All published articles of this journal are available on ScienceDirect.
Production and Characterization of L-Asparaginases of Streptomyces Isolated from the Arauca Riverbank (Colombia)
Abstract
Introduction:
L-asparaginase, is known as an anti-cancer agent, mainly used in acute lymphoblastic leukemia, which prevents the proliferation of tumor cells. This study shows that there are unexplored regions in Colombia that can be sources of obtaining this enzyme and that the optimization of the production of L-asparaginase from native isolates can be determined in the search for alternatives to commercial drugs.
Materials and Methods:
Selection and identification of Streptomyces among Actinobacteria isolated from the Arauca riverbank for L-asparaginase producers are described. In addition, the effect of carbon and nitrogen sources, pH, temperature and agitation rate are studied for L-asparaginase activity in liquid culture using Plackett-Burman design and Taguchi methodology. Kinetic characterization of a purified L-asparaginase and its cytotoxic potential are evaluated too.
Results:
Seven of seventy-eight actinobacterial strains were selected as L-asparaginase producing Streptomyces showing a high L-asparaginase/L-glutaminase ratio in liquid culture with lactose as substrate. The strain 112 identified as Streptomyces lacticiproducens was chosen for L-asparaginase production at these culture conditions: lactose 0.25%, L-asparagine 0,015%, malt extract 0,015%, pH 7.36, 32°C and 130 rpm. Enzymatic studies of the purified L-asparaginase showed that the optimal pH and temperature were 6 and 37.5°C, respectively. This purified enzyme had an IC50 of 36.74 µg/mL on THP-1 cells.
Conclusion:
S. lacticiproducens isolated from the Arauca riverbank is a new source for the production of a high activity L-asparaginase, creating expectation of its availability as a drug for the acute lymphoblastic leukemia treatment.
1. INTRODUCTION
According to the 2017 report of World Health Organization (WHO) , leukemia was one of the leading causes of mortality in the world after traffic accidents [1]. The American Cancer Society estimates that four out of every five children who have leukemia suffer from Acute Lymphoblastic Leukemia (ALL) [2]. In Colombia, it is estimated that every year, approximately five hundred people die between the ages of one and fifteen years, from different types of leukemia [3]. Based on the patient's prognosis, treatments for ALL include chemotherapy, steroids, intensive radiation therapy and bone marrow transplants [4]. However, chemotherapy is the most frequent treatment against ALL. Among the medicines, the enzyme L-asparaginase is the most used one [5]. L-asparaginase (L-asparagine amidohydrolase, EC 3.5.1.1) has an antineoplastic application that prevents the proliferation of cancer cells by deamination of the amino acid L-asparagine to ammonium ion and L-aspartic acid [6]. The decrease of this amino acid is essential for the synthesis of proteins of malignant cells causing its growth rate to decrease [6]. Currently, L-asparaginase obtained from Escherichia coli is used for patients diagnosed with ALL [7-9] however, prolonged administration of unpurified L-asparaginase, causes diseases related to the liver due to the presence of the L-glutaminase enzyme activity [10]. Some countries have chosen to produce this enzyme by genetic engineering in different microorganisms mainly of the genus Erwinia, an example is E. crysanthemi; the commercial drug known as Kidrolase® comes from the enzyme L-asparaginase produced by the microorganism mentioned above [10, 11]. This drug presents a high cost and therefore a limited use. Also, E. carotovora is another species used for producing L-asparaginase, which has a lower commercial value, without massive sales [12].
Therefore, due to the effectiveness of L-asparaginase as an anti-cancer agent, the search continues to obtain this enzyme through the use of microbial systems as it is considered an inexhaustible source of production at low cost/ high effectiveness and eco-friendly approach [12, 13]. Few microorganisms are used to produce L-asparaginase for use as a therapeutic enzyme with high activity at pH and physiological temperature [5]. The production of this enzyme has been reported in different types of bacteria, among them being Actinobacteria, specifically Streptomyces [4, 14]. These have the advantage of producing L-asparaginase in an extracellular manner [15, 16], with minimal or no L-glutaminase activity [5] and with the ability to show high activity at physiological conditions [15, 16], which favor a therapeutic application with reduced adverse effects.
Currently, some researchers have been concerned about the optimization of the production of L-asparaginase, taking into account the microorganism, culture medium, carbon and nitrogen sources, pH and incubation temperature [17-19]. The establishment of mathematical models that involve growing factors of the cells and the production of proteins foster optimal production of the enzyme [20]. In 2013, Thaer and Ellaiah showed that optimization of the concentration of the carbon and nitrogen source, pH and temperature are required for achieving an L-asparaginase production in Streptomyces comparable to E. carotovora L-asparaginase production [21]. However, few studies have established mathematical relationships between the variables mentioned above and the activity of the L-asparaginase obtained in liquid culture from Actinobacteria; this generates the need to have a predictive model when it comes to modifying the fermentation conditions for reduction of time and economic resources, which could lead us to optimize the production process. For this reason, the objective of this study was the optimization of L-asparaginase production of a strain selected from a bank of Actinobacteria and the characterization of such purified enzyme.
2. MATERIALS AND METHODS
2.1. Chemicals
The bacteriological agar used was from Oxoid (Hampshire, United Kingdom). The malt extract, peptone, lactose, maltose, inositol, xylose, ribose, yeast extract, L-asparagine, L-glutamine, glucose, β-mercaptoethanol and temed used were from ITW Reagents (Hannover, Germany). The L-asparaginase antibody was from GeneTex (Irvina, United States), Tris-HCl, Trichloroacetic Acid (TCA), Glycine, Ammonium Persulfate (APS), acrylamide and bisacrylamide were from Merck (Darmstadt, Germany). The MTT reagent ((3-(4,5-Dimethy lthiazol-2-yl)-2,5-Diphenyltetrazolium bromide)) was from Sigma Aldrich (Darmstadt, Germany). The positive control was L-asparaginase from E.coli (Medac, 10000 U, GmbH, Hamburg, Germany). The protein weight marker was Broad Range Protein from BioLabs (Ipswich, United States).
2.2. Biological Material
Seventy-eight strains of Actinobacteria (Actinobacteria strains bank, Bioprospection Group of the University of La Sabana) isolated from the Arauca riverbank that showed anti-fungal and antibacterial activity, were evaluated in the production of the enzyme L-asparaginase.
2.3. Selection of L-asparaginase and/or L-glutaminase Producing Strains
The selection of L-asparaginase/L-glutaminase producing strains was carried out by the plate assay developed by Gulati [22]. The agar medium ISP5 (Asparagine-Glycerol) was used, containing phenol red (0.009%) as an indicator.
2.4. Determination of L-asparaginase or L-glutaminase Activities in Liquid Culture
L-asparaginase activity was determined after fermentation and centrifugation of liquid media by Nessler method [5, 23]. The reaction contained 0.5 mL of supernatant after precipitation of cells by centrifugation, 0.5 mL of L-asparagine solution (0.04 M) and 0.5 mL Tris-HCl (0.05M, pH 8.4). The reaction was carried out at 37°C for 30 min. The reaction was stopped by adding 0.5 mL of trichloro acetic acid (TCA) (1.5M) and then centrifuged. The amount of ammonium cation released due to the reaction was colorimetrically determined by mixing 0.1 ml of supernatant, 3.7 mL of water and 0.2 mL of Nessler reagent. After that, absorbance at 405 nm was determined in a plate reader (BioTek Instruments, United States). The activity was expressed in enzymatic units (U) defined as the amount of enzyme that releases one μmol of ammonium per time unit (U = μmol /h) [23-25]. L-glutaminase activity was determined using L-glutamine (0.04 M) as substrate instead of L-asparagine.
2.5. Morphological Identification
The morphological characteristics of the Streptomyces were observed according to the method of Shirling and Gottlieb [25] in ISP2-agar media (malt extract-yeast extract), ISP3-agar (Oatmeal), ISP4-agar (starch-inorganic salts) and ISP5-agar (glycerol-asparagine) were compared with the International Streptomyces Project (ISP) [25]. Gram stain tests were performed to corroborate the morphological characteristics of Streptomyces [25].
2.6. Determination of the Effect of the Carbon and Nitrogen Source, pH, Temperature and Agitation Rate on the Production of L-asparaginase
Taking into account that the production of L-asparaginase is influenced by physical and chemical factors [26], six carbon sources at 1% (lactose, xylose, ribose, inositol and maltose) were evaluated in twenty-five selected strains with high production of L-asparaginase [21, 26, 27]. Glucose (1%) was used as a positive control and medium without carbon source as a negative control. Likewise, the effect of nitrogen source at 0.05% (meat extract, malt extract and peptone in combination with L-asparagine (0.05%)) [21, 26, 27] was evaluated. L-asparagine (0.1%) was used as a positive control and medium without a nitrogen source as a negative control. The enzymatic activity was evaluated by the Nessler methodology [28] in each one of the triplicate assays.
Using Plackett-Burman design [29] (Table S1), the carbon and nitrogen sources were evaluated as the main variables to understand the L-asparaginase activity. However, as an addition to this design, the effect of pH, the incubation temperature and the agitation rate were assessed to achieve the highest L-asparaginase activity. Consequently, two levels of each variable were performed to determine the effect of the five variables, carrying out twenty trials. The pH of the medium was adjusted at the beginning of the fermentation [5].
In order to obtain a considerable L-asparaginase activity in fermentation, the carbon and nitrogen source were optimized with the help of the Taguchi methodology [30] (Table S2). In this study, nine tests were conducted with an orthogonal array L9 [30-32]. The models were studied by Analysis of Variance (ANOVA) [28, 33].
In the experiments, the L-asparaginase activity (Y) was taken as the response variable. The coefficients of the second-order polynomial were calculated using the software Statistical Analysis System “SAS 9.4” (Raleigh, North Carolina, United States) [34], generating the following mathematical model of L-asparaginase activity for the selected strains. (Equation 1).
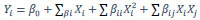 |
(1) |
2.7. Mathematical Modelling of L-asparaginase Production
To describe bacterial growth, the kinetic parameters
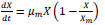 |
(2) |
Also, the m and n kinetic constants of the Luedeking-Piuret model for the production of L-asparaginase were graphically calculated [36, 37].
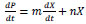 |
(3) |
In these equations,
The supplementary material contains the integrated equations of the models and the graphics, which were used to determine the kinetic constants and parameters.
2.8. L-Asparaginase Purification
The selected strain was inoculated into 200 mL lactose 0.25%, L-asparagine 0.015%, malt extract 0,015%, at pH 7.36, 32°C and 130 rpm for seven days in a 1L Erlenmeyer flask. Biomass-free fermented culture broth after centrifugation was taken (200 mL), filtered with 0.22 μm pore size membranes (Sartorius, Goettingen, Germany) and lyophilized (Labconco, Kansas City, United States) at -56°C and 0.140 mBar for 48 hours. The lyophilized fraction was suspended in 10 mL of Tris-HCl buffer at 0.05 M pH 8.6 and dialyzed overnight against the same buffer. After dialysis, the amount of protein in the samples was determined using Bicinchoninic Acid (BCA) as a reagent for protein quantification and using a standard of Bovine Serum Albumin (BSA) (2 mg/mL). The enzyme assay was made using the Nesslerization method as mentioned before [28], and the dialyzed sample stored at 4 °C for further purification. The samples were filtered with syringe membranes filters of 0.22 μm (Sartorius, Goettingen, Germany), and passed on a chromatographic DEAE cellulose column which was pre-equilibrated with buffer solution Tris-HCl at 0.05 M pH 8.6. The column was washed with two column volume of the above buffer and the adsorbed protein was isocratic eluted using a 0.5 M NaCl in 0.05 M Tris–HCl (pH 8.6). Fractions were collected at a flow rate of 0.8 ml min−1 before elution and 1 ml min-1 after elution (each fraction containing 3 mL) using BioLogicTM LP Starter Kit from Bio-Rad (Hercules, United States). The chromatographic runs were monitored for protein at UV (280 nm). The fractions were collected and examined for enzyme activity and protein content by procedures described earlier [28]. Fractions that showed a high activity of L-asparaginase were stored for later use [25, 38]. SDS-PAGE electrophoresis was performed according to the Laemmli method [38], with a 12% separation acrylamide gel and 4% stacking gel. The gel was stained with Coomassie brilliant blue R-250. The presence of L-asparaginase in the fractions was checked by western blot using a polyclonal antibody against L-asparaginase (GeneTex, Inc. United States). As a positive control, the L-asparaginase from E. coli was used (Medac, 10000 U, GmbH, Hamburg, Germany). Moreover, an Immobilon PVDF membrane (Merk, Bilerica, United States) was used, and the transfer was carried out in a Semi-Dry Blotter (Semi-Dry Blotting Systems EU-4000, C.B.S. Scientific Company INC, USA) at 100 V for 1 h. The membrane was incubated for 1 h in blocking buffer (TBST and 5% skim milk). Three washes were performed with TBST for three minutes each one and incubated with the antibody at 4°C for 12 hours. After this time, three washes were made with TBST for 5 min each one and Luminata Crescendo Western HRP Substrate (Merk, Belerica, United States) was added for three minutes. The membrane was revealed in MyECL imager (Thermo Fisher Scientific, Waltham, United States) [39, 40].
2.9. L-Asparaginase Characterization
The purified enzyme was incubated in 0.05 M Buffer solution from pH 4 to 10 and its activity was determined using the Nessler reagent. The buffer solutions used were: PBS (pH 4.5-7.5), Tris-HCl (pH 8.5) and glycine-NaOH (pH 9.5-10.5). For optimal temperature determination, the enzyme was incubated at temperatures from 25ºC to 60ºC in the buffer solution previously established [28]. The constant (Km) and maximal velocity (Vmax) was determined for the enzyme using the Michaelis-Menten kinetic model. Vmax and Km values were determined using nonlinear regression with SimBiology by Matlab (Version 2018b, Mathworks, United States) [28].
2.10. Cell Culture
For the cytotoxicity assay of the purified L-asparaginase, the THP-1 (ATCC® TIB-202 ™ - Acute monoblastic leukemia) and MRC-5 (ATCC® CCL-171 ™ - Human lung fibroblast) cell lines were used. The THP-1 and MRC-5 cells were seeded in a 96 well cell culture plates at a concentration of 1x104 cells per well in 100 μL of DMEM medium and incubated for 24 hours at 37°C. Then, 5% of the purified L-asparaginase at different concentrations from 10-200 ppm was added to the culture in triplicate and treated for 48 hours [28]. Cell viability was evaluated by the MTT test [40], where 15 μL of the MTT reagent in PBS buffer solution was added and incubated for four hours at 37°C. The medium with MTT was separated and the crystals formed were solubilized with 100 μL of DMSO and the absorbance was measured at 570 nm in a plate reader (BioTek Instruments, United States). The percentage of cellular inhibition was determined with equation 10. A non-linear regression was plotted between the percentage of inhibition and the logarithmic concentration and the IC50 was determined using the GraphPad Prism software (Version 7, GraphPad Software, USA).
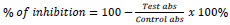 |
(4) |
3. RESULTS
3.1. Selection of L-asparaginase and L-glutaminase Producing Strains
Previously in the work of Arango et al. [38], seventy-eight isolates of the Arauca river banks in Colombia, belonging to the Streptomycetaceae were found to have antimicrobial activity. The isolation of the strains was made by pretreatments of the collected sediment river samples with calcium carbonate, phenol, thermal or heat, microwave and sonication. Those isolates showed both antibacterial and antifungal activity. The isolates produced metabolites with antibacterial activity against Methicillin-resistant Staphylococcus aureus-MRSA, Vancomycin-resistant Enterococcus faecium-VRE and extended-spectrum β-lactamase Klebsiella pnumoniae, together with antifungal activity against clinical isolates of Cladosporium cladosporioides and Epicoccum nigrum. Due to that, an analysis to determine an L-asparaginase production was performed on all these strains. According to other studies, high production of secondary metabolites could lead to better production of enzymes [38]. Therefore, from these results, 25 strains were highlighted due to high L-asparaginase/L-glutaminase activity presenting the production of the ammonium cation by the enzymatic action, which was determined by the color changing of phenol red indicator in the medium. The phenol red indicator was chosen because it does not inhibit the growth of Streptomyces strains [36].
3.2. Determination of L-asparaginase and L-glutaminase Activities in Liquid Culture
Through Nessler method, strains were evaluated to quantify L-asparaginase and/or L-glutaminase activity [41], showing that strains 5, 67, 112, 126, 220, 276 and 326 had higher activity of L-asparaginase than L-glutaminase from all 25 strains studied (Table 1); these results were expressed in enzymatic units of activity per mL (U/mL). These strains were chosen to continue with the research.
| Strain | L-asparaginase activity (U/mL) | L-glutaminase activity (U/mL) |
|---|---|---|
| 5 | 18.581±0.004 | 4.663±0.007 |
| 67 | 32.714±0.012 | 6.962±0.007 |
| 112 | 57.454±0.012 | 13.186±0.009 |
| 126 | 47.951±0.002 | 11.162±0.007 |
| 220 | 18.183±0.004 | 4.816±0.007 |
| 276 | 25.908±0.007 | 2.854±0.012 |
| 326 | 50.526±0.006 | 3.988±0.012 |
| 330 | 8.066±0.004 | 38.938±0.008 |
| 382 | 8.618±0.003 | 50.465±0.007 |
| 388 | 22.229±0.004 | 45.774±0.006 |
| 397 | 20.635±0.002 | 63.065±0.007 |
| 444 | 19.746±0.012 | 49.606±0.005 |
| 448 | 23.149±0.005 | 48.411±0.007 |
| 481 | 18.581±0.012 | 36.638±0.008 |
| 515 | 23.517±0.002 | 48.349±0.006 |
| 516 | 10.059±0.010 | 55.554±0.007 |
| 530 | 7.146±0.010 | 33.358±0.008 |
| 616 | 1.321±0.010 | 70.974±0.006 |
| 619 | 2.486±0.002 | 41.390±0.007 |
| 626 | 4.540±0.002 | 47.859±0.008 |
| 659 | 13.462±0.002 | 48.349±0.007 |
| 660 | 11.132±0.013 | 49.606±0.012 |
| 663 | 2.609±0.002 | 41.881±0.008 |
| 689 | 5.920±0.002 | 61.225±0.005 |
| 847 | 0±0.002 | 38.232±0.006 |
3.3. Morphological Identification
The characteristics of the seven selected L-asparaginase producing strains growing on different ISP agar media are described in detail in the supplementary material. Basically, all of the strains presented a satisfactory growth on ISP3 agar media (between 75 and 100% of the box area) with the production of diffusible pigments (except for the strain 326, which did not produce pigment), showing aerial mycelium and vegetative mycelium (except for the strain 326, which did not show vegetative mycelium). On the other ISP agar media, most of the strains grew between 51% and 74% of the box area, always producing aerial mycelium, but in some cases, the vegetative mycelium was not presentat all (for example, on ISP4). The production of diffusible pigments was not observed on ISP4 for any of the strains, whereas it was partial on ISP2 and ISP5 agar media.
The selected strains were Gram-positive (Fig. S1), showing the presence of hyphae with ramifications accompanied by fragments in the form of stick or coconut and formation of spores (see supplementary material), typical characteristics of the genus Streptomyces [42]. The selected Streptomyces are circular with regular and irregular borders growing on ISP agar media. They present pigmentation depending on the medium in which they are inoculated, which has been seen in other studies [43]. Each strain of Streptomyces presented above was identified by sequencing the 16S gene in a parallel study [38] and as previously reported, all strains showed an identity greater than 98.7% [44]. The strain 5 was identified as Streptomyces lunalinharesii, the strain 67 as Streptomyces puniciscabiei, the strain 112 as Streptomyces lacticiproducens, the strain 126 as Streptomyces phaeogriseichromatogenes, the strain 220 as Streptomyces phaeoluteigriseus, the strain 276 as Streptomyces griseofuscus and the strain 326 as Streptomyces mashuensis (Table S3).
3.4. Determination of the Effect of the Carbon and Nitrogen Source, pH, Temperature and Agitation Rate on the Production of the Enzyme L-asparaginase
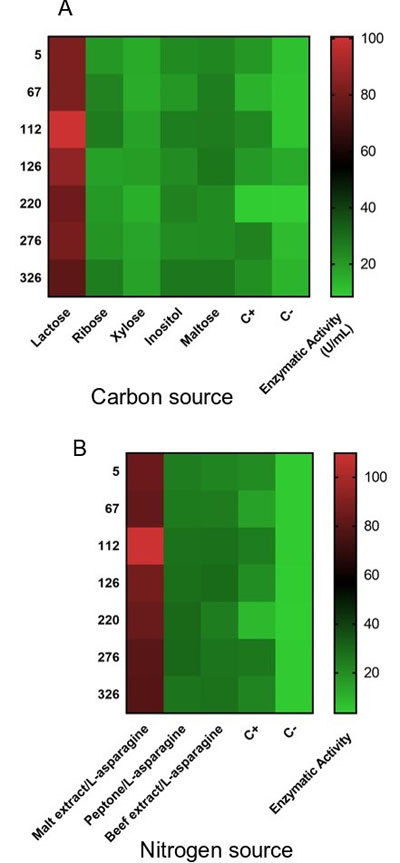
The production of ammonium cation was evaluated for each of the strains of Streptomyces with L-asparaginase activity in cultures with different carbon sources that were previously studied by different researchers [20, 33] (Fig. 1A). The seven strains showed higher L-asparaginase activity (U/mL) when lactose was used as a carbon source and for Streptomyces lacticiproducens (strain 112), its activity was 100.589 U/mL.
The effect of the nitrogen source on the activity of L-asparaginase (Fig. 1B) by the strains of Streptomyces showed a variation in the enzymatic activity depending on the source used. As a positive control, the amino acid L-asparagine was used. The liquid medium with malt extract and L-asparagine produced the highest L-asparaginase activity for each strain. Meena, et al. in 2015, reported that L-asparagine as a source of nitrogen stimulates the production of the enzyme [5].
According to (Table 2), all the models presented are acceptable to be represented as a curve-linear model due to their R2 have values close to 1, as desired. However, Streptomyces lacticiproducens (strain 112) has a behavior that indicates some troubles to be represented by this curve-linear model, as its R2 could be characterized as a low value to be classified by a good correlation. Moreover, the lack of fit of each model presented was represented to be higher than 0.05, therefore, all the models fit to the experimental data. Nevertheless, the lack of fit presented on strain 126 could lead a trouble to fit the model presented to the experimental data.
Through Plackett-Burman, twenty trials were implemented in the experimental design for each of the positive strains; the response to the experimental design was the activity of the enzyme L-asparaginase. Optimal values for each of the Streptomyces that maximize the activity of L-asparaginase are synthesized in (Table 3).
| Isolate | Parameters | ||||||||
| Lactose Concentration (%p/V) | Malt Extract/Asparagine Concentration (%p/V) | pH | Temperature (ºC) | Agitation Rate (rpm) | L-Asparaginase Activity Value Predicted (U/mL) | L-asparaginase Activity Experimental (U/mL) | Error (%) | ||
| 5 | 1.2519 | 0.1710 | 7.23 | 32.01 | 157.95 | 53.97 | 55.45 | 2.74% | |
| 67 | 1.4580 | 0.1720 | 7.22 | 31.72 | 155.02 | 59.24 | 61.18 | 3.27% | |
| 112 | 1.4460 | 0.1430 | 7.36 | 31.90 | 129.40 | 66.26 | 69.34 | 4.65% | |
| 126 | 1.4660 | 0.1640 | 7.24 | 31.72 | 155.02 | 54.33 | 55.21 | 1.62% | |
| 220 | 1.4620 | 0.1660 | 7.26 | 31.75 | 155.46 | 56.25 | 58.43 | 3.88% | |
| 276 | 1.4630 | 0.1650 | 7.25 | 31.74 | 155.65 | 57.45 | 62.31 | 8.46% | |
| 326 | 1.4340 | 0.1660 | 7.26 | 31.73 | 155.16 | 58.96 | 61.12 | 3.66% | |
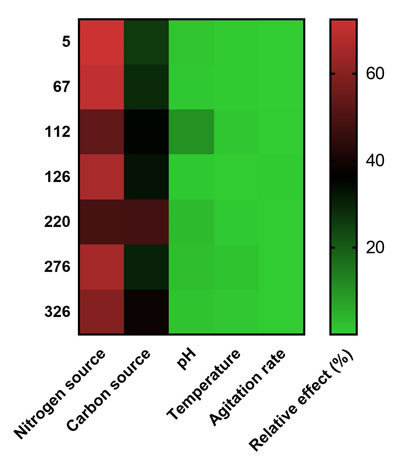
The Pareto chart (Fig. 2) illustrates the order of importance of the variables that affect the production of L-asparaginase in the Plackett-Burman experimental design. Among the variables evaluated, the most effective factors that positively affect the production of L-asparaginase, as indicated in the Pareto chart were the carbon and nitrogen source. These were identified as the most significant variables that affect the production of L-asparaginase and were selected for further optimization using the Taguchi methodology, which aims to optimize the process variables easily, economically and quickly since it requires fewer experiments in order to find the optimal condition as it can be seen in (Table 4) [30]. These values were adjusted to a mathematical relationship in order to adjust the values to a mathematical model; the other factors chosen previously in the Plackett-Burman design were kept constant in this part of the research.
It is observed how the behavior for the production of the L-asparaginase activity from the several strains of Streptomyces is different in each case. The evaluation of the Taguchi model was carried out by ANOVA. Significance was found in the regression for all strains at 95% confidence, which provides statistical evidence for an adequate correlation between the variables. It is observed how the activity (U/mL) is increased with the improvement of the process. According to protein concentration and enzymatic activity generated from each strain and comparing them with SDS-page electrophoresis (Fig. 3), it is observed that the enzyme related to S. lacticiproducens (strain 112) is highlighted more clearly than the other ones. Moreover, when all the strains were tested through chromatography column, an interesting phenomenon was observed; most of the enzymes reduced their activity considerably, except for the L-asparaginase from S. lacticiproducens. This enzyme after purification is the one increasing protein concentration and enzymatic activity. Therefore, due to these two parameters, protein concentration and enzymatic activity, S. lacticiproducens was selected as the best strain to produce L-asparaginase.
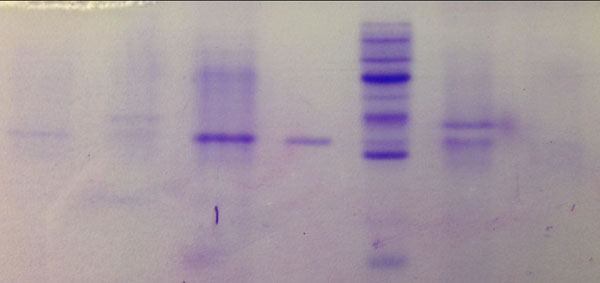
PC, Positive control. PM, Protein marker.
3.5. Mathematical Modelling of L-asparaginase Production
3.5.1. Microbial Growth
Logistic equation (Equation 2) was used for adjusting the data of growing of S. lacticiproducens (strain 112), for a later modelling of L-asparaginase production [45]. A significant increase in the production of L-asparaginase was observed after four days of fermentation and a maximum production was achieved after seven days, which begins after a gradual decrease of S. lacticiproducens biomass. A maximum amount of biomass 2.3
3.5.2. Product Formation
The production of L-asparaginase described by the Luedeking-Píret model and the kinetic parameters were evaluated using Equation 3 to a stationary phase (dX / dt = 0). The growth associated constant 'm' and the constant 'n', not associated to the growth were obtained (Fig. S3) as 291.75
These results showed that the constant associated with the degree of growth 'm' is greater than the constant not associated with non-growth, 'n'. Therefore, the production of L-asparaginase in this study was observed to be associated with bacterial growth. Matlab 2018b was used to estimate the parameters mentioned before using ‘lsqcurvefit’ function. The estimated parameters were used to test the model. The comparison of the predicted model with the experimental data of L-asparaginase activity obtained during seven days is shown in the supplementary material (Fig. S4).
3.6. L-asparaginase Purification
The crude extract filtered, lyophilized and dialyzed from S. lacticiproducens yielded 1.782 mg of protein/mL and 88.541 U/mL of L-asparaginase. The specific activity of this enzyme is about 49.695 U/mg of protein. After anion exchange chromatography, the collected fractions of L-asparaginase contained 0.312 mg of protein/mL and 83.789 U/mL of enzyme activity with a remarkable specific activity of 268.841 U/mg of protein. The specific activity of the purified L-asparaginase was found to be higher than Streptomyces fradiae NAES-82 [28] which is 30.63 U/mg of protein.
The molecular weight of the purified L-asparaginase of S. lacticiproducens was determined by using SDS-PAGE. It was found that the enzyme has an apparent molecular mass of 37 kDa. Only one distinctive band was observed and indicated of the homogenous form of L-asparaginase after enzyme purification (Fig. 4) in contrast with the several bands observed in the fermentation supernatant (Fig. 3).
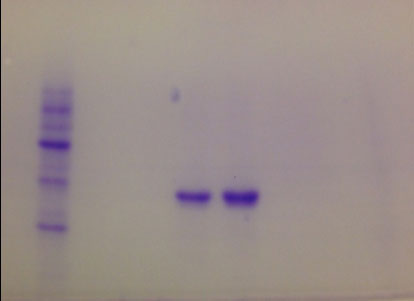
The presence of the enzyme was checked through Western Blot method, using L-asparaginase from E. coli as a positive control. A single band was obtained after development with a molecular weight of 37 kDa similar to positive control (Fig. S5). The other band observed was related to another subunit of L-asparaginase with a molecular weight of 75 kDa. It is important to say that an L-asparaginase antibody was used. This means that only L-asparaginase is revealed on the membrane.
Silver staining was used as a protein detector method. Thanks to its sensibility, proteins bind silver ions which can be reduced under appropriate conditions to build up a visible image made of finely divided silver metal [46]. As mentioned before, we have a single band on the purified protein from S. lacticiproducens and compared with a positive control, the same band range can be seen on the supplementary material.
3.7. Enzymatic Characterization
The enzyme showed maximum activity at pH 6 as it is shown in (Table 5). From pH 5.5 up to pH 6.0, the enzyme activity increases with a maximum activity of 97.27 U / mL (relative activity, 100%). At higher pH, the enzymatic activity decreases. The enzyme retains up to 23,1% activity at pH 8.5 compared to 24,8% at pH 7. Similar results were reported by Dhevagi and Poorani for L-asparaginase from Streptomyces sp. PDK7, which showed maximum activity between pH 6.0 and 8.5 [47, 48]. L-asparaginase is an amidase that is generally active and stable at neutral and alkaline pH [28] whereas, pH 5.0 to 9.0 were reported to be an interval for amidase activity L-asparaginase, purified from Streptomyces acrimycini NGP, which exhibited maximum activity at pH 6.0 [28].
The effect of temperature on the purified L-asparaginase activity is shown in (Table 5). It was active over a wide range of temperature conditions of 25-60 °C. The maximum activity of L-asparaginase was obtained at 37.5°C. At higher temperature, the activity of L-asparaginase decreased. Rahimzadeh et al. demonstrated that at 37°C, it is the optimal temperature for the activity of Bacillus PG03 [49]. However, the optimal temperature for the L-asparaginase activity obtained from Erwinia sp. is at 35°C [50].
Finally, regarding the Michaelis-Menten parameters, they were calculated as 0.0197 M for Km and 57.23 U/mL for Vmax taking into account the hydrolysis of L-asparagine. El-Naggar et al. in 2016 reported a Km value of 0.01007 mM and Vmax of 95.08 U/mL*min for purified L-asparaginase from Streptomyces fradiae NEAE-82 [28], it is important to remember that the lower the Km, the stronger the binding ability of the enzyme. Vmax is the limiting velocity as substrate concentrations get very large [28]
3.8. Cell Viability Assay
The THP-1 cells were exposed to diverse concentrations with E.coli L-asparaginase or with the purified enzyme of S. lacticiproducens (strain 112). It was observed that these present a decrease in living cells (metabolic active), compared to untreated cells after 48 hours of treatment, showing a good cytotoxic activity as the dose increases, which suggests that the L-asparaginase of S. lacticiproducens affects cell viability as it is seen in Table 6.
| pH | % of relative L-asparaginase activity | Temperature (°C) | % of relative L-asparaginase activity |
|---|---|---|---|
| 5.5 | 23.48 | 20 | 70,85 |
| 25 | 71,74 | ||
| 27,5 | 73,20 | ||
| 6.0 | 100 | 30 | 74,38 |
| 32,5 | 82,76 | ||
| 35 | 94,54 | ||
| 7.0 | 24,76 | 37,5 | 100,00 |
| 40 | 86,05 | ||
| 45 | 72,76 | ||
| 8.5 | 23,17 | 50 | 70,85 |
| 60 | 64,54 |
| Enzyme | THP-1 ATCC TIB-202 |
|---|---|
| IC50 Concentration (µg/mL) | |
| L-asparaginase from Streptomyces lacticiproducens | 36.74 |
| Medac® | 54.56 |
| Doxorubicin | 1.26 |
As it is shown in (Table 6), the enzymes present an IC50 between 1 and 60 µg/mL on THP-1 cell line. Comparing the IC50 obtained for the enzymes evaluated and doxorubicin on THP-1, it is evident that doxorubicin needs less concentration to cause the same cell damage than the L-asparaginase. Bhagat et al. in 2016 [51] reported that the purified L-asparaginase from Pseudomonas oryzihabitans showed cytotoxicity activity on MCF7-Breast carcinoma cell line and HeLa-Cervix carcinoma cell line with IC50 of 43.3 µg/mL and 50 µg/mL respectively, and Mahajan et al. in 2014 evaluated the cytotoxicity of purified L-asparaginase from Bacillus licheniformis, against Jurkat clone E6-1 with IC50 of 0.22 IU. All these reports are comparable with the results obtained with S. lacticiproducens.
4. DISCUSSION
Poorani in 2010 reported the L-asparaginase activity (2.7 U/mL*h) from Streptomyces EPD 27 without modification in fermentation [24]. Strains 5, 67, 112, 126, 220, 276 and 326, produced L-asparaginase at high levels, above 15 U/mL, but with a characteristic that they produce levels of L-glutaminase, lower than 20 U/mL. An addition to the morphological characteristics showed that Streptomyces phaeogriseichromatogenes and Streptomyces griseofuscus were reported as L-asparaginase producing strains. In 2010, Poorani [24] determined the L-asparaginase activity from crude extract (2.7 U/(mL*h)) [24]. However, strains such as Streptomyces lunalinharesii, Streptomyces puniciscabei, Streptomyces lacticiproducens, Streptomyces phaeoluteigriseus and Streptomyces mashuensis did not show antecedent as producers for L-asparaginase. Liu and Zajic in 1972 reported that lactose was the best carbon source to optimize the production of L-asparaginase from Erwina aroideae [52], however, there are few studies that reported lactose as the best carbon source for Streptomyces. In previous studies, it has been reported that L-asparagine is a suitable nitrogen source for the production of L-asparaginase by Streptomyces venezuelae, S. karnatakensis, S. collinus, S. gulbargensis and P. endphytica [37]. Several studies have shown that when using the combination of two nitrogen sources in the medium, the production of L-asparaginase increases [37, 53]. In the present study, it was observed that the microorganisms had a higher production of L-asparaginase in the media containing malt extract and L-asparagine for all sevens strains (Fig. 1B). In this case, Streptomyces lacticiproducens (strain 112) produced an enzymatic activity of 109.786 U/mL. Increasing the L-asparaginase activity was obtained when lactose was used as a carbon source and L-asparagine and malt extract were used as nitrogen sources. Through Plackett-Burman design, El-Naggar et. al in 2015 showed that an enzymatic activity of 49.874 U/mL was obtained at pH 7, and temperatures close to 30°C [33]. In our study, the enzymes produced by strains of Streptomyces showed higher enzymatic activities (around 60 U/mL) when the pH was around 7, the temperature was around 32°C and the agitation rate was between 130 rpm for isolate 112 and 155 rpm for the rest of the strains that were studied. After evaluation of the optimal concentration for lactose and L-asparagine/malt extract, S. lacticiproducens is the one with the highest enzymatic activity (86.4 U/mL). Therefore, through this parameter, S. lacticiproducens was selected as the better strain for producing L-asparaginase. Also, it was found that the production of L-asparaginase is related to the growth of the Streptomyces. Purification from the liquid culture was made when L-asparaginase was at its highest (seven days) comparable to El-Naggar et. al where they purified the enzyme from Streptomyces fradiae NAES-82 after seven days of culture [28]. The crude extract filtered, lyophilized and dialyzed from S. lacticiproducens had a specific activity for the enzyme of 49.695 U/mg of protein, while the recombinant E.coli W3110 L-asparaginase was 0.73 U/mg of protein [24]. After the elution of the enzyme from the DEAE cellulose anion exchange column, the specific activity of the enzyme was 268.841 U/mg of protein, which is higher than the specific activity reported for Streptomyces fradiae NAES-82 (30.63 U/mg of protein) [28]. The molecular weight obtained (37 kDa) was different from others reported in other microorganisms such as Streptomyces albidoflavus (28 kDa) [48], and Streptomyces fradiae NAES-82 (53 kDa) [28]. The optimal pH of the purified L-asparaginase from Streptomyces acrimycini NGP was 6 [28], which is the same as the L-asparaginase purified from S. lacticiproducen. On the other hand, the maximum activity of L-asparaginase was obtained at 37.5 °C, which is close to the optimal temperature for L-asparaginase produced from Bacillus PG03 [49]. Finally, Bhagat et al. in 2016 [51], reported that the purified L-asparaginase from Pseudomonas oryzihabitans showed cytotoxic activity on MCF7-Breast carcinoma cell line and HeLa-Cervix carcinoma cell line with IC50 of 43.3 µg/mL and 50 µg/mL respectively. The purified L-asparaginase from S. lacticiproducens (strain 112) showed cytotoxic activity on THP-1 cell line with IC50 of 36.74 µg/mL.
CONCLUSION
Seven strains of Streptomyces previously isolated from the Arauca riverbank were selected for the study of the effect of the carbon and nitrogen source, pH, temperature and agitation rate due to their high L-asparaginase/L-glutaminase activities ratio and mathematical models involving the effect of those variables. The strain 112 identified as S. lacticiproducens was selected as a producer of L-asparaginase in this study, taking into account the enzyme production achieved in liquid culture and the stability presented during the purification process. L-asparaginase specific activity increased almost 5.4 times after purification maintaining a very low L-glutaminase activity, and the purified enzyme showed an optimal pH and temperature of 6 at 37.5°C, respectively. Also, this purified enzyme had cytotoxic activity on TPH-1 cells, indicating that it can be a promising enzyme for ALL treatment.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
The authors gratefully acknowledges the Universidad deLa Sabana, for their financial support of this research (internalproject code Eng-158-2015) and for granting a scholarship toone of the authors.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors would like to thank to the staff members of microbiology and molecular laboratory of the Universidad de La Sabana for their help during this research.
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publishers Website along with the published article.


