All published articles of this journal are available on ScienceDirect.
Acaulospora longula Increases the Content of Phenolic Compounds and Antioxidant Activity in Fruits of Libidibia ferrea
Abstract
Background:
Several countries have made an association that how conventional and traditional medicines support the rational use of medicinal plants in different treatments. Brazil has stimulated the cultivation of and the development of the studies of Libidibia ferrea, a plant with medicinal properties, which are provided by the presence of bioactive compounds in its several parts, such as fruits. The synthesis of these biomolecules may increase with Arbuscular Mycorrhizal Fungi (AMF) inoculation. However, there have been no previous reports about the optimization in the production of biomolecules in the fruits of mycorrhizal L. ferrea.
Objective:
The aim of this study was to define the potential advantages of inoculation with AMF by increasing the production of biomolecules and antioxidant activity in fruits of mycorrhizal L. ferrea.
Methods:
The phytochemical and mycorrhizal analyses occurred after 32 months of transplanting to the field and the plants were inoculated or not with Acaulospora longula, Claroideoglomus etunicatum and Gigaspora albida and then by transplanting the seedlings to the field.
Results:
Compared to those from the control treatment, the concentration of phenols and tannins in the fruits of L. ferrea inoculated with A. longula increased 26.4% and 39.8% (p <0.05), respectively. The fruits also had higher antioxidant activity. In contrast, the concentration of phosphorus in the fruits of L. ferrea inoculated with A. longula and C. etunicatum was not incresead. Because the concentration of phosphorus in fruits was verified in this study.
Conclusion:
Mycorrhization increased the concentration of biocompounds and antioxidant activity in the fruits of L. ferrea, which is of great interest to the herbal medicine industry.
1. INTRODUCTION
The World Health Organization (WHO) considers traditional medicine as a complement to conventional health practices, as, in some countries, it is used as the first access that people have to health treatment. Traditional medicine involves a set of products and practices which are not part of conventional medicine, including the use of medicinal plants. Therefore, the WHO seeks to develop and stimulate strategies and policies in many countries to promote the quality, safety, and efficiency of medicinal plants [1].
In Brazil, the Política Nacional de Práticas Integrativas e Complementares do Sistema Único de Saúde (SUS) [2] established, in the National Policy and Software of Medicinal Plants, a definition of strategies for sustainable cultivation and management of the production, distribution, and use of medicinal plants [3]. Many of the plants covered in these strategies, including Libidibia ferrea (Mart. ex Tul.) L.P. Queiroz, is a legume native to Caatinga, popularly known as ironwood, which is used by the population to treat several illnesses, such as diabetes, inflammations and gastrointestinal diseases [4, 5]. Its medicinal properties, such as anti-inflammatory [6], antidiabetic [7], antimicrobial [8], and antioxidant [9], have been confirmed. The plant also has antitumoral action and slows tissue ageing [10, 11]. Such benefits are associated with the presence of phytomass of secondary metabolites like phenols, flavonoids, tannins and terpenes [12-14]. These are synthetized in small quantities in comparison to primary metabolites [15].
A sustainable way to optimize the accumulation of the bioactive compounds and antioxidant activity in the plants is inoculation with Arbuscular Mycorrhizal Fungi (AMF). These fungi belong to the Subphylum Glomeromycotina, Phylum Mucoromycota [16] and form a mutualistic symbiosis with plants [17] assisting them with increased nutrient absorption [18, 19], tolerance to stress [20, 21], and growth [22, 23]. Inoculation also increases the production of primary and secondary metabolites in the plants [24-26] and antioxidant activity [15]. These benefits occur with different legume species [27, 28], including some from the Caatinga biome [19, 22, 29]. AMF inoculation also maximized the accumulation of biomolecules in leaves and the stem bark of L. ferrea [14, 25]. However, such an increase in the fruits, which are the main part used for herbal medicine, has not been reported [30, 31]. Therefore, the study aimed to define the potential of AMF inoculation by increasing the production of biomolecules and antioxidant activity in the fruits of L. ferrea.
2. MATERIALS AND METHODS
2.1. Experimental Setup
The experiment was established in February 2013 in the Experimental Field of the Laboratory of Mycorrhizal Technology (LTM/UPE), located in the University of Pernambuco – Campus Petrolina, Pernambuco (9º23’54.1” S; 40º28’49.0” W). It was set up as a random block design with four inoculation treatments and six repetitions. The treatments were: pre-inoculation of the plants with Acaulospora longula Spain & N.C. Schenck (UFPE 21), Claroideoglomus etunicatum (W. N. Becker & Gerdemann) C. Walker & A. Schussler) (UFPE 06) or Gigaspora albida N.C. Schenck & G.S. Sm. (UFPE 01), and non-inoculated control (Silva et al. 2014a). These three AMF isolates were donated by the Laboratory of Mycorrhizas of the Department of Mycology of the Federal University of Pernambuco. The AMF were multiplied using the millet (Panicum miliaceum L.) as host and soil with 10% vermicompost. The inoculum produced was stored at 4ºC until the time of use.
Seeds were collected from L. ferrea plants native to Caatinga. Exsiccate of the plants are maintained at the Herbarium Vale do São Francisco. After germination, when the plantlets showed two definitive leaves, they were transferred to jars, which contained 1.2 Kg of soil + 5% of vermicompost to be inoculated, or not, with AMF (200 spores, hyphae and colonized roots). The plants remained in the experimental netting for 225 days before being transplanted to the experimental field [14].
The field area was ploughed and holes (40 x 40 x 40 cm) were opened, forming lines and rows spaced at 5 m. The plot of each treatment was composed of 16 holes (4 x 4) and surrounding the blocks, non-mycorrhizal plants of L. ferrea were planted [30]. The two central plants of each treatment plot were used for the analyses. For fertilization, each hole was fertilized with five liters of vermicompost and 150 g of simple superphosphate. Irrigation was performed using a semi-automatic drip (8.4 L H2O plant-1 h-1), which was activated on alternate days. The surface layer (0 to 20 cm) of the soil of the experimental area had the following characteristics: P, 10.38 mg dm-3; K, 0.24 cmolc dm-3; Ca, 1.4 cmolc dm-3; Mg, 0.5 cmolc dm-3; Na, 0.03 cmolc dm-3; Al, 0.00 cmolc dm-3; organic matter 0.41, g Kg-1; pH (H2O[1:2.5]), 6.2; electric conductivity, 0.21 mS cm-1.
2.2. Phytochemical Analyses
2.2.1. Plants Extracts
Thirty-two months after transplantation, the fruits were collected, cleaned and oven-dried (Quimis Ltda, Diadema, Brazil) for three days (at 45ºC). The dried fruits, including the bark and the seeds, were cut in four pieces (mean width: 13.81 mm and mean length 12.29 mm) using garden shears (Agass Ltd, China) and then crushed in a multiprocessor (Black+Decker®, United States) for 90 seconds. Subsequently, 2 g of the crushed fruits were transferred to amber flasks and 20 mL of methanol (80%, v/v) (Química Moderna Ltda, Barueri, Brazil) was added for 10 days at 20ºC [32]. After filtration, in gauze and qualitative filter paper, the extracts were stored in a freezer (-18ºC).
2.2.2. Total Phenols
For the dosing of the total phenols, the methanolic extracts were diluted 300 times in distilled water and 200 µL of the diluted extract was transferred to flasks. Then 1 mL of the Folin-Ciocalteau Reagent (10%, v/v) (Merck®, Germany) and 800 µL of sodium carbonate (7.5%, p/v) (Vetec Ltda, Duque de Caxias, Brazil) were added. The solution was vortex-stirred (Biomixer Ltda, São Paulo, Brazil) and left to rest. It was protected from the light for 30 minutes. Subsequently, it was measured in a spectrophotometer (765 nm). Tannic acid was used for the standard curve (y= 7.562 x + 0.0081, R2= 0.9996) [18].
2.2.3. Total Tannins
The method of casein precipitation (Vetec Ltda, Duque de Caxias, Brazil) was used for the quantification of tannins. In amber flasks, 3 mL of the methanolic extract and 0.5 g of casein (Vetec Ltda, Duque de Caxias, Brazil) were added. The solution was stirred (160 rpm) for 3 hours at 20oC and filtered through a qualitative filter paper. An analysis of total phenols in the filtered material was carried out, according to Orujei et al. [18]. The concentration of total tannins was calculated from the difference between the total phenol concentration initially found in each sample, described in the previous section, and the value of total phenol concentration after the casein precipitation.
2.2.4. Total Flavonoids
For the dosage of total flavonoids, the methanolic extracts were diluted in distilled water in the proportion of 1:3. In a flask, 100 µL of the diluted extract, 60 µL of glacial acetic acid (Química Moderna Ltda, Barueri, Brazil), 1 mL of pyridine-methanol solution (2:8, v/v), 250 µL of aluminum chloride solution (5%, p/v) (Vetec Ltda, Duque de Caxias, Brazil), and 1.10 mL of distilled water were added. The mixture remained at rest for 30 minutes to be subsequently read in a spectrophotometer (420 nm). Rutin (Sigma-Aldrich®, São Paulo, Brazil) was used for the standard curve (y= 0.5954 x + 0.045, R2= 0.9986) [33].
2.2.5. Total Proanthocyanidins
The methanolic extract was diluted 100 times and dosed for total proanthocyanidins by the acid vanillin method, in which 1 mL of the diluted extract was transferred to a flask. Here, 2 mL of vanillin 2% (Vetec Ltda, Duque de Caxias, Brazil) in H2SO4 (F Maia Ltda, Cotia, Brazil) 70% (v/v/water) was added. The mixture was vortex-stirred (Biomixer Ltda, São Paulo, Brazil) and remained at rest for 15 minutes in the dark. Subsequently, realized reading spectrophotometric (500 nm) [34], using catechin (Sigma-Aldrich®, São Paulo, Brazil) for the standard curve (y= 0.412 x + 0.0026, R2= 0.9926).
2.2.6. Total Saponins
In this analysis, flasks with 1 mL of the methanolic extract, 1 mL of cobalt chloride 0.2% (p/v/water) (Nuclear®, Diadema, Brazil), and 1 mL of concentrated H2SO4 (F Maia Ltda, Cotia, Brazil) were included [35]. The mixture was vortex-stirred (Biomixer Ltda, São Paulo, Brazil) and read in a spectrophotometer (Thermo Scientific®, United States) at a wavelength of 284 nm. Saponin (Inlab Ltda, São Paulo, Brazil) was used for the standard curve (y= 0.0009 x + 0.0045, R2= 0.9926).
2.2.7. Total Antioxidant Activity
The antioxidant activity was estimated by capturing the free radical DPPH (2.2-diphenyl-1-picrylhydrazyl). In this analysis, 0.1 mL of the methanolic extract was diluted 100 times in distilled water. It was mixed with 3.9 mL of the radical of DPPH (0.06 mM) (Sigma-Aldrich®, São Paulo, Brazil) in an amber flask, then vortex-homogenized (Biomixer Ltd. São Paulo, Brazil) and placed to rest in the dark for 30 minutes. Subsequently, the solution was read using a spectrophotometer (515 nm). DPPH (y= 0.0245x + 0.0033, R2= 0.9999) was used for the standard curve, expressing the results in g of DPPH remaining in the solution [36].
2.2.8. Total Soluble Carbohydrates
For the quantification of carbohydrates, 20 µL of the extract, 95 µL of distilled water, 50 µL of phenol (80%, p/v) (Vetec, Duque de Caxias, Brazil) and 2 mL of sulfuric acid (Química Moderna Ltda, Barueri, Brazil) were transferred to test tubes. The mixture was vortex-stirred and placed to rest in the dark for 10 minutes. Afterwards, the solution was read using a spectrophotometer (490 nm). Glucose (CRQ Ltda, Diadema, Brazil) was used for the standard curve (y= 0.221x + 0.0004, R2= 0.9921) [37].
2.2.9. Quantification of Gallic and Ellagic Acids
1 g of pulverized fruit and 50 mL of distilled water were transferred to a round-bottom flask (250 mL) and the mixture was placed for 30-minutes in a water bath (Lucadema®; São Paulo, Brazil), under reflux, at a temperature of 85-90 °C. The flask was cooled in water to ambient temperature (25 ± 2°C). The solution was filtered through cotton and transferred to a 50 mL volumetric flask, which was filled with distilled water. Posteriorly, in a 10 mL volumetric flask, 5 mL of the aqueous extract was transferred and the volume completed with distilled water. The sample was filtered through PVDF filters (membrane of polyvinylidene fluoride; Macherey-Nagel®) of 25 mm. 0.45 µm for the vials.
The gallic and ellagic acids were quantified by Liquid Chromatography (Ultimate 3000, Thermo Fisher Scientific®, United States) coupled to a Photodiode Array Detector (PDA; Thermo Fisher Scientific®, United States). Wavelengths of 270 nm and 254 nm were used to detect gallic acid and ellagic acid, respectively. A chromatographic column C18 (250 mm x 4.6 mm d.i, size of particle 5 µm; Dionex®, United States) and pre-column C18 (4 mm x 3.9 µm, Phenomenex®, United States), with an oven temperature at 24°C, were used to separate them. The mobile phase constituted of ultrapure water (A) and methanol (B), both acidified with 0.05% of trifluoroacetic acid, with the flow at 0.8 mL/min. The gradient applied was 0-10 min, 12.5-25% B; 10-15 min, 25-40% B; 15-25 min, 40-75% B; 25-30 min, 75-75% B; 30-33 min, 75-12.5% B. 20 µL of the samples was injected for quantification. The standards were purchased from Sigma-Aldrich® (São Paulo, Brazil) and the purities of the gallic and ellagic acids were 98% and 96%, respectively [38].
2.2.10. Phosphorus Concentration
The quantification of phosphorus in the fruits was conducted at the Department of Nuclear Energy, Federal University of Pernambuco, following a method of ascorbic acid, using a reagent which constituted of ascorbic acid (0.1 M), sulfuric acid (5 N), and ammonium heptamolybdate (4%). In this reaction, the phosphomolybdic acid formed in the reagent due to the presence of sulfuric acid and ammonium heptamolybdate was reduced, forming a blue color which was read in a spectrophotometer [39].
2.3. Mycorrhizal Analyses
In order to perform mycorrhizal analyses, soil samples were collected in three equally spaced points in each plot. The projection of the top of the rhizosphere of the plants had a depth of 0-20 cm. To assess mycorrhizal colonization, the roots were clarified in KOH (10%, p/v) and hydrogen peroxide (10% p/v) (F Maia Ltda, Cotia, Brazil) and colored with Trypan blue (0.05% in lactoglycerol, p/v) (Vetec Ltda, Duque de Caxias, Brazil) [40]. The percentage of colonization was obtained through the quadrant intersection method [41] using a stereomicroscope (Quimis Ltda., Diadema, Brazil) (40 x).
3. RESULTS
The production of mycorrhizal structures was similar between treatments (Table 1). However, the inoculation of the ironwood plants with A. longula increased the total phenols concentration in the fruits by 26.4% in relation to the fruits of the non-inoculated plants. A benefit was not found within the other inoculation treatments (Table 1). The production of total tannins was optimized in the plants inoculated with A. longula, with an increase of 39.78% against the control treatment (Table 1), which partially proves our initial hypothesis.
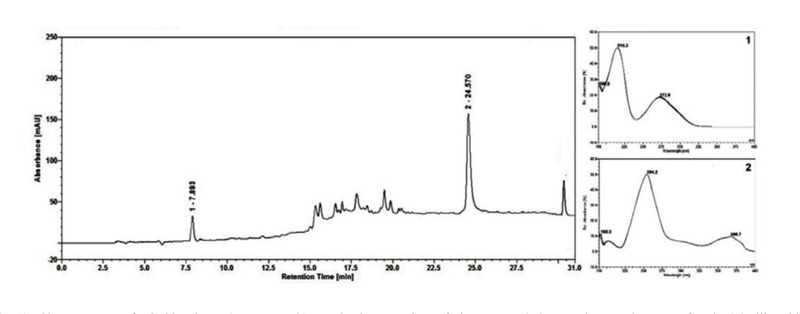
The AMF inoculation did not optimize the production of soluble carbohydrates, flavonoids, proanthocyanidins or total saponins (Table 1). The production of gallic and ellagic acids was not optimized by mycorrhization (Table 1, Fig. 1).
Considering the lowest concentration of DPPH remaining in the solution, the fruits of mycorrhizal L. ferrea with A. longula had higher antioxidant activity than the non-inoculated plants (Table 1). This confirms the negative correlation between the concentration of phenols and tannins and the concentration of DPPH (Table 2).
| Variable | Inoculation treatments | ||||
|---|---|---|---|---|---|
| Control | Acaulospora longula | Claroideoglomus etunicatum | Gigaspora albida | CV (%) | |
| Total phenols (mg g-1) | 235.50 bc | 297.80 a | 265.20 b | 217.60 c | 6.8 |
| Total tannins (mg g-1) | 156.40 bc | 218.60 a | 182.10 b | 148.80 c | 9.6 |
| Total flavonoids (mg g-1) | 158.60 a | 154.60 a | 146.40 a | 19.60 a | 10.6 |
| Total proanthocyanidins (mg g-1) | 12.43 a | 10.71 a | 11.32 a | 10.55 a | 11.2 |
| Total saponins (mg g-1) | 5.50 a | 4.33 a | 3.77 a | 4.40 a | 28.1 |
| Gallic acid (g %) | 0.70 a | 0.41 b | 0.16 c | 0.12 c | 11.4 |
| AAO (mg g-1 of remaining DPPH) | 89.20 a | 59.80 b | 104.90 a | 91.60 a | 15.9 |
| Ellagic acid (g %) | 0.18 a | 0.17 a | 0.11 b | 0.05 c | 12.7 |
| Phosphorus (mg Kg-1) | 375.70 b | 203.70 c | 452.20 ab | 524.40 a | 16.6 |
| Soluble carbohydrates (mg g-1) | 8.48 a | 7.56 ab | 7.00 b | 5.03 c | 9.3 |
| Mycorrhizal colonization (%) | 45.50 a | 38.60 a | 41.80 a | 39.70 a | 19.7 |
| - | Phosphorus | Phenols | Tannins | Flavonoids | Proantho | Saponins | GA | EA | Colonization | Carbohydrates | AAO |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Phosphorus | - | -0.7515** | -0.7166** | -0.4422* | ns | ns | ns | -0.6655 | ns | -0.5569* | 0.56* |
| Phenols | - | - | 0.9541** | 0.5351* | ns | ns | ns | 0,5001* | ns | 0.4588* | -0.5502* |
| Tannins | - | - | - | ns | ns | ns | ns | ns | ns | ns | -0.6138* |
| Flavonoids | - | - | - | - | ns | ns | 0.4820* | ns | ns | 0.8046** | ns |
| Proanthocyanidins | - | - | - | - | - | ns | 0.4708* | ns | ns | 0.4918* | ns |
| Saponins | - | - | - | - | - | - | ns | ns | ns | ns | ns |
| GA | - | - | - | - | - | - | - | ns | ns | 0.6952** | ns |
| EA | - | - | - | - | - | - | - | - | ns | ns | ns |
| Colonization | - | - | - | - | - | - | - | - | - | ns | ns |
| Carbohydrates | - | - | - | - | - | - | - | - | - | - | ns |
The concentration of phosphorus in fruits of plants inoculated with G. albida was increased by 39.55%. This did not occur for the plants inoculated with A. longula and C. etunicatum (Table 1).
4. DISCUSSION
Similar to this study, higher phenol concentrations in the fruits of Olea europea were reported after inoculation with Rhizophagus intraradices [42]. Santos and collaborators [25] reported the same results in the concentration of total tannins in the stem bark of L. ferrea when established in the field and inoculated with A. longula. In contrast, Silva and collaborators [30] verified an increase in the concentration of this phenolic group in the leaves of L. ferrea, which was established seven months earlier.
The optimization of the antioxidant activity, improved by the inoculation in the fruits of L. ferrea, was not found in the fruits of Solanum lycopersicum L. mycorrhizal [43]. In contrast, leaves of Inga vera inoculated with A. longula [15] had antioxidant activity potentialized by mycorrhization.
The chromatographic analysis of the aqueous extract of L. ferrea indicated the presence of gallic acid (retention time = 7.89 min) and ellagic acid (retention time = 24.57 min) compounds with a similarity between ultraviolet absorption spectrum, for gallic acid and ellagic acid of fruits from plants inoculated with A. longula, respectively (1 and 2 in Figure 1). This corroborated the results of Ferreira and collaborators [38].
Probably, the gallic and ellagic acids of the fruits of inoculated L. ferrea were synthetized and quickly directed for biosynthesis of tannins [10, 44], whose production was favored in this study by mycorrhization (Table 1). Therefore, it is important to develop studies with fruits of plants with a longer time scale in the field. This will verify whether the contents of gallic and ellagic acids are optimized by mycorrhization by considering that the production and composition of these compounds vary according to the plant’s development [10].
As expected, the higher concentration of total tannins was correlated to the production of total phenols (Table 2), considering that such molecules belong to the group of phenolic compounds [10]. Even though mycorrhization did not optimize the production of gallic acid and flavonoids (Table 1), the synthesis of these compounds had a positive correlation to the concentration of soluble carbohydrates (Table 2), which confirms that some sugars are precursors in the biosynthesis of some secondary metabolites [10].
Mycorrhization was not improved by the production of other compounds such as flavonoids, proanthocyanidins and saponins in the fruits of L. ferrea. Similarly, in leaves of Mimosa tenuiflora, a legume native to Caatinga, the mycorrhization did not favor the production of flavonoids either [45]. In the stem barks of L. ferrea, which were established in the field for 13 months, the biosynthesis of proanthocyanidins could not be optimized by mycorrhization either [25].
Different to this study, seedlings of L. ferrea placed in a greenhouse and inoculated with G. albida had a higher leaf concentration of total flavonoids in relation to the control, non-inoculated seedlings [14]. In other species of Caatinga, the increased production of flavonoids provided an inoculation with AMF [15, 22, 29], which leads us to understand that the inoculation with AMF influences the production of secondary metabolites in relation to the dependence of species, age and plant part studied.
In this study, the production of soluble carbohydrates, not optimized by AMF, can be related to the drain of these compounds to the production of secondary metabolites, such as phenols and tannins, which had a higher concentration in our study and are formed from the intermediaries of the metabolism of carbohydrates [10]. In addition, plants with a higher production of carbohydrates had lower accumulation of phosphorus (Table 2). A nutrient which was probably used in the biosynthesis of phenolic compounds. Such behavior was not expressed in the mycorrhizal leaves of Commiphora leptophloeos (Mart.) J.B. Gillet, whose concentration of soluble carbohydrates was reduced in the plants which produced more phenols because of inoculation [15].
In relation to phosphorus production in the fruits of mycorrhizal L. ferrea, similar results were found in mycorrhizal Pogostemon patchouli Pellet with native AMF, whose inoculation led to a higher leaf concentration of phosphorus [27]. In addition, this was similar to the fruits of Solanum lycopersicum L., which were associated with Rhizophagus intraradices [43].
In this study, the AMF, which increased the production of phenols and tannins, were not the same as the one which provided the highest accumulation of P in the fruits (Table 1). Similar behavior for the accumulation of essential oils was recorded in the fruits of dill (Anethum graveolens L.) and coriander (Coriandrum sativum L.) [46, 47]. In contrast, the fleshy fruits of S. lycopersicum had a higher accumulation of phenolic compounds and phosphorus with the mycorrhizal plant [43]. In mycorrhizal plants, the mechanisms of phosphorus accumulation and the direction of this nutrient for the biosynthesis of phytochemicals are probably different in dry and fleshy fruits (Table 1).
The rate of mycorrhizal colonization was no different in the treatment of this work. Nevertheless, A. longula was more efficient at producing biomolecules, which is probably related to the development of fungus, as indications from other studies showed that fungi of this species are more efficient than isolates of C. etunicatum and G. albida [48]. This same behavior had been recorded in another study carried out with the stem bark of L. ferrea by Santos et al. [25], which was the first report of the effect of mycorrhizal inoculation on the production of biomolecules in dry fruits of legumes. Here the behavior proved different from the previous knowledge held about the fleshy fruits of mycorrhizal plants, such as tomatoes [43].
CONCLUSION
Therefore, the use of A. longula produced fruits of L. ferrea with high phenolic content and higher antioxidant activity. Therefore, such a technological strategy can be used to install cultures aligned with the guidelines recommended by the WHO to produce herbal medicines complementing the actions of traditional medicine. Our results indicate a need to develop tests to verify whether the medicinal action of mycorrhizal ironwood fruit extracts is higher.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No Animals/Humans were used for studies that are the base of this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The authors confirm that the data supporting the findings of this research are available within the article.
FUNDING
None declared.
CONFLICTS OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors would like to thank the FACEPE (Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco) for granting the main author with a scholarship; and, in addition, to the CNPq (Conselho Nacional de Pesquisa e Desenvolvimento Tecnológico) and CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior).


