All published articles of this journal are available on ScienceDirect.
Malaria Burden and Trend Among Clients Seeking Healthcare in the Western Region: A 4-Year Retrospective Study at the Sefwi-Wiawso Municipal Hospital, Ghana
Abstract
Background:
Malaria cases continue to rise despite sustained efforts directed at eliminating the burden among Ghanaians. This study was aimed at describing the spectrum of malaria burden in a four-year (2013-2016) retrospective review among clients seeking care at the Sefwi-Wiawso Municipal Hospital in the Western Region of Ghana.
Materials and Methods:
The study analyzed secondary data extracted on 32,629 patients who were referred to the Laboratory for malaria testing from January 2013 to December 2016. Socio-demographic data included age and gender, department of test requisition and malaria results were obtained from the archived Daily Malaria Logbook records. Approval for the study was granted by the authorities of the Sefwi-Wiawso Municipal Hospital.
Results:
The overall confirmed malaria case was 8629 (26.5%), among under five 1,384 (58.7%), pregnant women 4451 (20.3%) and 14.1% among asymptomatic population. Significant gender disparity in the confirmation of suspected malaria cases was observed with males recording higher rate (45.8%) than females (36.7%). The peak of the malaria epidemic was observed in the wet season (195 cases per month), compared to the dry season (133 cases per month).
Conclusion:
Cases of malaria is increasing with high rates among vulnerable groups in the Western Region. There is the need to intensify efforts to reduce the burden in the study area especially among vulnerable groups.
1. INTRODUCTION
Malaria infection continues to be of significant public health importance worldwide and in the developing world in spite of sustained global efforts targeted at eliminating the disease [1, 2]. According to a recent report by the World Health Organization (WHO), 212 million new cases of malaria were recorded worldwide in 2015, out of which, the Africa region accounted for about 90% of the global malaria cases [3]. In the tropics, malaria transmission is said to be seasonal and holoendemic, affecting all people especially children under five years and pregnant women who are most vulnerable to the disease [4-7]. Malaria infection is associated with a broad spectrum of clinical and laboratory manifestations and these may include fever, impaired consciousness, seizures, vomiting, diarrhoea, respiratory distress, anaemia, thrombocytopaenia, hypoglycaemia, metabolic acidosis, and hyperlactataemia among others [8-10]. Although Ghana and some West Africa countries are currently considered to be in the control phase of the malaria elimination program, malaria incident rate continues to rise annually as evident in the periodic reports of the National Malaria Control Program (NMCP)-an agency tasked with the responsibility of reducing the malaria burden in the country [11-13]. In Ghana, about 2.3 million suspected malaria cases were recorded at the Out-Patient Department (OPD) in the first quarter of 2017 representing a 1.2% increase over the same period in 2016 [13]. It is against this backdrop that in the present study, using a single site analysis, we aimed to describe the spectrum of malaria burden in a four-year (2013-2016) retrospective review among Ghanaians visiting the Sefwi-Wiawso Municipal Hospital in the Western.
2. MATERIAL AND METHODS
2.1. Study Design and Data Capture
The study analyzed secondary data of patients who reported at the Sefwi-Wiawso Municipal Hospital for care and were subsequently referred to the Clinical Laboratory for testing and confirmation during the study period (January 2013-December 2016). The period of data extraction was from 1st March, 2017 to 30th April, 2017. The study included a total of 32,629 male and female participants with information available for extraction from archival records. Socio-demographic data included age and gender, department of test requisition and microscopy results were extracted. A positive result included the presence of malaria parasite and density determination by counting the number of parasites against 200 white blood cells in the thick blood film. A negative result was defined by the absence of malaria parasite in at least 200 fields of the stained slide of patients’ blood in the thick blood film. Data on malaria results were obtained from the Daily Malaria Logbook records.
2.2. Study Area
Sefwi-Wiawso Municipal is one of the twenty-two (22) Metropolitan, Municipal Districts in the Western Region of Ghana. Wiawso is the Administrative Capital of the Municipality. The Municipality was established by Legislative Instrument (L.I) 2015 and shares boundaries with Bibiani-Anhwiaso-Bekwai to the east, to the west with Akontombra District, to the north with Asunafo South in the Brong Ahafo Region and to the south with Juabeso District. According to 2010 population and housing census, the population of the municipality stands at 139,200 with 69,753 males and 69,447 females.
2.3. Definition of Seasonality
The major and minor rainy seasons were defined as months of March to June and July, September to October respectively whilst the dry seasons were defined as the months of August, November, December, January and February.
2.4. Data Analysis
Data extracted was entered into Microsoft Excel 2013 spreadsheet for cleaning and validation. Data was described using frequencies and proportions. Differences between proportions and trends analysis were tested using Fisher exact test, Chi-square test and Chi-square test for trend where appropriate. A p-value < 0.05 was considered statistically significant. IBM Statistical Package for the Social Sciences version 22.00 (SPSS Inc, Chicago, USA; (www.spss.com) was used for data analysis.
3. RESULTS
In four years, under review, a total of 32,629 malaria tests requisitions were made, out of which 8629 (26.5%) were diagnosed as having the malaria parasite. In the general population, the review observed a significantly year-on-year increasing trend of malaria confirmed cases, after an initial dip in 2014, with the peak year in 2016 (31.9%). Among 29,526 females who reported during the review period, 7,208 (24.4%) were diagnosed of malaria while 1,421(45.8%) out of 3,103 of the male population were infected with the malaria parasite. In both genders, the peak malaria infection was recorded in 2016 (Table 1).
| Parameter | Total | 2013 | 2014 | 2015 | 2016 | p-value |
|---|---|---|---|---|---|---|
| Total Patients | 32629 | 7465 | 7140 | 7962 | 10062 | – |
| Malaria cases | 8629(26.5) | 2163(28.9) | 1353(18.9) | 1904(23.9) | 3209(31.9) | <0.0001 |
| Female Patients | 29526 | 7371 | 6892 | 7096 | 8167 | – |
| Malaria cases | 7208(24.4) | 2121(28.8) | 1259(18.3) | 1506(21.2) | 2322(28.4) | 0.1970 |
| Male Patients | 3103 | 94 | 248 | 866 | 1895 | – |
| Malaria cases | 1421(45.8) | 42(44.7) | 94(37.9) | 398(45.9) | 887(46.8) | 0.0555 |
Within the period of 2013-2016, 1,384 malaria cases were recorded among children under five years, accounting for 58.7% out of 2,356 attendance amongst this age group. Similarly, children within 5 and 12 years age bracket reported a high rate of malaria infection 510 (60.4%). The proportion of malaria infection recorded among patients of 13-19 years was 30.6%. The rate of malaria infection in the older years was below 30.0%. Irrespective of the age group, the prevalence of malaria rose from a trough in the second year of the review (2014) and reached at peak in 2016 (Table 2).
| Parameter | Total | 2013 | 2014 | 2015 | 2016 | p-value |
|---|---|---|---|---|---|---|
| Total patients | 2356 | 1621 | 234 | 717 | 1243 | – |
| Under 5 | 1384(58.74) | 91(56.17) | 123(52.56) | 407(56.76) | 763(61.38) | 0.0101 |
| Total patients | 844 | 59 | 94 | 222 | 469 | – |
| 5-12 | 510(60.43) | 35(59.32) | 40(42.55) | 145(65.32) | 290(61.83) | 0.0599 |
| Total patients | 2707 | 621 | 586 | 618 | 882 | – |
| 13-19 | 828(30.59) | 230(37.04) | 125(21.33) | 155(25.08) | 318(36.05) | 0.4201 |
| Total patients | 16469 | 4257 | 4123 | 4136 | 3953 | – |
| 20-29 | 3693(22.42) | 1202(28.24) | 732(17.75) | 786(19.00) | 973(24.61) | <0.0001 |
| Total patients | 8015 | 2111 | 1822 | 1803 | 2279 | – |
| 30-39 | 1606(20.04) | 540(25.58) | 286(15.70) | 296(16.42) | 484(21.24) | 0.0021 |
| Total patients | 2238 | 255 | 281 | 466 | 1236 | – |
| ≥ 40 | 608(27.17) | 65(25.49) | 47(16.73) | 115(24.68) | 381(30.83) | 0.0002 |
The majority of the case mix, 22,005 (67.4%), were observed among pregnant women attending Antenatal care and OPD 7,076 (21.7%) of the hospital. The percentage case confirmation was the highest among request received from the Children’s ward (60.5%), followed by OPD (39.9%) and Male ward (31.7%). People who voluntarily tested for malaria without any sign and symptoms of the disease (14.1%) and pregnant women as part of the routine screening of Antenatal care (20.2%) presented with the lowest percentage malaria burden (Table 3).
| Parameter | Department | Malaria |
|---|---|---|
| Antenatal care | 22,005(67.4) | 4,451(20.2) |
| Children's ward | 995(3.1) | 602(60.5) |
| Female ward | 908(2.8) | 255(28.1) |
| Maternity ward | 946(2.9) | 282(29.8) |
| Male ward | 635(1.9) | 201(31.7) |
| Out-patient department | 7,076(21.7) | 2,829(39.9) |
| Voluntary counseling and testing | 64(0.2) | 9(14.1) |
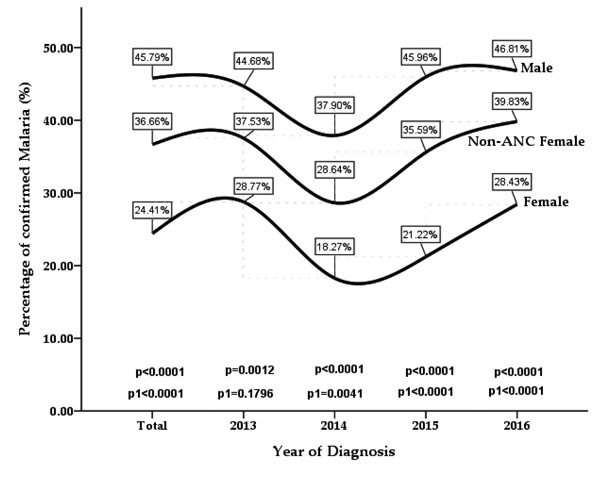
The percentage of malaria confirmed cases throughout the review period, ranged from 18.3% to 28.4% among the total female population, 28.6% to 39.8% in non-Antenatal female group and 37.9% to 46.8% among the male population. Significant gender variation in malaria infection burden was observed for the cumulative four-year period (p < 0.0001), with male patients recording a greater percentage of confirmed cases than their female counterparts. In general, significantly higher infection burden among the male group was observed year-on-year for the entire period of the review (2013, 2014, 2015 and 2016). Irrespective of gender, the infection density retarded in 2014 but peaked in 2016 (Fig. 1).
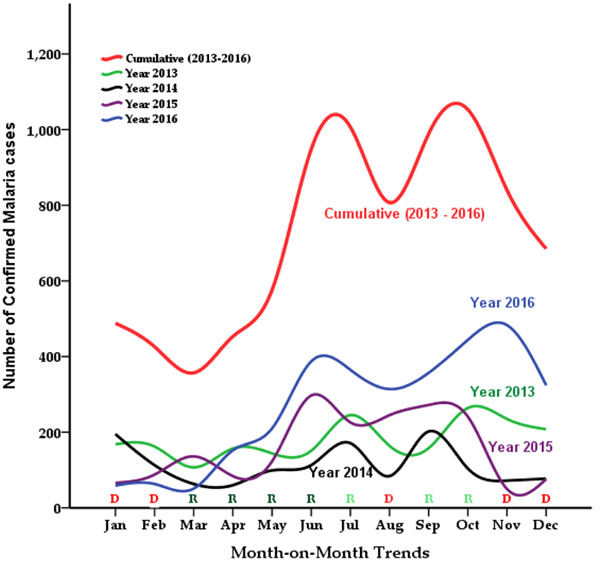
The modal points of the cumulative malaria burden within the period of the review were observed at the months of October (1,053 cases) and July (1,007 cases). The case difference between the trough month (March = 357) and the peak month (October) was in excess of 696 cases, representing a 194.9% increase in the case density. The general epidemiological pattern saw a rise in the infection burden each year at the month of June through to October and then a decline in the infection burden onward. However, In 2013 and 2014, there was a delay in the rise of cases in July (rainy season), and in 2016, there was a delay in the recession (Fig. 2).
4. DISCUSSION
Over the years, intense efforts have been made to curb the malaria menace in the Ghanaian population. However, the proportion of suspected malaria cases continues to rise in the country’s health facilities [12]. In the present study, a total of 32,629 patients were referred to the hospital’s laboratory for testing during the four (4) years under review (2013-2016). Among this study population, 8,629 participants recorded malaria infection, constituting an overall incidence of 26.5%. In addition to an increase in the proportion of infected patients, there was also an increase in the number of patients tested. Our results suggest a high burden of malaria among clients seeking medical care at Sefwi-Wiawso Municipal Hospital. Consistent with our finding, a recent study by Awine, Malm [11] reported that malaria morbidity accounted for about 40.0% of all outpatient attendance in Ghanaian health facilities. In a Ghana Demographic Health Survey (GDHS) report, malaria infection is said to range from 11.2% to 40.0% among Ghanaians hence rendering the country’s malaria situation highly endemic [14]. High malaria burden similar to the results of our study has been reported in other African countries. In Nigeria, Umaru and Uyaiabasi [15] reported an overall malaria infection rate of 35.7% among patients attending the General Hospital Makarfi in Kaduna-State. In Kenya, Jenkins, Omollo [7] recorded an infection rate of 28.0% in a study to ascertain malaria burden and its determinants in an endemic area of Kisumu County. The available data in the current review is limited in explaining the precise factors that could lead to the high malaria burden observed in our study. However, the transmission is shown to be driven by socioeconomic variables including poverty, unsanitary conditions, agricultural practice including irrigation, climatic conditions, availability of drugs and pesticides, use of mosquito bed nets, ignorance, poor behavioral attitudes towards the disease, migration and outdoor activities as well as human immunity among others [16-18].
In the present study, a significant gender disparity of confirmed cases was observed for the cumulative four-year period under review (p < 0.0001), with female patients recording a lower percentage of confirmed malaria cases than their male counterparts (Fig. 1). The incidence of malaria observed among the total female participants (24.4%) and female ward (28.1%) while in the male participants who reported at the facility during the review period, was 45.8% (Table 1). There is discordance in the literature on gender susceptibility to malaria infection. Male preponderance to malaria infection has been reported in previous studies, where males accounted for a greater proportion of malaria morbidity compared to the females [15, 19]. In contrast, other studies have also reported an overwhelmingly greater female susceptibility to malaria parasitemia than their male counterparts [7, 16].
Malaria remains number one cause of morbidity and mortality among children in Ghana and it is responsible for about 20% of childhood death [5]. Within the period under review, out of 2,356 children under five years with suspected malaria infection, 1,384 were confirmed accounting for 58.7% malaria infection amongst this age group (Table 2). Analysis of the departmental distribution of laboratory-confirmed malaria cases revealed the highest infection (60.5%) among patients from the Children’s ward (Table 3). Our findings, which were compared with results obtained by Kunihya et al. [16], indicate higher malaria susceptibility among the younger population compared to the teenage, twenty, thirty and forty age brackets with an observed incidence of 30.6%, 22.4%, 20.0%, and 27.2% respectively. Among the plausible explanations to this phenomena of higher malaria burden is the attribution to lowered immunity among the young compared to matured people. In areas of stable malaria endemicity, human immunity could develop over several years of exposure to the parasite, a phenomenon which could protect or reduce the risk of malaria infection in the older population [7, 16].
Of particular interest in the present study is the high rate of malaria parasitemia observed in pregnant women and in people who voluntarily tested for malaria at the facility. The malaria incidence among pregnant women who underwent routine antenatal screening was 20.2%, whereas in voluntary individuals, asymptomatic malaria was 14.1%. Asymptomatic malaria infection is quite common among Ghanaians. Kiptoo [20], recorded an infection rate of 5.5% among Antenatal Clinic attendees at Ridge Regional Hospital in Accra, Ghana. Atelu [21] in Navrongo reported Plasmodium falciparum carriage of 13.9% among an asymptomatic population. The asymptomatic malaria situation could render efforts aimed to control and eliminate malaria difficult, in that the parasites could remain undetected with the potential of silent transmission to other people during favourable seasons [22]. Malaria in pregnancy is associated with adverse maternal and fetal outcome. In pregnancy, sequestration of Plasmodium species in the placenta has been linked with low birth weight, pre-term delivery, miscarriages and stillbirths [23-27].
Irrespective of the age group, the prevalence of malaria rose from a trough in the second year of the review (2014) and peaked in 2016. The yearly trend of malaria infection, thus presented a U-shape epidemiological curve. Increasing malaria infection has been reported in Ghana [6]. Among factors postulated to account for the increasing national malaria burden among Ghanaians as captured in the 1st Quarter National Malaria Control Surveillance Bulletin–Issue 5, 2016, is the upsurge in the number of reporting health facilities in the District Health Information Management System (DHIMS)- a nationally adopted disease data capturing tool [12]. However, in this single site review, this explanation would not be plausible. In other jurisdictions, Roy and Khatun [28] in a longitudinal study have reported a decreasing malaria infection pattern among Asians from 2006 to 2011. In their opinion, the decreasing malaria phenomenon might be due to the substantial improvement made in the effort to combat malaria infection in the region.
Malaria transmission is perennial in tropical countries with different seasonal trends; the peak transmission period coincides with the period of major rains whereas the dry season records low malaria infection rate [16, 29, 30]. In Ghana, the major rainy season commences in March or April and runs until mid-July followed by a short dry period in July–August and the minor rainy season in September–October as well as a long dry season (harmattan) usually lasting from November through March [31]. Generally, the study recorded the highest number of malaria cases during the periods coinciding with the rainy seasons compared to those in the dry seasons over the review period (Fig. 2). The seasonal trend of malaria cases with a higher rate in the wet season observed in this study could be attributed to the relative increase in the number of breeding grounds for the Anopheles mosquitoes, increased oviposition by the vector leading to an increase in malaria parasitaemia during rainfall seasons, an assertion also corroborated by Egbendewe-Mondzozo, Musumba [32] and Darkoh, Larbi [33]. However, it is believed that extreme rainfall can reduce malaria infection rate by washing away mosquitoes from their breeding grounds hence reducing the numbers of disease vectors [33-35].
The present study has a limition due to its its cross-sectional design, and data reviewed did not take into consideration the parasite density and clinical symptoms, which may define the severity of the disease.
CONCLUSION
Malaria infection remains endemic among Ghanaians in the Western Region and with high rates among vulnerable groups (Children and pregnant women) in the Western Region. The trend points to seasonal variations tilted toward hyper infection in the wet seasons. There is therefore the need to intensify efforts to reduce the burden in the study area especially among vulnerable groups.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Approval for the study was granted by the authorities of the Sefwi-Wiawso Municipal Hospital.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Analysis of data was anonymous and non-linked, and no names and personal identifiers data were retrieved from the archives.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


