhofar, Oman, and their results indicated that 59% of samples were positive [55].
Studies indicate that there are different genera of the Rickettsia order, including Coxiella, Anaplasma, Ehrlichia, and Wolbachia in different arthropods in Iran. Coxiellosis, erlichiosis, anaplasmosis and SFG rickettsioses, which are zoonotic, increase the presence of their agents in the environment and the risk of human-animal transmission. The insufficient information about SFG Rickettsioses in Iran indicates the absence of pathogens in the population. However, there is a need for studies on the SFG Rickettsioses among a wide range of organisms across the country as it is difficult to determine the range of hosting, the zoonotic potential and actual prevalence of SFG Rickettsioses in Iran. Several studies indicate the presence of Rickettsia species in Iran; some of them are as follows:
Spitalska et al. [2004] examined tick-borne diseases in small domestic ruminants in Iran. Collected ticks were infected with Ehrlichia ovina, Theileria lestoquardi and Theileria spp [56].
Jalali et al. [2013] investigated the Anaplasma infection in blood samples of sheep in Ahvaz by the PCR-RLFP method. In 80% of samples, there was an infection caused by A. ovis and A. marginale [57]. Noaman et al. (2017) conducted an overview of Anaplasmosis in the Iranian ruminants [58].
Yousefi et al. [2017] investigated the molecular evidence of the presence of Anaplasma phagocytophilum as a tick-borne pathogen in small domestic ruminants in Iran. One percent of blood samples were positive. The research was the first report of this pathogen in Iran [59]. Parvizi et al., (2013) isolated Wolbachia pipientis from Phlebotomus kandelakii and Phlebotomus perfiliewi sandflies [60]. Karimian F., et al., [2018] studied wsp-based analysis of Wolbachia strains associated with Phlebotomus papatasi and P. sergenti (Diptera: Psychodidae) main cutaneous leishmaniasis vectors, the introduction of a new subgroup wSerg [61].
Q fever is caused by a small and intercellular gram-negative bacterium, called Coxiella burnetii. Domestic ruminants, including cow, sheep and goats, are considered the main reservoirs of C. burnetii to cause human infection. Infected ticks can play important roles in the transmission of bacteria among mammals, rodents and other mammals as well as wild and domestic birds. Q fever is domestic in Iran [23, 29, 62]. It has been reported that rodents have been infected by named infectious agents in Iran during the 1970s; however, there has been no report of rodent infections and transmission to humans in recent decades [62]. There are many reports about the infection of other animals and their products with C. burnetii in different provinces of Iran like Fars, Khuzestan, Yazd, Chaharmahal and Bakhtiari, Isfahan, Gilan and Mazandaran [23, 26, 27, 29].
In research by Rahimi in 2010, it was investigated that the prevalence of C. burnetii in cow's milk samples from dairy cattle of Fars, Kerman, Qom, Yazd and Khuzestan provinces. Eighteen percent of collected samples from Fars were infected [27].
In a study by Doosti et al. in 2014, the prevalence of C. burnetii in camels of Iran was studied. In this study, blood samples were taken from 130 camels; and the PCR method was used for their study. Ten percent of samples were positive [63].
A research was conducted by Parhizgari et al. in 2017, and they reviewed Emerging and re-emerging infectious diseases in Iran and declared that Q fever was an endemic disease caused by C. burnetii in Iran [64].
Rahmdel et al. [2018] investigated C. Burnetii in cow's milk samples in Shiraz (capital of Fars province) using the PCR method. Their results indicated that 3% of milk samples were contaminated, and cows' raw milk could be a potential source of C. burnetii in Shiraz in southern Iran [65]. Ghavami et al. (2018) investigated ITS1 spacer and Rickettsia infection rate in human fleas (Pulex irritans) using the molecular method. Their results indicated that 4.9% of 182 samples of human fleas were infected with Rickettsia spp [41]. Traub et al. (1974) studied the ecology of scrub typhus. In their study, they predicted that the infection could be present in the northern regions of Iran despite no evidence of infection [66].
In recent years, studies have indicated that mosquitoes can be potential vectors for Rickettsiae. According to studies, the importance of the presence of mosquitoes has multiplied in regions where there are many vector-borne diseases; and mosquitoes can also be considered as the vectors of Rickettsiae. Some studies on the potential of mosquitoes for transmitting Rickettsial diseases are as follows:
Among the first studies on the presence of Rickettsiae in mosquitoes, research reported the presence of intracellular microorganisms of Rickettsia in ovarian and testicle cells of Culex pipiens mosquito in 1924 [30]. Later, this bacterium was known as Wolbachia pipientis, a member of Anaplasmataceae family in Rickettsiales order. In addition, Yen introduced another type of transovarially transmitted Rickettsia using electron microscopy in 1975 [31].
In 2006, Horta et al. could develop R. Felis in the c6/36 cell of Aedes albopictus mosquito. Their results indicated that mosquito cells could be used to cultivate this rickettsia. In the study, 90 to 100% of cells were infected after adding pathogen to these cells after 15 days of incubation at 25 °C [33].
In a similar study in 2007, they examined the proliferation of transmitted Rickettsiae by arthropods in two types of mosquito cells. In this study, they used two cells, Aedes albopictus Aa23 and Anopheles gambiae Sua5B. They proliferated three species, namely Rickettsia felis, R. montanensis and R. peacockii in the cells. They found that the cells were also suitable for the cultivation of difficult - cultivable Rickettsiae such as R. peacockii [67].
Another study in 2011, examined the impact of Wolbachia on Culex quinquefasciatus. It was found that this kind of Rickettsia affected the ovary and reproduction of mosquito. The infected females are laying eggs before the usual time, and these eggs spend more time than healthy eggs. Healthy females produce more eggs than infected ones [68].
In a study in 2011, the R. Felis infection was investigated as a pathogen that was transmitted by mosquitoes in the future. They argued that due to the separation of this Rickettsia from people with fever in tropical regions, where there were numerous Ae.albopictus and Ae.aegypti mosquitoes, and they easily bit people, Aedes spp. mosquitoes could spread infection in the future [32]. In a study in the same year, they investigated the role of Wolbachia in controlling mosquito-borne diseases. The results indicated that Fruit fly Wolbachia strains could infect mosquitoes and affected the longevity, reproduction and pathogen entry to the mosquito's body [69].
Research in 2012 provided an examination of Rickettsia species in Anopheles of Africa. The real-time PCR and conventional PCR method were used by sequencing gltA, ompB, ompA and sca4 genes. DNA of Rickettsia was not separated from An.arabiensis, An.funestus and An.gambiae (molecular form M). R. felis was separated from An.gambiae molecular form S as the most important vector of malaria in Africa [7].
In a study in 2015, the researchers investigated a potential transmission of R.felis by An.gambiae mosquito. In this test, An.gambiae were infected with Rickettsia in the laboratory, and the infected mosquitoes were exposed to balb/c rats. The results indicated that the mosquitoes were able to transmit R. felis to rats; hence, mosquitoes could be potential vectors of this Rickettsia [70].
In another study in 2017, the researchers examined molecular characteristics of the species of mosquito-borne Rickettsiae in collected sample mosquitoes from a Korean civilian region. In this study, Rickettsiae were detected using real-time PCR and sequencing rss, gltA, 17kDa, ompB, and sca4 genes. Three new Rickettsia genotypes were detected from Mansonia uniformis, Culex pipiens, and Aedes esoensis [71].
In a research in Africa in 2017, a new species of Wolbachia was detected in malaria vectors. They could isolate this bacterium from 5 Anopheles species [72].
In one of the latest studies in this field in 2019, the researchers studied R. felis in mosquitoes in China. They investigated three species, including Culex pipiens, C. tritaeniorhynchus, and Aedes albopictus in terms of Rickettsia; and all three species were positive [73].
CONCLUSION
All these studies indicate the higher importance of mosquitoes in regions where there are Rickettsia infections. There are few studies on the presence of Rickettsial diseases in Iran.
According to the results of this study, mosquitoes in this part of the country are not considered as vectors of Rickettsia spp. and Coxiella burnetii currently. We can attribute the negative outcomes to several main factors, such as the inability of mosquitoes to support pathogens in order to establish, grow and replicate within their bodies and to have different geographic conditions.
In general, limited studies have been conducted on Rickettsial diseases in Iran, and since Rickettsia spp. has been reported from different vectors in Iran and other neighboring countries, there is a need for further studies on this field in tropical areas of Iran, especially in mosquitoes as a possible vector with high abundance and mobility.
Given the fact that there are numerous travels to different regions of the world with Rickettsia infection such as Southeast Asia, and Middle Eastern countries; and people enter Iran from neighboring countries such as Pakistan with the possibility of infection, there is a need for more comprehensive and accurate studies on this field, especially in Southern provinces of Iran such as Sistan and Baluchestan, Fars, khozestan, Bushehr, and Hormozgan.
Rickettsial disease should be considered important and travelers, who are returning from neighboring countries especially Saudi Arabia travelers, for the Hajj ritual with symptoms of fever and headache, should be screened. The importance of these diseases and their causes should be taken seriously into consideration before they create serious problems for the society.
It is suggested that a comprehensive study at a wider level should be conducted in other endemic areas of Iran in order to define the prevalence of Rickettsial infection rates in mosquitoes. In this case, we will be able to understand the exact role of mosquitoes in the cycle of transmission of these pathogens to humans in Iran.
AUTHORS' CONTRIBUTIONS
ZH and AS; Designed and performed experiments, analyzed data and co-wrote the paper. ZH, AS, KA, and HA; Performed experiments and co-wrote the paper. AS, KA, and HA; Supervised the research.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No human or animals were used in this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The data that support the findings of this study are available from the corresponding author, [AS], upon reasonable request.
FUNDING
This paper has partly been derived from, and supported by Ms. Zahra Hoseini Master of Science thesis (Grant No: 1396-01-04-16676) in Medical Entomology, at School of Health, Shiraz University of Medical Sciences, Shiraz, Iran.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Authors are thankful to the assistance of vice-chancellorship for research and technology at Shiraz University of Medical Sciences (SUMS). Also we are really thankful of Dr. Ehsan Mostafavi from pasture institute of Iran for giving us the positive control of Coxiella burnetii and Rickettsia spp.


