All published articles of this journal are available on ScienceDirect.
Anti-Toxigenic Effect of Lactic Acid Bacteria Against Aspergillus spp Isolated from Wheat Grains
Abstract
Introduction:
Many fungi infect the wheat grains. Under field and or storage conditions from temperature and humidity, some fungi can produce aflatoxins (AFs), which may cause acute or chronic diseases. Therefore, there is a necessary and urgent need to find an effective and safe way to reduce or remove AFs.
Objective:
The objective of this study was the evaluation of Lactobacillus rhamnosus, Lactobacillus gasseri, and Lactobacillus plantarum for their ability to reduce and or remove AFs produced by Aspergillus flavus and Aspergillus parasiticus, which were isolated from wheat grains, as well as control of AFs produced on affected wheat grain by A.parasiticus spores only.
Methods:
LAB, isolated from some local dairy products, were cultured in MRS for the evaluation of their ability to remove AFs, produced by A. flavus and A. parasiticus on (YES) media, in addition to the treatment of wheat grains by LAB cells to prevent AFs produced by A. parasiticus.
Results:
The L. rhamnosus strain gave the highest reduction rates of AFs produced by A. parasiticus that were 62.6, 44.4, 43.3, and 52.2% for AFG1, AFB1, AFG2, and AFB2, respectively. While in the case of A. flavus, the reduction was 50.4, 42.7, 40.6, and 36.8% in the same order of toxins. When applied, these strains with wheat grains were affected by A. parasiticus, the inhibition rates of AFs were ranged between 61.4 and 75.8% with L. rhamnosus strain and 43.7 to 52.1% with L. gasseri, while L. plantarum strain ranged from 55.5 to 66.9%.
Conclusion:
According to this study, L. rhamnosus is considered one of the best strains in this field. Therefore, the present study suggests applied use of LAB as a treatment to prevent AFs production in wheat grains.
1. INTRODUCTION
Aflatoxins (AFs) are a group of secondary metabolites produced by Aspergillus flavus, A.parasiticus, A. nomius, A. bombycis, and A. pseudotamarii [1, 2]. High temperature and high relative humidity are conducive for the growth of fungi producing AFs on stored grains. The incidence of AFs in foods and feeds is relatively high in the tropical and sub-tropical regions due to the warm and humid weather conditions that provide ideal conditions for the growth of the aflatoxigenic molds. The four major naturally produced AFs are known as aflatoxin B1 (AFB1), aflatoxin B2 (AFB2), aflatoxin G1 (AFG1), and aflatoxin G2 (AFG2). AFs have many bad effects on humans and animals. If ingested, AFs may cause acute or chronic diseases, such as carcinogenic, mutagenic, teratogenic, estrogenic, hemorrhagic, nephrotoxic, hepatotoxic, neurotoxic, as well as immunosuppressive effects [3-5]. Numerous studies around the world indicated the contamination of most cereal products, with one or more types of AFs, inducing large economic losses [6, 7]. Once the contamination has occurred by AFs, many methods must be established and applied for controlling or reducing AFs for the prevention and detoxification in order to preserve the safety of products intended for human consumption. Many microorganisms, including bacteria, yeasts, and molds, are capable of reducing toxins in foods and feeds, therefore, many studies used lactic acid bacteria (LAB) to bind toxins in vitro and in vivo [8-10].Therefore, this study aimed for the isolation and identification of fungi from wheat grains then investigated the ability of three strains from LAB (L. rhamnosus, L. gasseri and L.plantarum) isolated from some local dairy products for the reduction of AF s produced by A. parasiticus and A. flavus, which were isolated from wheat grains, in addition to the treatment of wheat grains by these strains for inhibition AFs produced by A. parasiticus.
2. MATERIALS AND METHODS
2.1. Materials
2.1.1. Wheat Grain Samples
Fifty (50) samples for wheat grains were collected from local markets of Cairo and Giza. Samples were subjected to mycological examination.
2.1.2. Lactic Acid Bacteria Strains
In this study, three strains from LAB ( Lactobacillus rhamnosus, Lactobacillus gasseri and Lactobacillus plantarum) were used which were isolated from some local dairy products. These strains were identified according to their morphological and cultural characteristics [11] and using -50CHLAPI- identification system (BioMerieux). [12].
2.2. Methods
2.2.1. Isolation of Fungi from Wheat Grains
Potato dextrose agar (PDA) medium was used for the isolation of fungi according to the method described by Pitt and Hocking [11] for un-sterilized wheat grain, while sterilized wheat grain, with the help of sterile forceps, were surface sterilized for 5 min in 1% sodium hypochlorite, then rinsed three times with sterilized distilled water. These grains were dried on sterilized filter paper.
2.2.2. Purification and Identification of Fungal Isolates
All the isolated fungi, as well as the microsocial structures on PDA medium, were identified by studying the cultural characteristics, according to previous studies [13, 14]. The frequency of fungi and relative percentage of particular species within a genus of fungi was calculated using the formula of Ghiasian et al. [15].
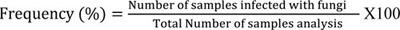 |
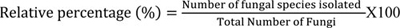 |
2.2.3. Determination of Toxicity for Some Isolated Fungi
The isolated Aspergillus spp. (A. parasiticus and A. flavus) were tested for their ability to produce AFs by cultivation on Yeast Extract Sucrose Broth (YESB) broth for 8 days at 28˚C, according to Frisvad et al [16].
2.2.4. Preparation of Bacteria Culture Cells
The isolates of LAB were propagated overnight (16 h) in 100 overnight (16 h) in 100 ml MRS broth (pH 6.8).A cell free solution was obtained by centrifugation of culture at 5000 rpm at 4°C 20 min, followed by filtration of supernatant through a sterilized filter (0.2 µm, Millipore), to take all cells.
2.2.5. Effect of LAB Cells on AFs Production
A loopful of LAB from the Man-Rogosa-Sharpe medium was separately incubated in tryptic soy broth at 37 °C for 18 h. Then, one mL of culture medium (density equivalent to 0.5 MacFarland standard) was added to a 100 mL Erlenmeyer flask containing 25 mL of yeast extract sucrose broth with 105 spores/mL each for A. parasiticus and A. flavus. Mycelial mass and AFs production rates were measured according to the method described previously [17]. After incubation, a medium containing LAB and fungus was filtered, then mycelia weight was measured, as AFs were extracted from filtrates according to the method described previously [18, 19], where extraction was carried out using 20 mL of chloroform (with 20 ml filtrate), and homogenization for 3 min in a separation funnel. The chloroform phase was filtered through Whatman filter paper with sodium sulphate anhydrous and concentrated to dryness using a hot plate then determined using HPLC.
2.2.6. Preparation of Treated Wheat Grains Samples
Autoclaved wheat grains were used in the following seven experimental groups (100gin each): (1) Positive control: wheat grains inoculated with a spore suspension of A. parasiticus were prepared according to Sharma et al. [20]. Groups 2, 3, and 4 negative controls: wheat grains inoculated with 2.5mL for each of L. rhamnosus, L. gasseri, and L. plantarum, respectively; groups 5, 6, and 7 wheat grains infected by A.parasiticus spore suspension plus L. rhamnosus, L. gasseri, and L. plantarum, respectively. All the inoculations were performed by the sprinkling of grains with microbial suspensions, followed by incubation for seven days at 28 ºC [21]. On the other hand, the quantity of AFs in wheat grains samples was measured by using the Immun affinity column (Aflatest®-P affinity column) for extraction as well as HPLC for the determination of AFs concentrations [22].
2.2.7. Statistical Analyses
All data were statistically analyzed using the General Linear Model procedure of the SPSS ver. 18 (IBM Corp, NY). The significance of the differences among treatment groups was determined by Waller–Duncan k-ratio. All statements of significance were based on the probability of P-value <0.05, which was considered to be statistically significant.
3. RESULTS AND DISCUSSION
3.1. Isolation and Identification of Fungi
The results recorded in Table 1 revealed that the fungi-contaminated wheat grains samples were grown on PDA disinfected and non-disinfected grains. The Total Fungi Count (TFC) isolated were 382 and 312 isolates in non-sterilized and sterilized wheat grain, respectively. Also, data showed that the most isolated fungal species were identified as Aspergillus, Pencillium, Fusarium, and Alternaria.
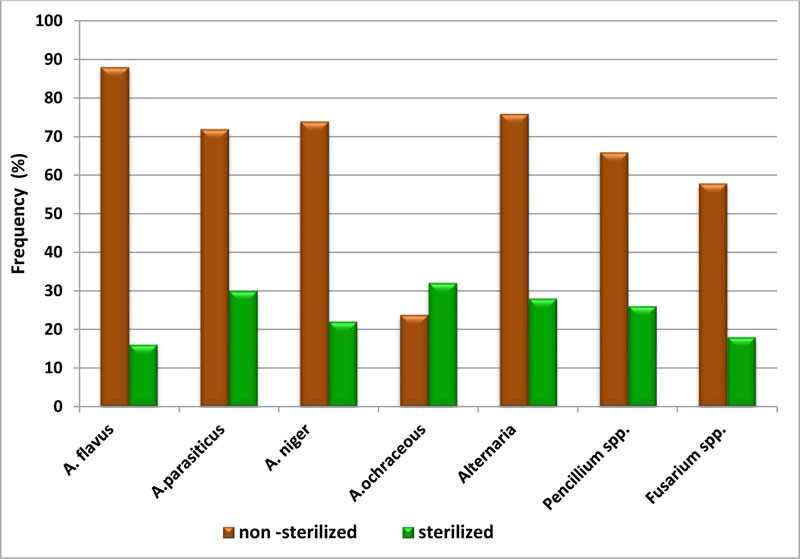
The frequency of isolated fungi from non – sterilized and sterilized wheat grain samples shown in Fig. (1) revealed that the highly frequent species were A. flavus (88%) and A.parasiticus (72%) as well as A. niger (74%) in non–sterilized wheat grains. On the other hand, the results indicated that the highly frequent species in sterilized wheat grains samples were A. ochraceous (32%) and A. parasiticus (30%). These results are in agreement with previous studies [12, 23]. This is mostly related to the physiology of fungi and their adaptation to the different matrices and environmental conditions [24]. Fungal spoilage is caused by two factors: (biotic) living, which include insects, birds, rodents, and microorganisms, and (non-biotic), non-living, which include temperature, humidity, and time [25]. Deterioration of grain is mainly affected by moisture content, temperature, relative humidity, storage conditions, fungal growth, and insect pests. In grain, the occurrence of fungi, especially A. flavus and Fusarium spp., facilitated by hot and humid conditions, poses a risk through the production of mycotoxins. In order to maintain high quality maize for both short- and long-term storage, wheat must be protected from weather, growth of microorganisms, and pests [26].
In Egypt, the weather gives a chance for the growth of Aspergillus species and other fungi on grains as it is characterized by high temperature and high relative humidity. Also, contamination occurs through small amounts of spores contaminating the grain as it goes into storage from the harvest in handling and storage equipment or from spores already present in storage structures. However, wheat grains were cultivated in the winter season (November /December) and harvested in the summer, which enhanced the chances of pre-harvest contamination. Also, the farmers have used old traditional farming practices, which can enhance fungal infections [27].
3.2. Screening of AFs Production by Some Isolated Strains
In this study, the isolates of A. flavus and A.parasiticus from surface-sterilized wheat grains were investigated to produce of AFs. The results in (Table 2) clarify that 6 and 8 strains of A. flavus and A.parasiticus were producers for AFs, respectively.
| Name of the Fungi | Non –sterilized Wheat Grain | Sterilized Wheat Grains | ||||
| No. of Samples Infected | No. of Isolates | R.P (%) |
No. of Samples Infected | No. of Isolates | R.P (%) |
|
| A.flavus | 44 | 98 | 25.6 | 8 | 14 | 10.1 |
| A.parasiticus | 36 | 56 | 14.6 | 15 | 22 | 15.8 |
| A.niger | 37 | 77 | 20.1 | 11 | 25 | 17.9 |
| A.ochraceous | 12 | 14 | 3.7 | 16 | 16 | 11.5 |
| Alternaria | 38 | 44 | 11.5 | 14 | 28 | 20.1 |
| Pencillium spp. | 33 | 55 | 14.4 | 13 | 13 | 9.3 |
| Fusarium spp. | 29 | 38 | 9.9 | 9 | 21 | 15.1 |
| TFC count/5seeds | 382 | 139 | ||||
| Type of Fungal | Numbers | Concentration of AFs (ng/100ml) | |||||||
| AFG1 | AFB1 | AFG2 | AFB2 | ||||||
| Max. | Min. | Max. | Min. | Max. | Min. | Max. | Min. | ||
| A. flavus | 14(6)* | 35.15 | 8.31 | 75.23 | 21.77 | 24.19 | 13.8 | 32.15 | 6.23 |
| A. parasiticus | 22(8)* | 37.38 | 6.4 | 57.42 | 8.21 | 33.1 | 11.22 | 20.8 | 7.77 |
The production of AFs by A. flavus and A.parasiticus depends on the possession of AFs genes. The isolates varied widely due to the prescience of seven toxigenic gene (aflR, aflS, aflQ, aflP, aflD, aflM, and aflO). The isolates of toxigenic A.flavus possessed at least 5 (out of 7) of these genes that were not detected in most of the isolates, as there were variations in AFs type [28]. In A. flavus and A. parasiticus, AFs pathway genes are clustered within a 75-kb region of the fungal genome on chromosome III [29, 30]. AFs biosynthesis is coded by an 80 kb long DNA sequence, as a cluster containing 30 putative genes characterized in both A. flavus and A. parasiticus.The variations in Toxin Type among aflatoxigenic fungi this due to the metabolic behavior of strains according to the molecular genetics and phylogenetic relationships. A. flavus differs from A. parasiticus by loss of a portion of the gene, aflU (cypA), involved in G type AFs production as well as A. flavus and A. parasiticus group forms a polyphyletic assemblage containing isolates of different morph types and having the ability to produce AFs [31].
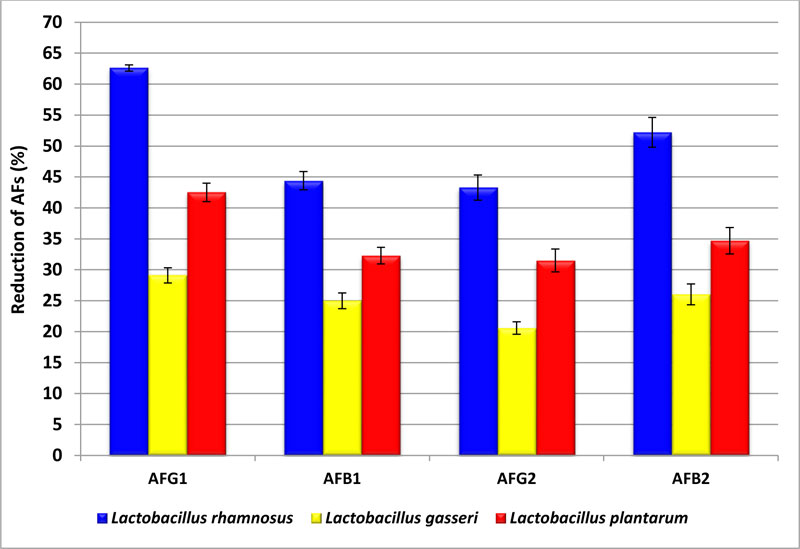
3.3. Control of AFs Produced in Liquid Media by Addition Some LAB Strains
In this study, three strains from LAB were used to control the AFs produced on Yeast extract sucrose (YES) media by A. flavus and A. parasiticus. The ability of LAB strains to reduce AFs produced by A. parasiticus ranged from 20% to 62.6%. The results in Fig. (2) indicated that the highest rate of reduction of AFs were with L. rhamnosus strain that removed AFG1, AFB1, AFG2, and AFB2 up to 62.6, 44.4, 43.3, and 52.2%, respectively. While in the case of L. gasser, the strain rate of reduction of toxins was decreased to 29.1, 25.0, 20.6, and 26.03% with AFG1, AFB1, AFG2, and AFB2, respectively. However, L. plantarum strain reduction was 42.5% and 31.5% for AFG1 and AFG2, respectively.
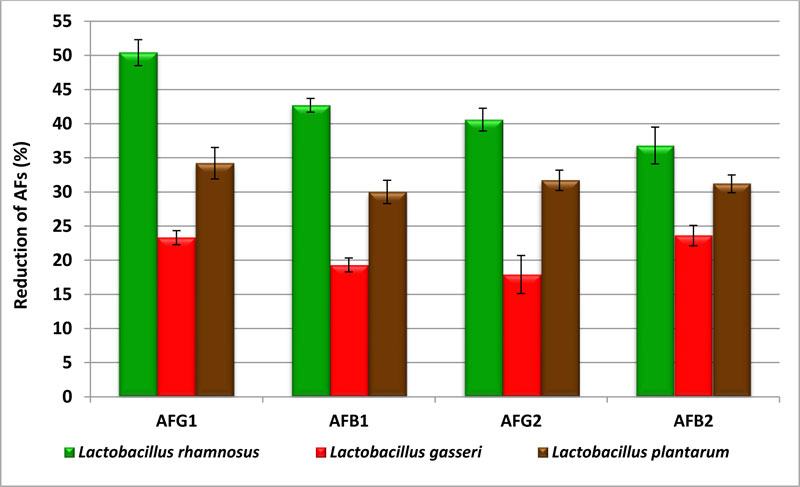
The data in Fig. (3) showed the impact of LAB on AFs produced by A.flavus isolated from wheat grains. When treated with the YES media by Lrhamnosus, AFG 1 and AFG2 reduced to 50.4 and 40.6%, respectively. The production of AFB1 and AFB2 decreased to 42.7 and 36.8%, respectively. The highest rate reduced with L. plantarum was 34.2% with AFG1. The results showed that L.gasseri has the ability to degrade AFs contents in medium ranging from 17.9 to 23.6%. These LAB strains used in this study have a significantly higher reduction on AFs when used in liquid media. Also, there were significant differences depending on the type of toxin. Analysis of variance and Duncan analysis showed significant of (p≤0.05) on the reduction of AFs (Tables 3 and 4).
The interaction between ingredients of LAB cell wall with AFs may be due to the adsorbance of AFs through treatments affecting LAB wall polysaccharides, lipids, and proteins causing an increase in the binding with AFs, whereas LAB cell wall binds the toxin with non-covalent weak bonds accompanied by some electrostatic attraction through lactinine like protein, polysaccharides, and peptidoglucan. The binding between AFs and bacterial cell wall ingredients modifies AFs structures and gets a new structure form. This mechanism was with four types of AFs because the activity of this type of AFs depends on the same active groups, such as double bonds, OH, CHR3R, etc [32-34].
| Source | SS | df | MS | F | P |
| Intercept | 49387.65 | 1 | 49387.65 | 4272.239 | 0.0000 |
| LAB | 3949.242 | 2 | 1974.621 | 170.813 | 0.0000 |
| Toxin | 871.5945 | 3 | 290.5315 | 25.13219 | 0.0000 |
| LAB*toxin | 182.8627 | 6 | 30.47711 | 2.636398 | 0.0415 |
| Error | 277.4432 | 24 | 11.56013 | - | - |
| Total | 54668.8 | 36 | - | - | - |
| Source | SS | df | MS | F | P |
| Intercept | 36627.58 | 1 | 36627.58 | 3665.609 | 0.0000 |
| LAB | 2810.304 | 2 | 1405.152 | 140.6246 | 0.0000 |
| Toxin | 201.1808 | 3 | 67.06028 | 6.711248 | 0.0000 |
| LAB*toxin | 192.4517 | 6 | 32.07528 | 3.210024 | 0.01853 |
| Error | 239.8133 | 24 | 9.992222 | - | - |
| Total | 40071.33 | 36 | - | - | - |
3.4. Control of AFs Production in Affected Wheat Grains with Spores of A. parasiticus
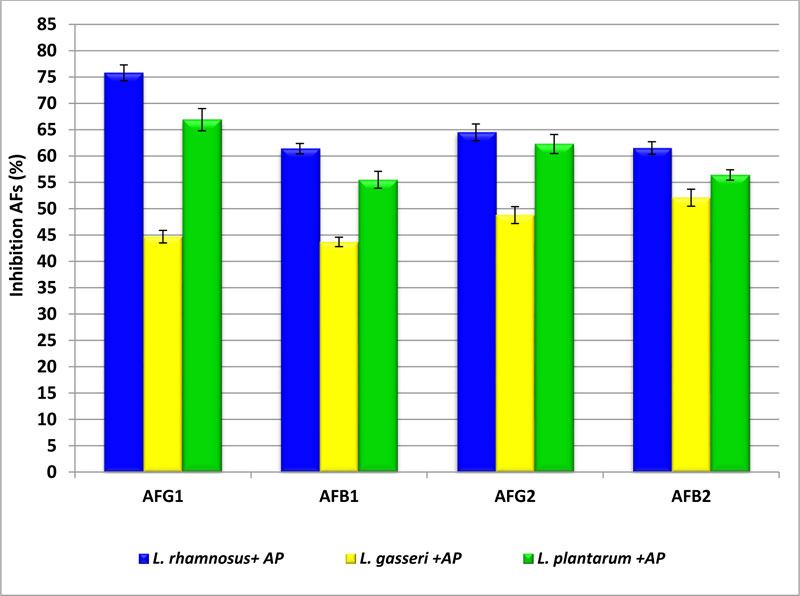
In this study, three strains for LAB were used for inhibition of the production of AFs on wheat grains. Data presented in Fig. (4) showed the percentages of inhibition of AFs in wheat affected by A. parasiticus spores after treatment (incubation) with LAB cells. When wheat grains were treated with L.rhamnosus strain, the reduction in AFG1, AFB1, AFG2, and AFB2 was 75.8±1.5, 61.4±1.0, 64.5±1.6, and 61.5±1.2%, respectively. While in the case of wheat treated with L. gasseri, the reduction of AFs was 44.7±1.2, 43.7±1.0, 48.8±1.6, and 52.1±1.6% in AFG1, AFB1, AFG2, and AFB2, respectively. AFs were removed by L. plantarum to 66.9±2.1and 62.3±1.0% with AFG1 and AFG2, respectively. On the other hand, AFB1 and AFB2 inhibition is 55.5±1.9 and 56.4±1.0%, respectively. The obtained data reflected that L.rhamnosus strain was the most efficient organism in inhibition of toxins then treatment by L .plantarum. The inhibition of AFs in wheat grains may be due to the biotransformation potential for toxins by LAB strains as well as the interaction between LAB and the accumulation of toxin through inhibition of their biosynthesis,whereas the production of AFs by A. parasiticus depends on expression genes responsible for producing of AFs which may be due to the effect of adding LAB strains [35-37].
CONCLUSION
The present study investigated the ability of L.rhamnosus, L.gasseri, and L.plantarum isolated from some local dairy products to control AFs produced by A.flavus and A.parasiticus, which were isolated from wheat grains. According to this study, L. rhamnosus is considered to be one of the best strains in this field. So that the use of LAB, which already has Generally Regarded as Safe (GRAS) status, should be encouraged for reduction and or removal of AFs. Moreover, the present study suggests applied use of LAB as a treatment to inhibit and or prevents AFs production in wheat grains.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The data supporting the findings of this research are available in the manuscript.
FUNDING
This work was supported by the National Research Centre, Egypt (Project No. 11040205) and was performed in accordance with the eleventh research plan for National Research Centre.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


