All published articles of this journal are available on ScienceDirect.
Antifungal Action of Methylene Blue Involves Mitochondrial Dysfunction and Disruption of Redox and Membrane Homeostasis in C. albicans
Abstract
Candida albicans is known to cause infections ranging from superficial and systemic in immunocompromised person. In this study, we explored that the antifungal action of Methylene blue (MB) is mediated through mitochondrial dysfunction and disruption of redox and membrane homeostasis against C. albicans. We demonstrated that MB displayed its antifungal potential against C. albicans and two clinical isolates tested. We also showed that MB is effective against two non- albicans species as well. Notably, the antifungal effect of MB seems to be independent of the major drug efflux pumps transporter activity. We explored that MB treated Candida cells were sensitive on non-fermentable carbon source leading us to propose that MB inhibits mitochondria. This sensitive phenotype was reinforced with the fact that sensitivity of Candida cells to MB could be rescued upon the supplementation of ascorbic acid, an antioxidant. This clearly suggests that disturbances in redox status are linked with MB action. We further demonstrated that Candida cells were susceptible to membrane perturbing agent viz. SDS which was additionally confirmed by transmission electron micrographs showing disruption of membrane integrity. Moreover, the ergosterol levels were significantly decreased by 66% suggesting lipid compositional changes due to MB. Furthermore, we could demonstrate that MB inhibits the yeast to hyphal transition in C. albicans which is one of the major virulence attribute in most of the hyphal inducing conditions. Taken together, the data generated from present study clearly establishes MB as promising antifungal agent that could be efficiently employed in strategies to treat Candida infections.
INTRODUCTION
Candida albicans is a commensal organism which generally resides within human body, but causes superficial and systemic infections during immunocompromised conditions [1]. Some of the predominantly used antifungals against this pathogen include azoles, polyenes, allylamines and echinochandins. Despite the ever increment in cache of numerous antifungal of the above classes, they are unable to meet the requirement of adequate antifungal therapy to warfare against the Candida infections in the patients because of their enunciated side effects [2-4]. Moreover, due to undue augmentation in the usage of these drugs, the leading intricacies in the treatment of patients are development of Multidrug resistance (MDR) apart from severe side effects, high cost, lesser efficiency [5]. Thus the remedies to exterminate these fungal foes are getting hampered and, there comes an urgency to develop newer effective drugs to fight against these fungal pathogens.
The limited repertoire of antifungals has nowadays shifted the research interest to either synthetic or natural drugs. Methylthionine hydrochloride or 3,7-bis (dimethy-lamino) phenothiazin-5-ium chloride commonly known as methylene blue (MB,) the very first lead chemical structure of phenothiazine and other derivatives, is used as dye in biological sciences having lower toxicity, high photo stability and could be easily eliminated from the body [6]. It was the first synthetic compound that was used as a drug [7] and was found to be effective as antimalarial compound [8, 9]. MB has been used for diagnostic procedures and the treatment of multiple disorders; including methemoglo-binemia, cyanide and carbon monoxide poisoning [10, 11]. MB has also been used as antifungal against murine models of C. albicans induced vaginal candidiasis by applying photodynamic therapy [12, 13]. However, mechanistic insights into the antifungal action of MB against C. albicans have not yet been fully elucidated. In this study, we have attempted to find out the mechanism of antifungal action for MB against the human fungal pathogen C. albicans and its clinical isolates. We also explored that MB is efficiently active against Candida tropicalis and Candida kruzei as well. We have further demonstrated that antifungal action of MB is possibly due to mitochondrial dysfunctioning and alteration in redox status and membrane homeostasis which is also the target of many known antifungals.
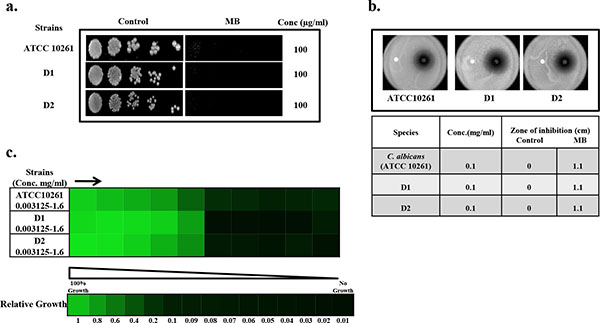
Drug susceptibility assays against C. albicans in the presence of MB.(a) Spot assay of C. albicans reference strains (ATCC 10261) and clinical isolates (D1, D2) in the absence (control) and presence of MB. (b) Disc diffusion assay against C. albicans reference strains (ATCC 10261) and clinical isolates (D1, D2) of C. albicans and their respective zone of inhibitions in the absence (control) and presence of MB. For control, discs were spotted with the solvent of MB (water) (c) Broth microdilution assay to determine the MIC80 of C. albicans reference strains (ATCC 10261) and clinical isolates (D1, D2) in presence of MB. Data is quantitatively displayed with colour (see colour bar), where each shade of colour represents relative optical densities of the cell. The minimum drug concentration that inhibits growth by 80% relative to the drug-free growth control is indicated as MIC80 for each strain.
MATERIALS AND METHODS
All Media chemicals YEPD (Yeast Extract Peptone Dextrose), Agar, Horse Serum, n- heptane, was purchased from Himedia (Mumbai, India). Methylene Blue (MB), Sodium Chloride (NaCl), Potassium Chloride (KCl), Mannitol, di-Sodium Hydrogen Orthophosphate, Potassium Di-hydrogen Orthophosphate, di-Potassium Hydrogen Orthophosphate, Sodium Hydroxide, D-Glucose, Sodium Dodecyl Sulphate (SDS) were obtained from Fischer Scientific.
Growth Media and Strains Used
The reference strain of C. albicans used in this study was ATCC 10261. The clinical isolate strains of C. albicans were D1, D2, and non albicans species including D9 (Candida tropicalis), and D46 (Candida krusei). All the strains of C. albicans were cultured in YEPD broth with the composition of yeast extract 1% (w/v), peptone 2% (w/v) and dextrose 2% (w/v). For agar plates 2% (w/v) agar (Himedia, Mumbai, India) was added to the media. For morphological switching the growth media used were serum (10% serum in YEPD), SLAD (0.17% yeast nitrogen base without amino acids and ammonium sulfate, 2% glucose, 50µM ammonium sulfate, 2% Bacto Agar) and spider media (1% mannitol, 0.4% K2HPO4, 1% Nutrient Broth). All Candida strains were stored in 30% (v/v) glycerol stock at -80˚C. The cells were freshly revived on YEPD broth and transferred to agar plate. The cells were grown at 30˚C on agar plate before each study to ensure the revival of the strains.
Drug Susceptibility Testing
Drug susceptibility was tested using spot assay, minimal inhibitory concentration (MIC) and filter disc assay as described below:
Spot Assay
Spot assays for the strains were determined using a method as described elsewhere [14, 15]. Briefly, for the spot assay 5μL of fivefold serial dilutions of each yeast culture (each with cells suspended in normal saline to an OD600 nm of 0.1) was spotted onto YEPD plates in the absence (control) and presence of the drugs. Growth was not affected by the presence of solvent used in the examination (data not shown). Growth difference was measured after incubation at 30˚C for 48 hours. The concentrations used in this study are specified in figure legends.
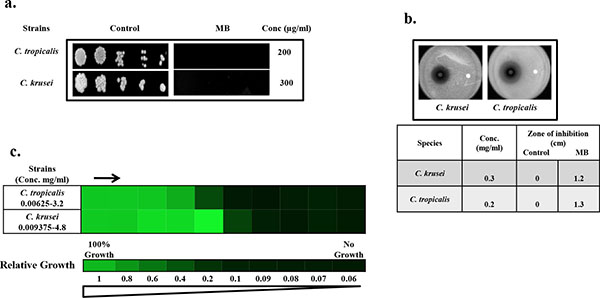
Drug susceptibility assays against non-albicans species of Candida in the presence of MB. (a) Spot assay of C. krusei and C. tropicalis; in the absence (control) and presence of MB. (b) Disc diffusion assay against C. krusei and C. tropicalis; and their respective zone of inhibitions in the absence (control) and presence of MB. For control, discs were spotted with the solvent of MB (water). (c) Broth microdilution assay to determine the MIC80 of C. krusei and C. tropicalis in presence of MB. Data is quantitatively displayed with colour (see colour bar), where each shade of colour represents relative optical densities of the cell. The minimum drug concentration that inhibits growth by 80% relative to the drug-free growth control is indicated as MIC80 for each strain.
Minimum Inhibitory Concentration (MIC)
MIC was determined by broth dilution method as described in method M27-A3 from the Clinical and Laboratory Standards Institute (CLSI) formerly NCCLS (National Committee for Clinical Laboratory Standards) [16]. Briefly, 100μl of media was placed at each well of the 96 wells plate following with the addition of the drug with the remaining media and then was serially diluted. 100μl of cell suspension (in normal saline to an OD600 0.1) was added to each well of the plate and OD600 was measured after 48 hours at 30˚C. The MIC80 was defined as the concentration at which the 80% of the growth was inhibited.
Filter Disc Assay
The filter disc assay was performed as described elsewhere [17]. The drug was spotted in a volume of 5-10μL at the indicated amount in the figure legends and the diameters of the respective zones of inhibition were measured after incubation of the plates for 48 hours at 30˚C.
Rhodamine 6G Efflux
The efflux of R6G was determined by using protocol described elsewhere [18]. Briefly, approximately 1x106 yeast cells from an overnight-grown culture in the absence (Control) and presence of MB at its sub inhibitory concentration (25µg/ml) were transferred to YEPD medium and allowed to grow for 5h. Cells were pelleted, washed twice with phosphate-buffered saline (PBS) (without glucose), and resuspended as a 2% cell suspension, which corresponds to 108 cells (w/v) in PBS without glucose. The cells were then de-energized for 1h in 2-DOG (5 mM) and 2,4 DNP (5 mM) in PBS (without glucose). The de-energized cells were pelleted, washed, and then resuspended as a 2% cell suspension (w/v) in PBS without glucose, to which R6G was added at a final concentration of 10μM and incubated for 40 min at 30°C. The equilibrated cells with R6G were then washed and resuspended as a 2% cell suspension (w/v) in PBS without glucose. Samples with a volume of 1 ml were withdrawn at the indicated time and centrifuged at 10,000 x g for 1 min. The supernatant was collected, and absorption was measured at 527nm. Energy dependent efflux (at the indicated time) was measured after the addition of glucose (2%) to the cells resuspended in PBS (without glucose). Glucose-free controls were included in all the experiments.
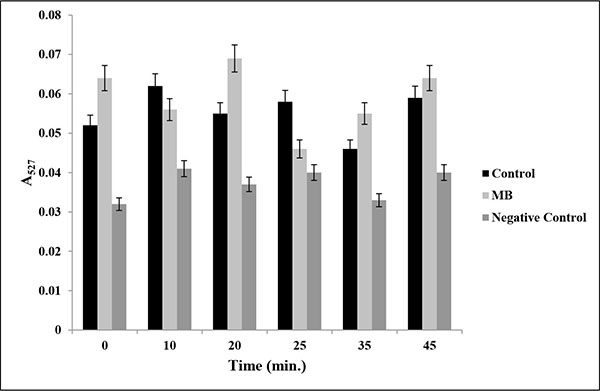
Drug efflux assay in presence of MB. Extracellular concentrations of R6G for C. albicans (ATCC 10261) cells grown in absence (control) and presence of MB (25μg/ml) calculated as described in material and methods. Negative control represents C. albicans de-energized cells without glucose. Mean of OD527 ± SD of three independent sets of experiments are depicted on Y-axis with respect to time (minutes) on X-axis. (P value > 0.05).
Transmission Electron Microscopy (TEM)
Damage to the cells treated with MB at its MIC80 value was observed by using TEM (Zeiss EVOMA10). The cells (~106 cells) were administered to the media with and without MB and were incubated for 24h at 30˚C. Sample preparation and analysis were performed by using the method as described elsewhere [19, 20]. Briefly, all cells were fixed with 2% glutaraldehyde in 0.1% phosphate buffer for 1 h at room temperature (20˚C). Washed with 0.1 M phosphate buffer (pH 7.2) and post-fixed 1% OsO4 in 0.1 M phosphate buffer for 1 h at 4˚C. Then the cells were dehydrated in acetone and dropped on round glass cover slip with hexamethyldisilizane (HMDS) and dried at room temperature then sputter coating with gold and observed under the TEM (Zeiss EVOMA10) at 30K magnification.
Quantitation of Ergosterol
Sterols were extracted by the alcoholic KOH method and the percentage of ergosterol was calculated as described previously [21, 22]. Briefly, a single C. albicans colony from an overnight YPD agar plate culture was used to inoculate 50 ml of YEPD in the presence and absence of MB. The cultures were incubated for 16 h with shaking at 30°C. The stationary-phase cells were harvested by centrifugation at 2,700 rpm for 5 min and washed once with sterile distilled water. The net wet weight of the cell pellet was determined to which 3ml of freshly prepared 25% alcoholic potassium hydroxide solution (25 g of KOH and 35 ml of sterile distilled water, brought to 100 ml with 100% ethanol), was added to each pellet and vortex mixed for 1 min. Cell suspensions were transferred to sterile borosilicate glass screw-cap tubes and were incubated in an 85°C water bath for 1 h. Following incubation, tubes were allowed to cool to room temperature. Sterols were then extracted by the addition of a mixture of 1 ml of sterile distilled water and 3 ml of n-heptane followed by vigorous vortex mixing for 3 min. The heptane layer was transferred to a clean borosilicate glass screw-cap tube and stored at -20°C. Both ergosterol and 24(28)-DHE absorb at 281.5 nm, whereas only 24(28)-DHE absorbs at 230 nm. Ergosterol content is determined by subtracting the amount of 24(28)-DHE (calculated from the OD230) from the total ergosterol plus 24 (28)-DHE content (calculated from the OD281.5). Ergosterol content was calculated as a percentage of the wet weight of the cells with the following equations: % Ergosterol + % 24 (28)-DHE = [(A281.5/290) ( F] / pellet weight; % 24(28)-DHE = [(A230/518) ( F] / pellet weight and % Ergosterol = [% ergosterol + % 24(28) DHE] - % 24(28) DHE, where F is the factor for dilution in petroleum ether and 290 and 518 are the E values (in percent per centimeter) determined for crystalline ergosterol and 24(28)-DHE, respectively.
Yeast to Hyphal Transition
Studies of hyphal induction on C. albicans were carried out on hyphal induction media like Spider (1% nutrient broth, 1% mannitol, 0.2% K2HPO4), 10% (v/v) horse serum and SLAD (0.17% yeast nitrogen base without amino acids and ammonium sulfate, 2% glucose, 50µM ammonium sulfate, 2% Bacto Agar). The dimorphic switching was performed using the protocol described elsewhere [23]. Briefly, the culture was grown overnight at 30˚C in YEPD broth before each study. The revived cells were harvested by centrifugation at 5000g rpm for 3 minutes and washed twice and incubated at 37˚C for 6h with PBS to induce starvation. After incubation, the cells were transferred to the required media for hyphal growth and hyphae were observed under microscope at magnification 40X.
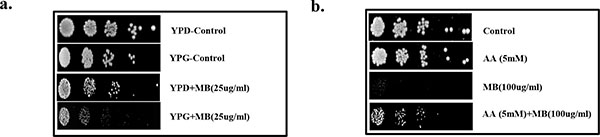
Effect of MB on mitochondrial function and redox status. (a) Spot assay demonstrating depleted growth of C. albicans in presence of MB (25µg/ml) when grown with glycerol as non fermentable source in contrast with the growth in presence of glucose. (b) Reversion in the growth of C. albicans at MIC80 concentration of MB when grown along with antioxidant AA at 5mM.
RESULTS
MB is an Efficient Antifungal Against C. albicans
To check the antifungal effects of MB against C. albicans, we first of all performed drug susceptibility testings. Three independent methods to check the drug susceptibility were performed viz. spot assay, filter disc assay and broth microdilution assay. From both spot assay and filter disc assay, it was confirmed that MB was efficiently acting as antifungal against C. albicans at 100µg/ml (Fig. 1a and 1b). Broth microdilution assay which determined the MIC80 value also showed similar result when performed against C. albicans (Fig. 1c). The antifungal activity of MB was further assessed against two clinical isolates of C. albicans referred as D1 and D2 and we found that both the strains displayed enhanced susceptibility to MB at 100µg/ml (Fig. 1). Thus, all the drug susceptibility testing results indicate that MB is inhibitory against reference as well as clinical isolates of C. albicans.
MB is Also Effective Antifungal Against Non-albicans Species of Candida
To further elaborate our study, we test whether MB has similar effects against non-albicans species of Candida as well. By performing the spot assay and disc diffusion assay, it was clear that MB was efficiently inhibiting the Candida tropicalis and Candida krusei when grown at the concentration of 200 and 300µg/ml respectively (Fig. 2a and 2b). Broth microdilution assay was also performed for the verification where the MIC80 values against non-albicans species were found to be in the range of 200-300μg/ml and correlates with spot and filter disc assays (Fig. 2c). However, Candida glabrata and Canddia parapsilosis remained resistant to MB even at concentration above 600μg/ml (data not shown).
Antifungal Action of MB is Not Linked with MDR Efflux Transporters
Overexpression of Multidrug Efflux transporter is one of the predominant mechanisms that is used by Candida to avoid antifungal drug action [24]. To study the role of major drug efflux pumps, if any, in the antifungal mechanism of MB, we performed Rhodamine 6G (R6G) efflux assay. R6G is well known substrate of efflux pumps and can be efficiently used to study the efflux activities. Our data suggest that there was no significant difference (P value > 0.5) in the extracellular R6G concentration in the absence (control) or presence of MB (Fig. 3).
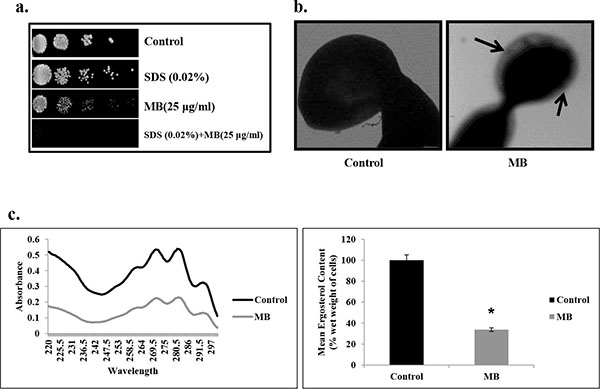
Effect of MB on membrane composition. (a) Spot assay demonstrating the attenuated growth of C. albicans in the presence of MB with membrane perturbing agent SDS (0.02% w/v). (b) Transmission electron micrographic image showing the tampered membrane integrity of C. albicans in presence of MB as described in Materials and methods. (c) Left panel shows UV spectrophotometric sterol profiles of C. albicans scanned between 220 and 300 nm from a culture grown for 16 hours with and without MB (25µg/ml). Right panel shows relative percentages of ergosterol content in the absence (control) and presence of MB (25μg/ml). Mean of % ergosterol levels is calculated as described in materials and methods normalized by considering the untreated control as 100 (absolute value of 0.002) ± SD of three independent sets of experiments are depicted on Y-axis and * depicts P value <0.05.
Antifungal Activity of MB is Linked with Mitochondrial Dysfunction and Altered Redox Status in C. albicans
MB has been previously shown to disrupt the mitochondrial integrity [25], therefore, we checked whether mitochondrial functioning of Candida cells is also affected in the presence of MB. For this, spot assays were performed in the media where glycerol is acting as sole non-fermentable carbon source in place of glucose (fermentable sugar) and it was observed that the growth of C. albicans was inhibited in the presence of MB in comparison to the cells grown in the presence of glucose (Fig. 4a). Next, we tested whether the observed disturbances in mitochondria is also linked with any alteration in redox status of the cell. For this, Candida cells were spotted in the presence of ascorbic acid (AA) which is a known antioxidant. We observed that the antifungal activity of MB could be rescued in the presence of AA which suggests that the antifungal activity of MB is also linked with the alteration in the redox status (Fig. 4b).
MB Disrupts Membrane Homeostasis in C. albicans
Since many of the current therapeutic drug regimes target the membrane integrity, hence we checked whether MB has any influence on the membrane integrity of C. albicans. Firstly, we performed the spot assay with SDS (membrane perturbing agent) and found that the C. albicans was unable to grow in the presence of MB when grown in SDS (Fig. 5a). This was further confirmed through TEM experiment which clearly depicts that MB disrupts the membrane integrity of C. albicans (Fig. 5b). Furthermore, we assessed whether the alteration in the membrane integrity due to MB could also have any influence on its membrane composition. We estimated the ergosterol levels and found that there was decrement in ergosterol content significantly by 66% (P value ≤ 0.05) in presence MB (Fig. 5c).
MB Hinders the Morphogenetic Switching in C. albicans
Yeast to hyphal switching is also one of the major factors which govern the virulence in C. albicans. Thus the effect of MB on this morphological switching was checked by providing various hyphae inducing conditions in liquid as well as solid media at 37˚C. We observed that filamentation was completely lacking in MB treated cells and appears only in yeast form in contrast to the untreated Candida cells suggesting that MB is an active inhibitor of morphogenetic switching for this dimorphic fungus (Fig. 6).
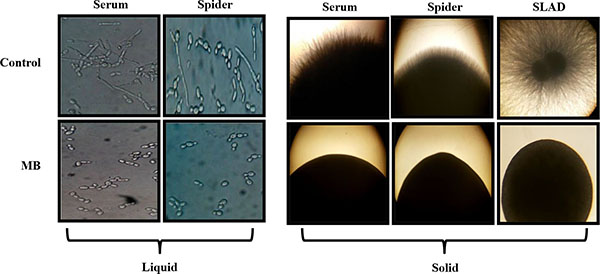
Effect of MB on Dimorphic Switching of C. albicans. Left panel shows Hyphal morphogenesis in the liquid hyphal inducing media (YEPD with 10% Serum and Spider media) in the absence (control) and presence of MB (25µg/ml) in the C. albicans (ATCC 10261) at 4 hours. Right panel shows hyphal morphogenesis in the solid hyphal inducing medium (Spider agar medium, 10% Serum and SLAD medium) in the absence (control) and presence of MB (25µg/ml) in the C. albicans (ATCC 10261) at 4 hours.
DISCUSSION
MB is already approved by US Food and Drug Administration (FDA) for acute acquired methemoglo-binemia, enzymopenic hereditary methemoglobinemia and prevention of urinary tract infections in aged patient and also for intra-operative visualization of nerves, nerve tissues, and endocrine glands as well as of pathologic fistulae. It is also used for the deterrence and treatment of neurotoxicity induction in cancer patients [26]. MB can be administered through various routes viz. orally, intravenously, intraperitoneally [27], intraduodenally [28, 29]. Despite having such diverse course of benefits for human as a drug, lesser studies have been conducted to elaborate the antifungal effects of MB. Thus, the present study was conducted with the aim to determine the antifungal effects against human fungal pathogen Candida albicans and also against its non albicans species viz. C. krusei and C. tropicalis. We proposed that MB acts as antifungal against human fungal pathogen C. albicans through dysfunctioning of mitochondria, altering redox status and disruption of membrane integrity to deplete the ergosterol level. We further, confirmed that MB inhibits the yeast to hyphal transition which is essential for survival and for virulence in the host body [30, 31].
Firstly, we demonstrated the antifungal activity of MB and found that MB was efficiently showing its anti-candidal effect not only against C. albicans but also for non albicans species of Candida. Three independent drug susceptibility testings; spot assay, filter disc assay and broth microdilution assay were conducted to demonstrate that MB was antifungal against C. albicans reference strain as well as clinical isolates at 100µg/ml Fig. (1) and inhibits the growth of C. krusei and C. tropicalis at 300 and 200µg/ml respectively (Fig. 2). Interestingly, MB showed no effect on C. glabrata and C. parapsilosis even upto 600µg/ml (data not shown) suggesting there might exists alternative mechanisms to cope up with MB stress. There are many reports which suggest that current therapeutic drugs clinically available are losing their effectiveness. For instance fluconazole shows its antifungal activity in C. albicans (ATCC10261) at 16µg/ml which goes up to the range of 85-110µg/ml for the resistant strains of Candida [32]. Similarly, MIC values for amphotericin B and caspofungin lies in the ranges 0.03-0.125µg/ml and 0.25-2µg/ml respectively [33, 34]. Moreover, a recent study suggests that resistance rates of Candida for amphotericin B, fluconazole, itraconazole, and voriconazole were 2.9%, 5.9%, 4.2% and 2.5%, respectively [35]. Under such circumstances, it becomes compelling to look for new drugs such as MB and explore their targets.
Our results prompted us to gain further insights into the inhibitory mode of action for MB. Since, one of the most common mechanisms of action for the MDR still remains the overexpression of efflux pumps [24], therefore, we first of all check the effect of MB on the MDR efflux transporter activity through R6G efflux assay. The data shows that MB was ineffective to inhibit efflux of R6G and there was no significant difference between the control and treated cells (Fig. 3). Thus, we hypothesized for existence of any alternate mechanism by which MB was showing its antifungal effects.
MB being a known inhibitor of mitochondrial respiratory chain (MRC) [7, 36] prompted us to check whether its antifungal activity is also linked with the mitochondria dysfunction. Thus we explored that MB antifungal action is linked with the disruption of mitochondrial integrity and found when grown in the presence of glycerol (the sole non fermentable carbon source) C. albicans cells were showing hypersensitivity to the MB (Fig. 4a). Mitochondria is most common organelle that acts as the important source for the production of reactive oxygen species (ROS) and any disturbances in functioning of mitochondria is often associated with altered redox homeostasis of the cell [31, 37]. Therefore, the mitochondrial dysfunction in presence of MB was further tested for its effect, if any, on redox status of the cell [38]. This was confirmed by performing spot assay in presence of AA, a known antioxidant. Interestingly we observed that the hypersensitivity of Candida cells could be rescued when spotted along with ascorbic acid (Fig. 4b). These results clarified that the inhibitory action of MB is associated with the disruption of mitochondria and change in the redox status of the cell. Considering the fact that fungal antioxidant system (superoxide dismutases, glutathione reductase, stress signaling pathway, etc.) plays a critical role in maintaining cellular integrity from toxic reactive oxygen species [39, 40], alteration in mitochondria functioning and redox status of the cell needs to be further validated.
Cell membrane is another crucial target for many of the known antifungals belonging to the class of azoles, polyenes and allylamines. This fact propelled us to study the influence of MB on the cell membrane of C. albicans. For this, firstly spot assay in the presence of known membrane perturbing agent viz. SDS was performed and found that C. albicans was showing hypersensitivity to MB in the presence of SDS (Fig. 5a). This observation led us to visualize membrane integrity through TEM experiment which further confirmed and corroborate with hypersensitivity to SDS (Fig. 5b). These results necessitated to find out whether the disruption in membrane integrity due to MB has any influence on its membrane composition. Hence we checked the ergosterol content of Candida cells which is an important determinant of the membrane structure. MB perturbs membrane structure and function was further evident from the fact that we observed 66% reduction in ergosterol level in the cells treated with MB (Fig. 5c). This establishes the fact that MB treatment to C. albicans leads to the disruption of membrane veracity leading to depletion in ergosterol level.
Finally we assessed the effect of MB on yeast to hyphal transition of C. albicans, a major virulence attribute to establish infection [41, 42]. Our results demonstrated that MB could efficiently inhibit the formation of hyphae in most of the hyphae inducing conditions such as spider media, serum and SLAD at 37°C in contrast to control cells which were able to express the filaments in similar conditions (Fig. 6).
CONCLUSION
Considering the ever-increasing burden of MDR which still remains a major concern for C. albicans, alternative therapeutic options such as MB that can be used as antifungal agents could be a wise strategy. Taken together, the present clearly establishes the antifungal nature of MB that can be exploited for improving the current therapeutic strategies.
ABBREVIATIONS
| 2,4 DNP | = 2,4 Dinitrophenol |
| 2-DOG | = 2-Deoxy D-glucose |
| MB | = Methylene Blue |
| R6G | = Rhodamine 6G |
| TEM | = Transmission Electron Microscopy |
| YEPD | = Yeast Extract Peptone Dextrose |
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
ACKNOWLEDGMENTS
S.H. thank for the financial assistance in the form of Young Scientist award (SR/FT/LS-12/2012) from Science and Engineering Research Board (SERB), New Delhi. We acknowledge the assistance of Jasvir Singh, IARI, New Delhi for assisting us in TEM experiments. We are grateful to Luqman A. Khan and Nikhat Manzoor, Jamia Millia Islamia, New Delhi for providing Candida reference strain as generous gift. We also thank Sumathi Muralidhar, Regional Sexually Transmitted Disease Research Centre, Safdarjung Hospital, New Delhi for helping us with clinical isolates of C. albicans and non-albicans strains. We thank Prof. S. M. Paul Khurana, Dean, Faculty of Science, Engineering & Technology for encouragement and providing the available facilities for research in the institute.


